Abstract
Background. Systemic lupus erythematosus (SLE) is an autoimmune disease that has been linked to the progression of periodontitis, negatively affecting patients’ quality of life. Further studies are required to determine the correlation between these conditions and to support the incorporation of periodontal control in patients with SLE.
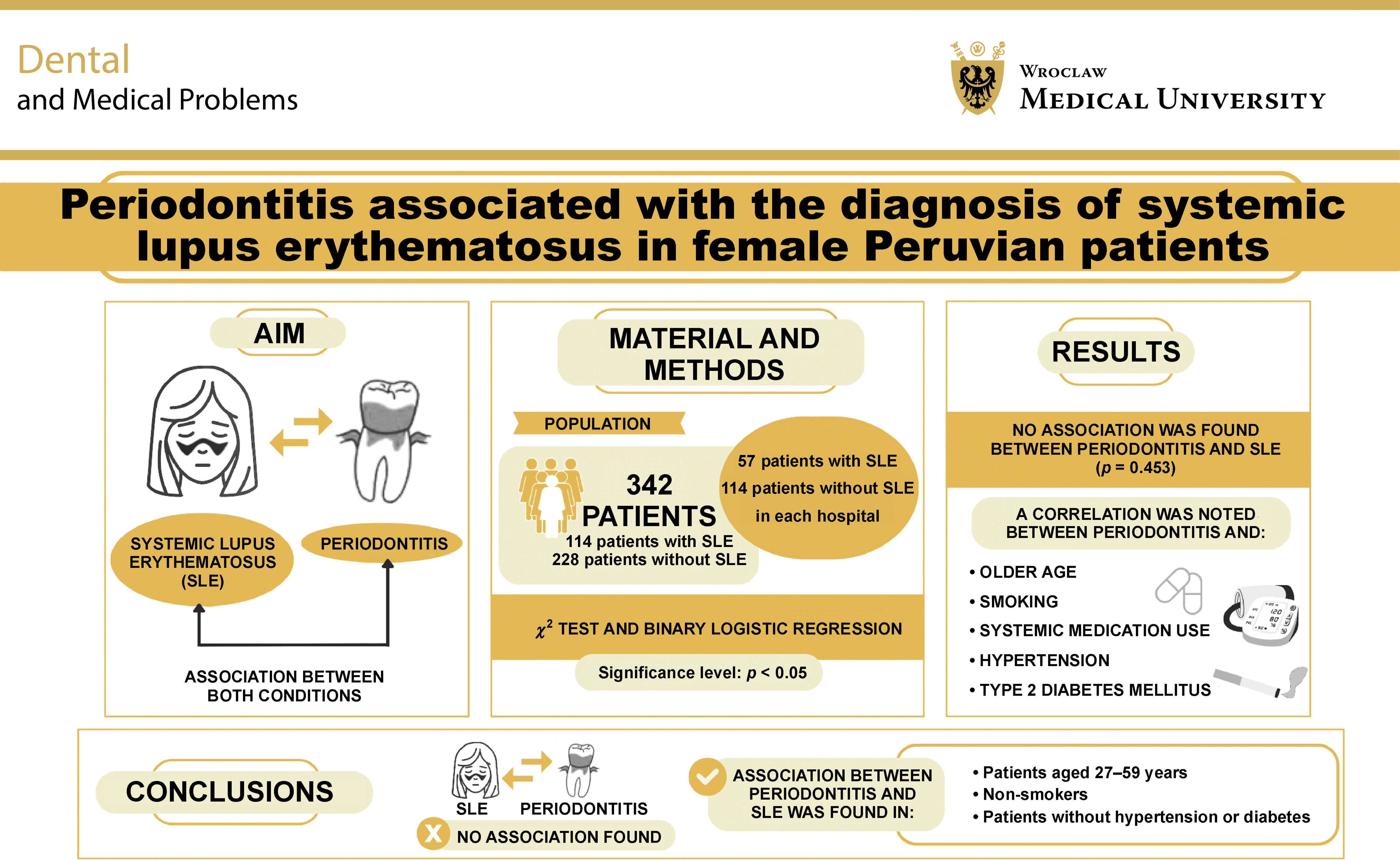
Objectives. The aim of the study was to determine the association between periodontitis and the diagnosis of SLE in adult female patients in Peru attending outpatient rheumatology consultations.
