Abstract
Improving diagnostic precision in periodontology and implantology is essential for better patient care, scientific advancement and study consistency, while always considering the worst-case scenario for diagnosis.
Keywords: periodontics, diagnosis, bitewing radiography, periodontal attachment loss
Introduction
An accurate diagnosis is the foundation of effective periodontal and peri-implant treatment planning.1, 2 The concurrent presentation of two distinct yet related pathologies pose a significant and often underestimated diagnostic challenge in periodontology. An important and underdiscussed problem therefore persists in both the literature and clinical practice – the difficulty of establishing an accurate diagnosis when two pathologies occur simultaneously.3, 4 The current classification systems,5, 6 while attempting to be comprehensive, struggle to provide clear guidance for clinicians when these entities coexist, leading to potential diagnostic inaccuracies, compromised treatment planning and inconsistencies in epidemiological reporting.
Although these conditions may represent points along a biological continuum, they remain distinct entities with different prognostic implications, treatment priorities and documentation standards.1, 7, 8 Misclassification can lead to inappropriate treatment decisions, erroneous prevalence data and inconsistency in clinical research. A recurring diagnostic error arises when clinicians report the less severe disease simply because it is visually obvious (e.g., erythema and bleeding on probing (BoP) suggesting gingivitis), while the presence of clinical attachment loss (CAL), radiographic bone loss (RBL) or deep periodontal pockets is overlooked.7 The same occurs in implant dentistry9: a case with soft-tissue inflammation around an implant may be labeled “peri-implant mucositis”, even when radiographs reveal progressive bone loss, indicating clear peri-implantitis. This confusion is reinforced by ambiguous terminology or insufficient diagnostic criteria in publications, causing inexperienced or inattentive clinicians to misinterpret cases and hindering standardization across studies.
Problem of coexisting pathologies
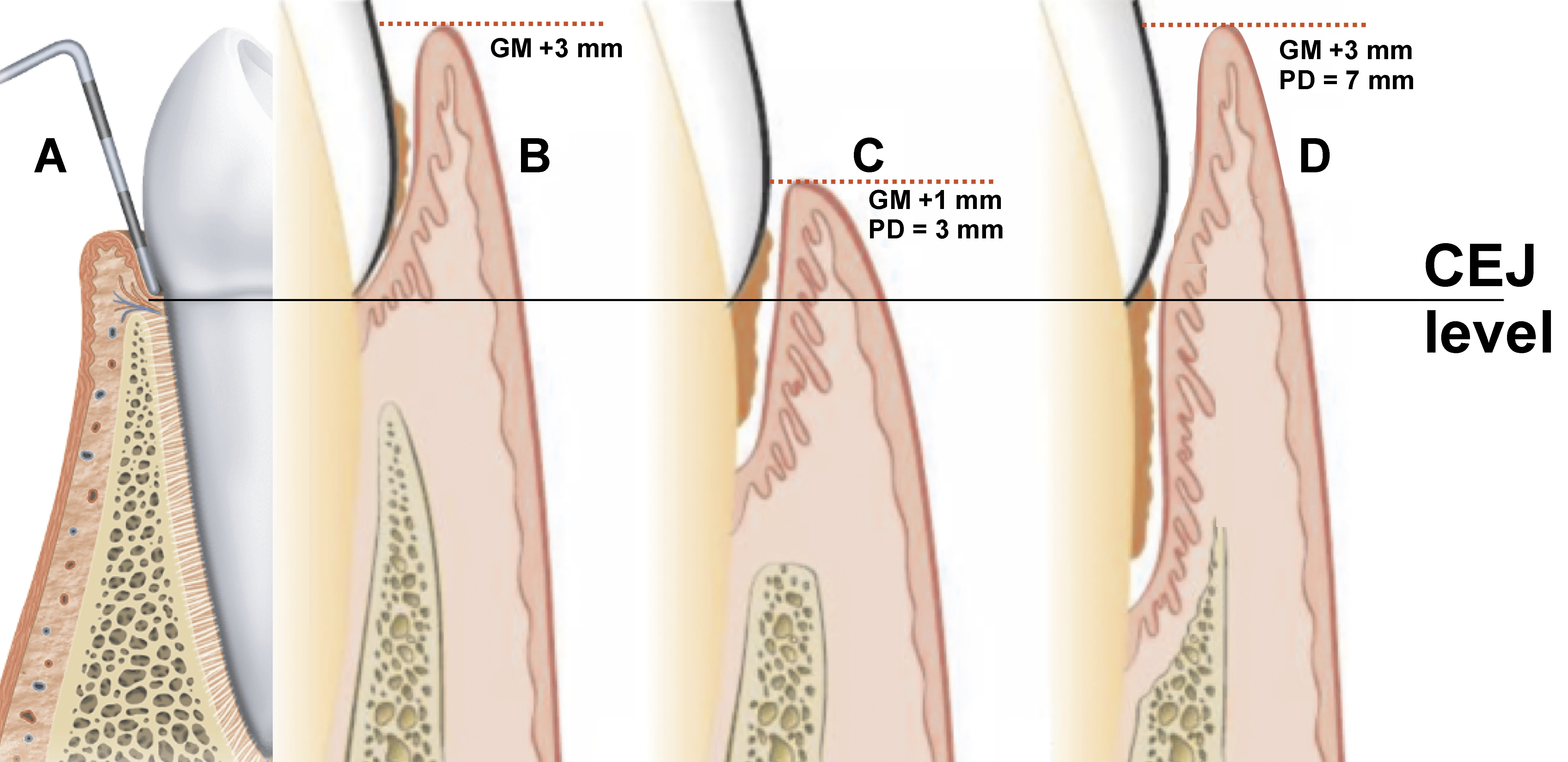
