Abstract
Background. Ensuring vaccine safety is crucial for maintaining public confidence, particularly in relation to non-life-threatening adverse events (AEs) that may impact quality of life and contribute to vaccine hesitancy. While systemic reactions to coronavirus disease 2019 (COVID-19) vaccines are well documented, oral AEs remain underexplored, despite emerging reports linking vaccination to taste disturbances and other oral manifestations.
Objectives. The aim of the study was to explore oral AEs following COVID-19 vaccination and to assess their potential to constitute signals of disproportionate reporting (SDRs) that warrant further investigation and validation.
Material and methods. A hybrid frequentist–Bayesian signal detection approach was applied to analyze oral AEs following COVID-19 vaccination using data from the US Vaccine Adverse Event Reporting System (VAERS). A two-stage analysis was conducted. The primary analysis identified SDRs, and the secondary analysis assessed reporting biases, including selective reporting bias and notoriety bias.
Results. Oral AEs were reported 2.4 times more frequently following COVID-19 vaccination compared with other vaccines, with taste-related SDRs showing the highest disproportionality. The most prominent SDRs included taste disorder (proportional reporting ratio (PRR) = 23.96), ageusia (PRR = 17.79), hypogeusia (PRR = 6.25), dysgeusia (PRR = 3.22), and oral lichen planus (PRR = 3.12). Females and middle-aged adults exhibited the highest reporting rates, whereas severe, hospitalized and fatal cases were less frequent. Co-occurrence analysis of gustatory and olfactory AEs suggests notoriety bias, likely influenced by increased public awareness. Regression analyses indicated that female sex and older age were associated with increased odds of oral SDRs, while state-level political inclination had no significant effect.
Conclusions. The findings highlight demographic disparities in the reporting of oral AEs. However, they should not be interpreted as undermining vaccine safety. Future research should focus on clinically significant oral SDRs, such as oral lichen planus, to distinguish true safety signals from potential reporting biases. Improved awareness and communication are essential for contextualizing these findings within dental practice.
Keywords: COVID-19 vaccines, oral manifestations, pharmacovigilance, adverse drug reaction reporting systems, Bayesian analysis
Introduction
Throughout history, anti-vaccination movements have actively spread misinformation regarding vaccine safety, often amplifying reports of adverse events (AEs) to undermine public confidence in immunization programs.1, 2 These tactics have evolved with advancements in communication technologies, particularly through social media, leading to widespread vaccine hesitancy.3 To counteract misinformation and ensure public trust, national pharmacovigilance systems play a crucial role in monitoring post-vaccination AEs, identifying safety signals and distinguishing true vaccine-related risks from coincidental events.4, 5 The Vaccine Adverse Event Reporting System (VAERS) in the United States serves as a cornerstone of passive surveillance, allowing healthcare providers and the public to report potential vaccine-related AEs.6 Among these, oral manifestations such as taste disorders, paresthesia and ulcers have emerged as notable concerns following coronavirus disease 2019 (COVID-19) vaccination.7, 8, 9, 10, 11
A growing body of evidence suggests that certain AEs may be disproportionately reported due to increased public awareness, regulatory alerts or media attention, a phenomenon known as notoriety bias.4, 12 This bias can lead to an overestimation of AE incidence in passive surveillance databases, as individuals become more likely to report events they perceive as vaccine-related. Taste-related AEs are particularly susceptible to notoriety bias, as loss of taste and smell are widely recognized diagnostic symptoms of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection.13, 14, 15 Previous studies have shown that the reporting rate of taste-related AEs in VAERS was from 3 to 17 times higher during the pandemic compared with the pre-pandemic period.7 This raises concerns about whether taste-related AEs following COVID-19 vaccination reflect a true biological association or are influenced by heightened public vigilance.