Material and methods. This cross-sectional study was conducted between July and October 2023 and included 342 patients from the Department of Rheumatology of the Regional Teaching Hospital of Trujillo, Peru, and Víctor Lazarte Echegaray Hospital, Trujillo, Peru. Intra- and inter-rater calibration for the clinical measurement of periodontitis yielded a κ value of 0.846 in both cases. The rheumatology specialist established the diagnosis of SLE. The χ2 test and binary logistic regression were used to analyze the results, with a significance level set at p < 0.05.
Results. No association was found between periodontitis and SLE (p = 0.453). However, correlations were noted between periodontitis and patients aged 27–59 years (p = 0.006), smoking (p = 0.035), systemic medication use (p < 0.001), arterial hypertension (p = 0.018), and type 2 diabetes mellitus (T2DM) (p = 0.003).
Conclusions. No association was established between periodontitis and SLE. However, correlations were observed between periodontitis and SLE in specific subgroups, including patients aged 27–59 years, non-smokers, and those without hypertension or diabetes. Furthermore, a higher prevalence of periodontitis was noted in older patients, smokers, those receiving systemic medication for SLE, and patients with hypertension or T2DM.
Keywords: risk factors, systemic lupus erythematosus, autoimmune disease, periodontal disease, periodontitis
Introduction
Periodontal disease is a multifactorial chronic inflammatory condition that represents a significant global public health problem, as it negatively affects patients’ quality of life1 and leads to progressive destruction of the tooth-supporting apparatus, making it a leading cause of tooth loss.2
The global prevalence of periodontal disease ranges from 20% to 50%.3 It occurs with greater frequency and severity in certain age groups, systemic conditions and educational levels.4 In this context, the disease produces a systemic inflammatory burden that may influence existing conditions such as arterial hypertension,5 diabetes mellitus, coronary heart disease,6 and cerebrovascular accidents.7
Systemic lupus erythematosus (SLE) is considered a prototypical autoimmune disease associated with increased disability and mortality.8 It is a complex condition that presents challenges for diagnosis and treatment,9 and is characterized by an exacerbated response of B and T cells, as well as a loss of tolerance to autoantigens. Defective production and clearance of antibodies, the circulation and deposition of immune complexes in tissues, as well as the activation of cytokines and complements contribute to clinical manifestations ranging from mild fatigue and joint pain to severe health damage.8
Typical manifestations of SLE include skin rash, malar (butterfly) rash, arthritis, pleurisy, serositis, alopecia, and lupus nephritis. The response to treatment is often variable and difficult to predict, likely due to the complex immune dysregulation involved in SLE pathogenesis.10, 11 Early diagnosis is crucial to prevent outbreaks and resulting tissue damage; however, delays in diagnosis remain common.10, 12
Although SLE has no cure, it can be effectively managed with pharmacological therapy, with the primary goals being the prevention of organ damage and the reduction of secondary comorbidities.13 Recommendations for SLE management were published by the European League Against Rheumatism (EULAR) in 2008 and updated in 2019 in collaboration with the American College of Rheumatology (ACR) based on emerging new data. These guidelines indicate that lupus patients can receive hydroxychloroquine, prednisone, methotrexate, azathioprine, mycophenolate, belimumab, rituximab, and cyclophosphamide.12
Oral manifestations are included among the diagnostic criteria for SLE. The prevalence of xerostomia, dental caries, mucositis, cheilitis, and oral mucosal ulceration ranges from 81.3% to 87.5% in patients with SLE. Additionally, up to 93.8% of patients with SLE present with periodontitis.14, 15
Periodontitis and SLE have been reported to share genetic and environmental risk factors. Systemic lupus erythematosus induces an imbalance between pro-inflammatory and anti-inflammatory cytokines, which contributes to tissue damage. It also activates autoreactive B cells and promotes dysregulation of other immune cells, including macrophages, neutrophils, CD4+ T cells, and dendritic cells, which may influence the subgingival microbiota.16, 17
It is estimated that at least 5 million people worldwide are affected by lupus in some of its manifestations.18 Several studies suggest a possible association between the pathology of SLE and periodontal disease19, 20; however, the available evidence remains limited. Therefore, the main objective of this study was to determine the association between periodontitis and the diagnosis of SLE in female Peruvian patients, and to provide information about the associated factors such as age group, smoking habits, use of systemic medication for SLE, hypertension, and diabetes. These findings may contribute to improving clinical care guidelines, emphasizing multidisciplinary collaboration between dentists and rheumatologists, and ultimately enhancing the quality of life of affected patients.