The core of the problem lies in the hierarchical relationship and symptomatic overlap between these diseases. In natural dentition,5, 6 gingivitis (Figure 1, Figure 2A,B) is defined as the reversible inflammation of the gingiva without CAL, and periodontitis is characterized by inflammation with CAL and RBL. Gingivitis is fundamentally, but not necessarily, the precursor or co-manifestation of periodontitis. In implant dentistry, peri-implant mucositis is the reversible inflammation of the peri-implant soft tissues without bone loss, while peri-implantitis is characterized by inflammation and progressive peri-implant bone loss (CAL and RBL).
When a patient presents with established periodontitis (Figure 2C) – defined by the presence of CAL and RBL – alongside active, site-specific gingivitis, the most severe diagnosis should be considered periodontitis. Similarly, the presence of peri-implantitis (bone loss present) supersedes and includes the inflammatory component of peri-implant mucositis.
Thus, to ensure diagnostic accuracy, the principle of “worst diagnosis prevails” must be emphasized. When signs of both gingivitis and periodontitis are present (Figure 2D), the correct diagnosis is periodontitis, characterized by a high level of gingival inflammation and, typically, a high percentage of BoP. Similarly, when peri-implant mucositis and peri-implantitis coexist, the peri-implantitis diagnosis should take precedence. The “worst diagnosis prevails” rule is a safety-first approach to dental health. It means that if a patient shows signs of both a minor, treatable/reversible condition (such as gingivitis) and a serious, permanent/irreversible condition (such as bone loss (CAL and RBL) – periodontitis), the dentist must treat the case as the most serious condition. This ensures that the most damaging aspect of the disease is never overlooked.
Diagnostic confusion for clinicians (specialists, dentists and dental students) arises, because the less severe, reversible condition (gingivitis/peri-implant mucositis) is often the most visually apparent manifestation of active inflammation. This can lead to the underestimation of the severity of the underlying, irreversible tissue destruction – namely CAL and RBL – associated with periodontitis/peri-implantitis. Alternatively, clinicians may correctly diagnose periodontitis, yet fail to account for the influence of gingival inflammation on the probing depth (PD). This can lead to the overestimation of CAL, based on the simplified calculation: CAL = PD − GM (gingival margin). When GM is coronal to the cementoenamel junction (CEJ), it is assigned a positive value; when apical to CEJ, a negative value.1 This potential inaccuracy can be mitigated by radiographic assessment, particularly with bitewing radiographs.