While numerous studies have examined AEs following COVID-19 vaccination, most have focused on systemic reactions such as fever, fatigue and anaphylaxis, with relatively little attention given to less common AEs, including oral manifestations.16, 17 Prior analyses of passive surveillance databases have identified common patterns of oral AEs in major regions, such as the United States, the European Union and Australia; however, no study has comprehensively assessed their disproportionality using advanced signal detection methods.9
The present study aimed to conduct a disproportionality analysis of oral AEs following COVID-19 vaccination using a hybrid frequentist–Bayesian signal detection approach, thereby contributing to the long-term pharmacovigilance of novel vaccine technologies that remain in global use. By identifying true signals of disproportionate reporting (SDRs) and assessing the impact of factors such as sex, age, vaccine type, and state-level political inclination, this study sought to enhance understanding of vaccine safety monitoring. Additionally, by investigating the potential role of notoriety bias in the increased reporting of gustatory AEs, the findings may help refine pharmacovigilance methodologies and improve risk communication strategies in future vaccination campaigns.
Material and methods
Study design
A disproportionality analysis was conducted in October 2024 using individual case safety reports (ICSRs) from VAERS. The study focused on oral AEs reported after COVID-19 vaccination to identify SDRs and validate them using an advanced statistical approach.
The analysis was performed and reported following the REporting of A Disproportionality analysis for drUg Safety signal detection using individual case safety reports in PharmacoVigilance (READUS-PV) guidelines to ensure transparency, reproducibility and methodological rigor.18
Data source
The Vaccine Adverse Event Reporting System is a passive surveillance database jointly managed by the Centers for Disease Control and Prevention (CDC) and the Food and Drug Administration (FDA). It collects AE reports from healthcare professionals, vaccine manufacturers and the general public.19 The Medical Dictionary for Regulatory Activities (MedDRA) is a hierarchical coding system used by VAERS to classify AEs for efficient data retrieval.20
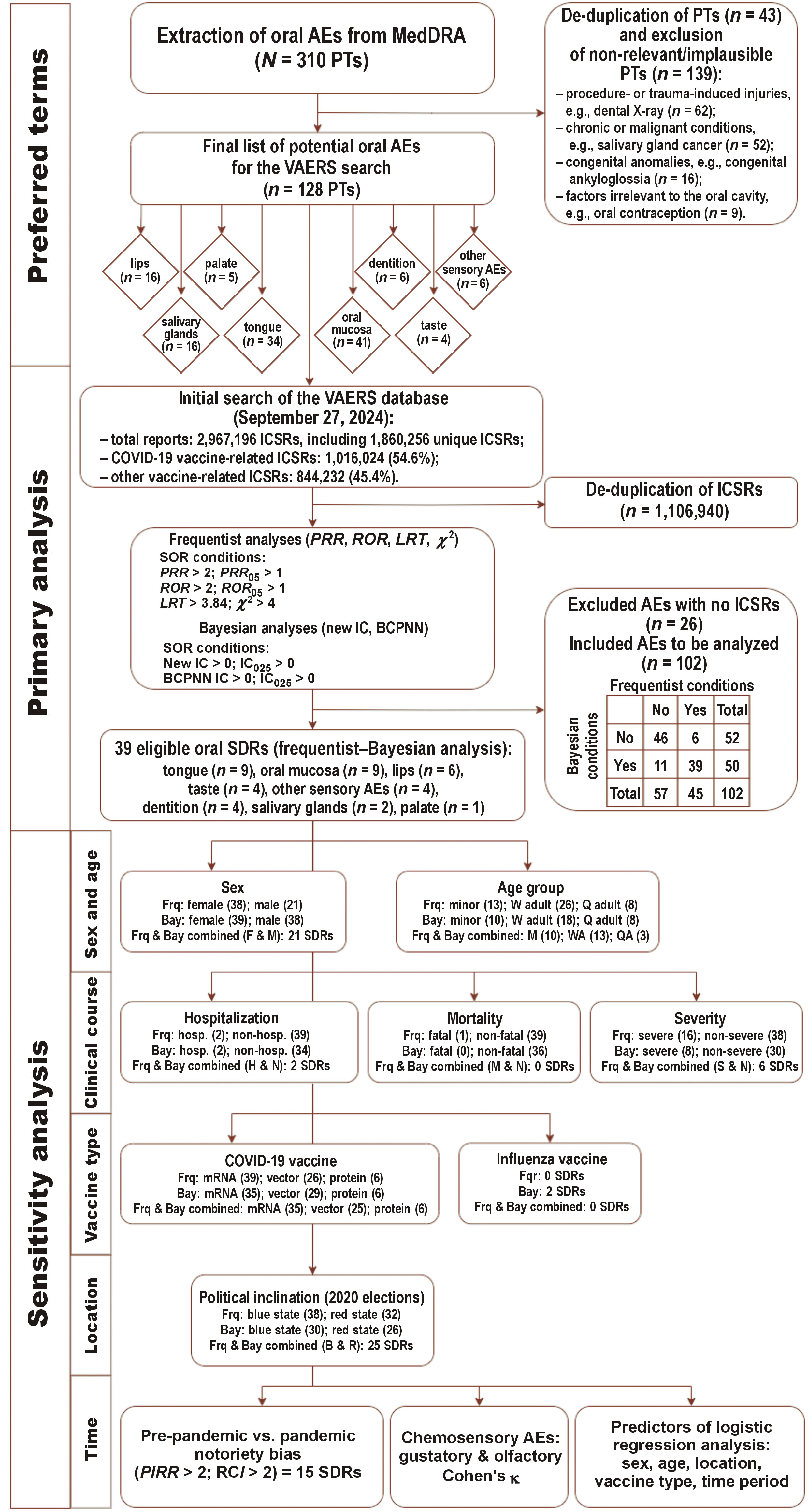