Material and methods
The present cross-sectional observational study was conducted at the Department of Rheumatology of the Regional Teaching Hospital of Trujillo, Peru, and Víctor Lazarte Echegaray Hospital, Trujillo, Peru, between July and October 2023.
The sample consisted of 342 patients, including 114 diagnosed with SLE and 228 with other rheumatological diseases. The sample size was calculated using a formula for group comparison, based on the data obtained from a pilot study conducted on 40 patients, using the following parameters: n1 – number of patients without SLE; n2 – number of patients with SLE; k = 2 (number of patients without SLE per patient with SLE); α = 0.05 (type I error); β = 0.20 (type II error, corresponding to 80% power); zα/2 = 1.96 (normal value with a type I error of 5%); zβ = 0.842 (normal value with a test power of 80%); p1 = 0.526 (prevalence of periodontitis among patients without SLE); p2 = 0.680 (prevalence of periodontitis among patients with SLE); and ε = 0.154 (difference in prevalence between groups). A non-probabilistic convenience sampling method was used, selecting the same number of cases from each hospital.
The study included female adults aged over 18 years who attended outpatient consultations in the rheumatology services of the aforementioned hospitals. Pregnant women, hospitalized women, and those who did not agree to participate were excluded.
The study protocol was approved by the Faculty of Human Medicine of Antenor Orrego Private University (Resolution No. 2598-2023-FMH-UPAO) and the Bioethics Committee of Antenor Orrego Private University (Resolution No. 0658-2023-UPAO), Trujillo, Peru, as well as by the Research Committee of the La Libertad–ESSALUD Healthcare Network (PI No. 121 CIYE-O.C.I.Y. D-RALL-ESSALUD-2023) and the Research Ethics Committee of Regional Teaching Hospital of Trujillo, Peru (September 19, 2023). All procedures complied with the principles established in the Declaration of Helsinki and the General Health Law of Peru (No. 26842).
All patients were informed about the objectives of the study and provided written informed consent prior to participation. The diagnosis of SLE and its severity, as well as the diagnosis of other rheumatological diseases, were established by a rheumatology specialist based on clinical examination, signs and symptoms, and corresponding blood and urine tests, such as antinuclear antibody (ANA), direct Coombs, anti-Smith (anti-Sm), and DNA antibody (anti-dsDNA) tests.
Subsequently, the principal researcher evaluated the presence of periodontitis according to the classification of Page and Eke,21 taking into account the following criteria: no/mild periodontitis (≥2 interproximal sites with clinical attachment loss ≤3 mm or probing depth ≤4 mm); moderate periodontitis (≥2 interproximal sites with clinical attachment loss ≥4 mm or ≥2 proximal sites with probing depth ≥5 mm); and severe periodontitis (≥2 interproximal sites with clinical attachment loss ≥6 mm and ≥1 interdental sites with probing depth ≥5 mm). To avoid information bias, intra- and inter-rater reliability were assessed through calibration of the principal investigator with an expert in periodontology affiliated with the Stomatology Program at Antenor Orrego Private University. The evaluation was conducted on 12 patients, yielding a κ value of 0.846 for both calibrations.
Statistical analysis
The obtained data was processed using the IBM SPSS Statistics for Windows software, v. 26.0 (IBM Corp., Armonk, USA). The correlation between periodontitis and SLE was determined using the χ2 test of independence. In addition, associations between study variables, adjusted for covariates, were evaluated using binary logistic regression (Wald test). The level of statistical significance was set at p < 0.05.
Results
In the present study, 342 female patients aged 18–92 years (mean (M) = 48.89; standard deviation (SD) = 16.27) who attended the rheumatology services of the abovementioned hospitals between July and October 2023 were evaluated. The prevalence of periodontitis in the study sample was 56.7% (Table 1).