The literature’s reliance on separate definitions can inadvertently promote the notion that clinicians must choose between diagnoses, rather than recognize the more severe, irreversible disease as the definitive diagnosis that encompasses the active inflammatory component. For instance, a site with established periodontitis that has recently become inflamed is not merely gingivitis; it is periodontitis with active inflammation (or a high level of gingival inflammation/BoP). This ambiguity has direct implications for treatment planning. An exception arises when a patient is observed over an extended period and the clinical parameters of periodontitis remain stable throughout follow-up; in such cases, the diagnosis may be revised to periodontal health or gingivitis in a reduced periodontium.
Recognizing the more severe condition is not merely a semantic distinction; it directly determines appropriate therapy, recall intervals, risk communication with patients, and the ability to monitor disease progression. Misdiagnosing an active case of periodontitis as gingivitis, for example, may result in inadequate therapy, failing to address the need for root surface debridement and the management of attachment loss.
Critical role of the probing depth (PD) and inflammation: Misdiagnosis arising from PD interpretation
The probing depth alone does not equate to CAL, RBL, or overall disease severity1, 6, 10; it must be interpreted within the context of gingival inflammation and tissue swelling. Inflammatory enlargement commonly produces pseudopockets, which may mimic increased PD in the absence of true attachment loss. Therefore, clinicians must distinguish between the increased PD resulting from edematous gingiva and the increased PD associated with the apical migration of the junctional epithelium, which reflects true periodontal breakdown. Failure to account for the inflammatory component may lead to the overdiagnosis of periodontitis in cases of gingivitis or underdiagnosis when inflammation masks the existing bone loss.
To accurately differentiate and identify the more severe condition, the clinical measurement of PD must be interpreted within the essential context of gingival inflammation/BoP and the radiographic bone levels. The probing depth is defined as the distance from GM to the base of the gingival sulcus/periodontal pocket, and its clinical significance changes substantially in the presence of inflammation:
1. Increased PD without BoP: a PD of 4–5 mm without BoP usually indicates a stable, treated, or fibrotic periodontal pocket (“pocket of residual depth”) that may not require immediate invasive intervention, particularly in cases of resolved/stable periodontitis (a reduced periodontium). If PD exceeds 5 mm, a “red flag” should be raised to investigate the underlying cause at that specific site. Possible explanations include a smoker with deep PD, the presence of a pseudopocket (which is uncommon without BoP, but can occur), or measurement errors. Radiographic assessment, preferably with bitewing radiographs, is essential to confirm true CAL or RBL. If radiographs do not show CAL/RBL, the site likely represents a pseudopocket, which is generally associated with a high level of BoP.
2. Increased PD with BoP: a PD over 3 mm accompanied by BoP indicates an active pathological site. This active inflammation signals that the measured PD reflects an ongoing disease process, such as active gingivitis/peri-implant mucositis or periodontitis/peri-implantitis. In cases of coexisting pathologies, clinicians must prioritize the measurements that indicate irreversible damage. This involves assessing CAL, using an accurate reference point – typically CEJ for the teeth – and confirming the findings with RBL assessment. Radiographic bone loss is measured from CEJ to the crestal bone, and should not exceed approx. 2.0 mm, which corresponds to the average supracrestal tissue attachment (STA), formerly known as the biological width.
The measurement of CAL, combined with the assessment of BoP, distinguishes the two conditions: (A) gingivitis/peri-implant mucositis – an increased PD is primarily due to gingival swelling (a pseudopocket), with no CAL present; (B) periodontitis/peri-implantitis – an increased PD results from both inflammation and the apical migration of the junctional epithelium – or from migration alone – leading to irreversible CAL and RBL.