The dataset was downloaded from the official VAERS website in October 2024 and included all ICSRs submitted between January 1990 and September 2024. The raw dataset comprised 2,967,196 ICSRs. After removing 1,106,940 duplicate records, 1,860,256 unique ICSRs remained. Of these, 1,016,024 (54.6%) were related to COVID-19 vaccines, and 844,232 (45.4%) were associated with other vaccines (Figure 1).
Variables
To analyze oral AEs, an anatomophysiological framework was applied, categorizing AEs by anatomical regions (lips, palate, tongue, dentition, salivary glands, and oral mucosa) and functions (taste and other sensations). A MedDRA search at the preferred term (PT) level identified 128 PTs relevant to the oral cavity. This framework was described in a previous study7 and is presented in Figure 1.7
Oral AE reports were stratified by sex (female/male), age group (infants, children, young adults, middle-aged adults, pre-seniors, seniors, and elderly) according to the United States Census Bureau classification,21 clinical course (severe, hospitalized or fatal) according to the definitions provided in the VAERS user guidelines,6 vaccine type (mRNA, vector-based and protein subunit), and state-level political inclination (blue (Democratic) or red (Republican)) as indicated by the outcomes of the US 2020 presidential election.22 Lastly, to assess temporal trends, the study period was divided into pre-pandemic (1990–2019) and pandemic (2020–2024) phases.
Statistical analysis
Statistical analyses were conducted in 2 stages. The primary analysis assessed disproportionate reporting of 128 potential oral AEs to identify true SDRs by applying both the frequentist and Bayesian conditions. This resulted in the identification of 39 oral AEs as true SDRs, as they met the predefined signal detection criteria.
The frequentist criteria for identifying true SDRs were as follows:
– proportional reporting ratio (PRR) ≥2, with the lower bound of the 95% confidence interval (95% CI) ≥1;
– reporting odds ratio (ROR) ≥2, with the lower bound of the 95% CI ≥ 1;
– likelihood ratio test (LRT) ≥3.84;
– χ² test with Yates’s correction ≥4;
– total number of reports >3.
The Bayesian criteria were:
– new information component (IC) method, requiring both the point estimate and the lower limit of the 95% CI for the IC (IC025) to be >0;
– Bayesian Confidence Propagation Neural Network (BCPNN) method, requiring both the point estimate and the IC025 to be >0.