Table 2 shows the association between periodontitis and SLE (p = 0.002). The percentage of patients with SLE who presented with periodontitis (68.4%) was twice as high as those without periodontitis (31.6%). When stratified by age group, a significant association between periodontitis and SLE was observed only in patients aged 27–59 years (p < 0.001). Regarding smoking habits, a significant correlation between periodontitis and SLE was found only among non-smokers (p = 0.017).
As shown in Table 3, no association was found between periodontitis and SLE in patients who used medications (p = 0.110) or in those who did not use medications (p = 0.861). However, a significant correlation was identified between periodontitis and SLE in patients without hypertension (p = 0.003) and in those without type 2 diabetes mellitus (T2DM) (p = 0.006).
Nevertheless, the associations identified through the bivariate analysis may be influenced by confounding factors such as age, smoking habits, medication use, and comorbidities. Therefore, a multivariate analysis was performed.
The multivariate binary logistic regression analysis identified no association between periodontitis and SLE (p = 0.453) (Table 4). However, significant differences in the prevalence of periodontitis were observed between older and younger adults (p = 0.006), smokers and non-smokers (p = 0.035), patients receiving medication and those not receiving medication (p = 0.000), hypertensive and non-hypertensive patients (p = 0.018), and diabetic and non-diabetic patients (p = 0.003). Furthermore, compared with their respective reference groups, the odds of periodontitis were 4.475-, 2.601-, 19.941-, 2.072-, and 3.760-fold higher in older adults, smokers, patients receiving medications for SLE, and those with hypertension and diabetes, respectively.
The multivariate analysis is more robust than the bivariate analyses presented in Table 2 and Table 3. As shown in Table 4, no association between periodontitis and SLE was observed after adjusting for age group, smoking status, medication use, and comorbidities. Furthermore, the analysis allowed for the evaluation of the association between these factors and periodontitis, regardless of SLE status.
Discussion
Systemic lupus erythematosus is a complex multisystem autoimmune condition that poses different challenges for diagnosis and treatment planning.8 Some studies have reported an association between SLE and periodontitis, particularly in terms of pathogenesis, physiological mechanisms and risk factors.16, 17 Dentists play an important role in the evaluation and management of these patients, taking into account the specific conditions associated with the disease.22
In the present study, the bivariate analysis revealed an association between periodontitis and the diagnosis of SLE. Similar results have been reported by Bolstad et al.,23 Rutter-Locher et al.,24 Wu et al.,25 and Calderaro et al.26 This may be explained by the fact that both diseases share genetic and environmental factors, as well as similar pathophysiological characteristics and inflammatory profiles. In this study, the number of patients diagnosed with SLE who presented with periodontitis was more than twice that of those without periodontitis, which is consistent with the findings of Bolstad et al. and Rutter-Locher et al., who reported that the risk of developing periodontitis in patients with SLE is approximately twice as high.23, 24
Regarding age groups, the association between periodontitis and SLE was observed only in patients aged 27–59 years. In this regard, Bolstad et al. found that SLE was associated with a higher risk of periodontitis in young patients aged 20–30 years.23 On the other hand, Wu et al. found no association between periodontitis and SLE across age subgroups.25 The findings of the present study may be explained by the larger proportion of adult patients with SLE attending the rheumatology service compared to younger and older age groups. Thus, future studies with balanced age group distributions are necessary to validate these findings, as the higher number of patients aged 27–59 years may have increased the statistical power. Furthermore, adult patients may have been under the effect of long-term medication, which could increase their susceptibility to periodontitis.