In summary, differentiating transient inflammation from permanent tissue destruction requires careful distinction between PD and CAL/RBL. The probing depth is a purely topographic measure, representing the distance from GM to the base of the periodontal pocket. As such, it is highly sensitive to the gingival phenotype and the inflammatory edema, which can produce pseudopockets without actual structural loss. In contrast, CAL and RBL are definitive clinical indicators of periodontal stability, reflecting the apical migration of the junctional epithelium relative to a fixed anatomical landmark – CEJ. While a deep PD signals the presence of a clinical niche, only CAL, corroborated by RBL, confirms a diagnosis of periodontitis. This distinction is essential: PD reflects the ‘depth’ of the current clinical state, often associated with BoP, whereas CAL represents the cumulative history of irreversible attachment loss.
Radiographic assessment – the decisive factor
The definitive, non-negotiable criterion for identifying the “worst” pathology is the radiographic evidence of bone loss. Bitewing radiographs (Fig. 2E) are essential for the accurate assessment of the alveolar bone, offering a clear, undistorted view of the interproximal bone crest relative to CEJ or the implant–abutment interface for implants.1 Properly taken bitewings are crucial for determining whether an increased PD corresponds to true bone loss. In comparison with periapical radiographs, which are often affected by variable angulation, bitewings provide more precise visualization and measurement of the interproximal crestal bone levels.
For diagnosing periodontitis,1, 5, 6 bitewing radiographs are necessary to confirm that the measured CAL corresponds to RBL, the two most critical parameters in periodontal diagnosis. In a patient with deep pockets and BoP, radiographic evidence confirms that the condition represents true periodontitis rather than merely gingival inflammation. The interdental bone level provides a definitive proof of the irreversible nature of the disease, taking precedence over superficial inflammatory signs.
However, while bitewing radiographs are the primary tool for assessing the crestal bone, other imaging modalities are valuable in specific clinical situations. Periapical radiographs are required to evaluate the full root length and the periapical status, whereas cone-beam computed tomography (CBCT) may be indicated for assessing complex three-dimensional (3D) defects, furcation involvement or anatomical relationships prior to advanced surgical interventions.
Final comments
Diagnostic accuracy depends on the careful evaluation of both clinical parameters and radiographic evidence. Without appropriate imaging, clinicians may misclassify gingivitis as periodontitis or fail to detect early peri-implantitis beneath apparently healthy soft tissue. The literature remains inconsistent in defining periodontal and peri-implant diseases when multiple signs coexist. Some studies consider increases in PD and inflammation sufficient to indicate disease progression, whereas others prioritize radiographic confirmation.
Until clearer diagnostic hierarchies and standardized operational definitions are widely adopted, variability in diagnosis will persist. This inconsistency affects not only clinical communication, but also the interpretation of epidemiological data, meta-analyses and evidence-based treatment recommendations. The irreversible condition – periodontitis or peri-implantitis – must always be considered the primary diagnosis when there is evidence of CAL/RBL, regardless of the severity of the reversible inflammatory component.
Clinical decision-making should be based on a tripartite assessment: (1) CAL/RBL (bitewing radiographs); (2) PD; and (3) BoP. This structured approach is fundamental for moving beyond the current diagnostic dilemma and ensuring that patients receive appropriate, definitive care targeting the most destructive aspect of their disease.
Conclusions
In light of these challenges, this editorial urges the periodontal and implantology communities to place greater emphasis on:
– assigning the most severe diagnosis when multiple conditions coexist;
– interpreting PD in the context of gingival inflammation;
– utilizing appropriate, standardized bitewing radiographs to confirm bone loss; and
– clarifying diagnostic criteria in publications and educational materials.
Enhancing diagnostic precision is essential not only for delivering high-quality patient care, but also for advancing the scientific literature and ensuring consistency across clinical studies.
Ethics approval and consent to participate
Not applicable.
Data availability
All data for this study are included within the article.
Consent for publication
Not applicable.
Use of AI and AI-assisted technologies
Not applicable.