A secondary (sensitivity) analysis was conducted on the 39 true SDRs to control for potential reporting biases, including selective reporting bias and notoriety bias. Disproportionality estimates were stratified by sex, age, clinical severity, vaccine type, vaccine schedule, state-level political inclination, and time period.
To examine potential notoriety bias in gustatory AEs, Cohen’s kappa coefficient was calculated to assess the agreement between gustatory and olfactory AEs, stratified by time period, sex, age, and vaccine type. Specifically, agreement coefficients were compared between pre-pandemic and pandemic periods, as well as between COVID-19 and non-COVID-19 vaccines during the pandemic. Multivariable logistic regression analyses were conducted to estimate adjusted odds ratios (AORs) for true SDRs, adjusting for sex, age, state-level political inclination, vaccine type, and time period.
All statistical analyses were conducted using the R software, v. 4.4.2 (https://www.r-project.org).23
Results
As of September 2024, VAERS contained 1,860,256 unique ICSRs, of which 56,501 reported oral AEs (30.37 cases per 1,000 ICSRs). The crude PRR of oral AEs following COVID-19 vaccination was 2.40 (95% CI: 2.35–2.44), indicating a higher reporting frequency compared with other vaccines. Females exhibited a higher PRR (2.29) than males (1.67), and younger vaccine recipients had lower PRRs than older groups. Non-severe, non-hospitalized and non-fatal cases showed higher PRRs than severe, hospitalized and fatal cases. Proportional reporting ratios were nearly identical in red and blue states (Table 1).
Primary analysis
Of the 128 oral AEs evaluated, 45 met the frequentist criteria, 50 met the Bayesian criteria, and 39 satisfied both, thereby qualifying as true SDRs. These SDRs were distributed across tongue (n = 9), oral mucosa (n = 9), lips (n = 6), taste (n = 4), other sensations (n = 4), dentition (n = 4), salivary glands (n = 2), and palate (n = 1).
Within taste-related AEs, taste disorder showed the highest PRR (23.96), followed by ageusia (17.79), hypogeusia (6.25) and dysgeusia (3.22). Other sensory AEs also exhibited high PRRs, including oral anesthesia (16.62), burning mouth syndrome (4.67), oral paresthesia (3.72), and oral hypoesthesia (3.50).
For oral mucosal AEs, aphthous ulcer (4.43), oral lichen planus (3.12) and oral pruritus (2.65) were identified as true SDRs. In contrast, oral herpes (1.51) and oral candidiasis (1.39) did not meet the criteria for true SDRs (Table 2).
Sensitivity analyses
Sex
The frequentist criteria identified 38 true SDRs in females and 21 in males, while the Bayesian criteria identified 39 true SDRs in females and 38 in males. Twenty-one SDRs were confirmed by both methods across both sexes.
Taste disorder had the highest PRR in both females (19.23) and males (28.06). In females, this was followed by dental paresthesia (18.30) and ageusia (15.97), while in males, tongue erythema (23.04) and ageusia (17.25) were the most prominent (Supplementary File: Table S1 (https://doi.org/10.17605/OSF.IO/3S8KW)).
Age
Infants and toddlers (0–4 years) showed only 1 true SDR (oral hypoesthesia (PRR: 26.77)), while children and adolescents (5–17 years) had 4 true SDRs, namely taste disorder (20.71), ageusia (11.10), aphthous ulcer (9.53), and mouth swelling (3.71).
Young adults (18–34 years) exhibited 6 true SDRs: taste disorder (27.72); ageusia (11.34); tongue discomfort (5.87); tongue pruritus (5.66); mouth swelling (2.83); and oral paresthesia (2.14). Similarly, middle-aged adults (35–49 years) showed 7 true SDRs, such as taste disorder (14.46), ageusia (9.50), tongue discomfort (5.48), tongue pruritus (4.82), mouth swelling (4.37), toothache (3.05), and oral paresthesia (2.08). Moreover, pre-seniors (50–64 years) had 8 true SDRs: taste disorder (12.86); ageusia (11.72); tongue discomfort (5.46); tongue pruritus (5.19); oral paresthesia (2.34); dysgeusia (2.31); oral hypoesthesia (2.21); and oral discomfort (2.03).