Regarding smoking habits, an association between periodontitis and SLE was observed only in non-smokers. This atypical result could be due to the small number of smoking patients diagnosed with SLE. In this regard, various authors, including Calderaro et al.,26 have reported greater probing depth in smoking patients, possibly due to alterations in gingival microbiota, immune response and periodontal tissue repair.27, 28 Chen et al. also reported an association between smoking and active skin rash in patients with SLE.27
No association was found between periodontitis and SLE in patients receiving pharmacological treatment for SLE or in those not receiving treatment, which is consistent with the findings of Calderaro et al.26 The pharmaceuticals most frequently administered to these patients include corticosteroids, immunosuppressants and antimalarial agents. Evidence suggests that these drugs may affect the periodontium due to their anti-inflammatory and immunosuppressive properties, potentially increasing susceptibility to microbial infections.29, 30
Some studies have indicated that periodontitis is a risk factor for the onset and progression of systemic diseases, including cardiovascular diseases (34%), T2DM (19–33%) and respiratory tract infections (50%).31, 32 When evaluating lupus patients with T2DM and hypertension, an association was found between periodontitis and SLE in individuals who did not present these comorbidities. This finding may be explained by the small number of hypertensive and diabetic patients included in the sample. In contrast, Bolstad et al. reported an association between periodontitis and SLE in patients with T2DM.23 Periodontitis and T2DM have a documented bidirectional association; furthermore, T2DM can promote the progression of periodontitis, which is associated with the invasion of host tissues by bacteria or degradation products into the systemic circulation.33, 34 Further studies with larger samples sizes are needed to clarify the association between periodontitis and SLE in patients with these comorbidities.
Furthermore, to strengthen the statistical analysis, binary logistic regression was performed. The analysis simultaneously evaluated multiple variables and covariates, providing a more accurate estimate of the probability of periodontitis and the associated factors, in addition to determining the risk of developing periodontitis from cofactors using odds ratios (ORs). Based on the results, no association between periodontitis and SLE was identified, in contrast to the results of the χ2 test. This discrepancy may be explained by the influence of covariates in the bivariate analysis, and lack of such influence in the multivariate model. Due to this contradiction, further studies with larger sample sizes and longitudinal designs are necessary to obtain more robust conclusions. On the other hand, the binary logistic regression analysis allowed us to observe an association between periodontitis and age (older adults), smoking, systemic medication use, hypertension, and T2DM. Notably, patients receiving medication for SLE were nearly 20 times more likely to develop periodontitis. This finding may reflect the effects of corticosteroids and immunosuppressants, such as prednisone and mycophenolate mofetil, on immune defense mechanisms in the oral cavity.35 In addition, glucocorticoids suppress the number and function of osteoblasts, leading to damage to bone turnover and compromising homeostasis in the periodontium.29
Although this study included a large sample of patients, its significant limitation is the unequal distribution of participants across covariate categories (e.g., 48 smokers vs. 294 non-smokers), which may have affected statistical power. In this sense, the associations observed with these variables should be interpreted with caution and only serve as preliminary information for the development of future studies.
Based on the results of this study, early diagnosis, treatment and periodontal control are recommended for patients with SLE. Future research should include larger samples and longitudinal designs, as the cross-sectional nature of this study does not allow for causal inference. In addition, collaboration between rheumatologists and periodontists is essential for the multidisciplinary management of patients with SLE. As suggested by Wu et al.,25 maintaining good periodontal health has a positive effect on controlling SLE, especially in patients undergoing immunosuppressive therapy, thus improving quality of life and increasing survival rates.
Conclusions
No overall association was identified between periodontitis and SLE in the sample population. However, a correlation was observed in specific subgroups, including patients aged 27–59 years, non-smokers, and those without hypertension or diabetes. In addition, a higher prevalence of periodontitis was noted in older patients, smokers, those receiving systemic medication for SLE, and patients with T2DM and hypertension.
Ethics approval and consent to participate
The study protocol was approved by the Faculty of Human Medicine of Antenor Orrego Private University (Resolution No. 2598-2023-FMH-UPAO) and the Bioethics Committee of Antenor Orrego Private University (Resolution No. 0658-2023-UPAO), Trujillo, Peru, as well as by the Research Committee of the La Libertad–ESSALUD Healthcare Network (PI No. 121 CIYE-O.C.I.Y. D-RALL-ESSALUD-2023) and the Research Ethics Committee of Regional Teaching Hospital of Trujillo, Peru (September 19, 2023). All procedures complied with the principles established in the Declaration of Helsinki and the General Health Law of Peru (No. 26842).
Data availability
This work corresponds to the thesis required to obtain the professional degree of Dental Surgeon from Antenor Orrego Private University (UPAO), Trujillo, Peru.
The data supporting the findings of this study are available in the institutional repository of UPAO at https://hdl.handle.net/20.500.12759/36331.
Consent for publication
Not applicable.
Use of AI and AI-assisted technologies
Not applicable.