Seniors (65–79 years) had 4 true SDRs, namely taste disorder (11.52), ageusia (10.43), tongue discomfort (4.99), and oral paresthesia (2.13), whereas ageusia (7.54) was the only true SDR identified among individuals aged ≥80 years (Supplementary File: Table S2).
Clinical course
Severe cases had 8 true SDRs, while non-severe cases showed 30 true SDRs. Six SDRs were shared between both groups, including ageusia (severe (PRR: 10.04) vs. non-severe PRR: 22.38)), hypogeusia (6.81 vs. 6.49), taste disorder (31.94 vs. 21.12), tongue discomfort (5.12 vs. 8.81), tongue pruritus (3.61 vs. 6.59), and mouth swelling (2.28 vs. 3.48) (Supplementary File: Table S3).
Hospitalized cases showed only 2 true SDRs, compared with 34 among non-hospitalized cases. Both hospitalized and non-hospitalized cases had jointly 2 true SDRs, including ageusia (hospitalized (PRR: 11.73) vs. non-hospitalized (PRR: 18.30)) and taste disorder (9.65 vs. 25.40, respectively) (Supplementary File: Table S4).
Mortality cases showed no true SDRs, while non-mortality cases exhibited 36 true SDRs. Among fatal cases, the Bayesian criteria were not met by any oral AE, while the frequentist conditions were met by ageusia only (PRR: 7.81) (Supplementary File: Table S5).
Vaccine schedule
Primary COVID-19 vaccination doses were associated with 35 true SDRs, while booster doses showed 10 true SDRs. Nine SDRs were shared between both schedules, including ageusia (primer (PRR: 17.79) vs. booster (PRR: 17.68)), hypogeusia (6.20 vs. 7.47), taste disorder (24.37 vs. 14.14), toothache (3.21 vs. 3.79), tongue blistering (2.73 vs. 3.26), tongue erythema (7.95 vs. 6.49), lip pain (2.45 vs. 2.24), aphthous ulcer (4.45 vs. 3.82), and mouth swelling (3.51 vs. 3.43) (Supplementary File: Table S6).
Vaccine type
The mRNA-based and viral vector-based COVID-19 vaccines exhibited 35 and 25 true SDRs, respectively, whereas protein subunit vaccines showed 6 true SDRs, as follows: ageusia (PRR: 18.74); taste disorder (14.93); glossodynia (4.53); dry mouth (4.21); dysgeusia (4.09); and oral paresthesia (2.31).
Among mRNA-based vaccines, taste disorder had the highest PRR (23.77), followed by ageusia (17.58), oral anesthesia (16.13), dental discomfort (15.23), dental paresthesia (12.54), hyperesthesia of teeth (11.85), tongue discomfort (9.11), tongue erythema (8.01), tongue pruritus (6.87), and hypogeusia (6.13). Similarly, for viral vector-based vaccines, taste disorder came first (26.07), followed by oral anesthesia (22.28), ageusia (20.55), dental paresthesia (16.71), dental discomfort (13.92), and hyperesthesia of teeth (10.28). When evaluating influenza vaccines, no true SDRs were found (Supplementary File: Table S7).
State-level political inclination
Blue (Democratic) states exhibited 30 true SDRs, and red (Republican) states showed 25 true SDRs. Both types of states shared 25 true SDRs, including taste disorder (blue (PRR: 23.20) vs. red (PRR: 25.22)), ageusia (17.27 vs. 20.56), hypogeusia (9.06 vs. 4.51), dysgeusia (3.11 vs. 3.30), oral paresthesia (3.72 vs. 3.56), oral hypoesthesia (3.57 vs. 3.32), and aphthous ulcer (4.71 vs. 4.38). Five AEs were unique to blue states: dental discomfort; dry tongue; tongue eruption; lip disorder; and palatal swelling (Supplementary File: Table S8).
Co-occurrence of chemosensory AEs
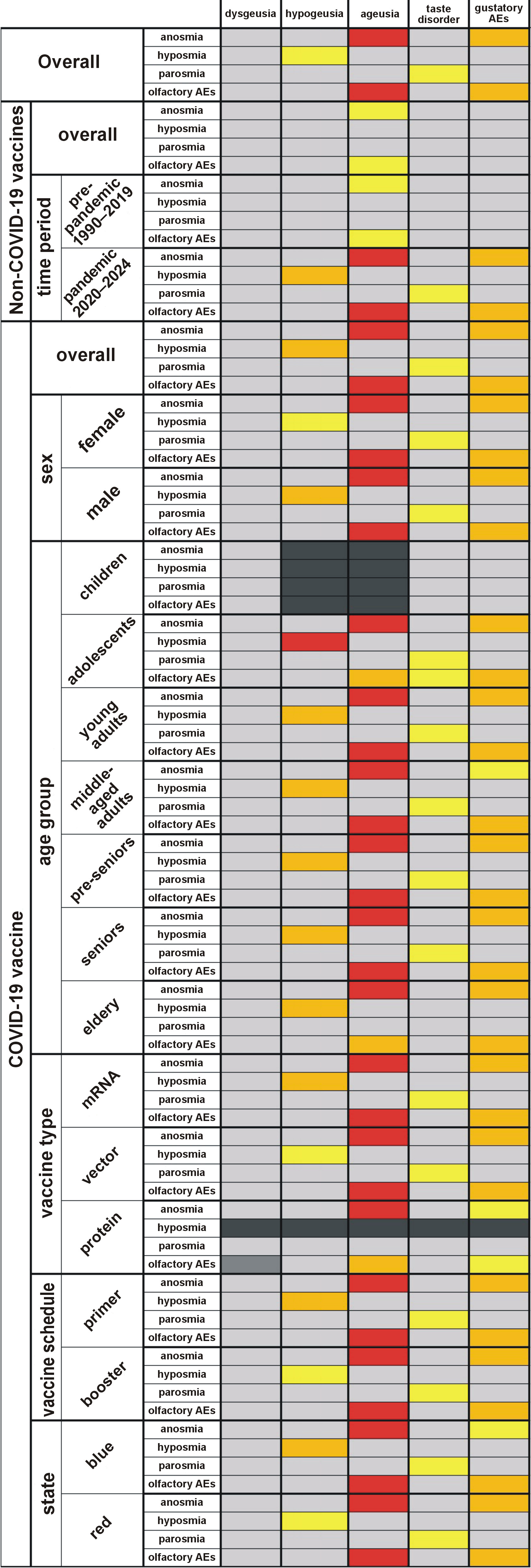
Overall, gustatory and olfactory AEs co-occurred with a Cohen’s kappa (κ) of 0.459, indicating moderate agreement. The strongest agreement was observed for ageusia and anosmia (κ = 0.699), followed by hypogeusia and hyposmia (κ = 0.399), and taste disorder and parosmia (κ = 0.258) (Supplementary File: Table S9).
For non-COVID-19 vaccines, a considerable difference was observed between the pre-pandemic (January 1990–December 2019) and pandemic (January 2020–October 2024) intervals. The co-occurrence of any gustatory AE and any olfactory AE was slight in the pre-pandemic period (κ = 0.115) but moderate in the pandemic period (κ = 0.500). Ageusia and anosmia exhibited fair agreement in the pre-pandemic period (κ = 0.283), but substantial agreement was noted between the two in the pandemic interval (κ = 0.721). Similarly, hypogeusia and hyposmia showed no agreement pre-pandemic (κ < 0.001), and moderate agreement in the pandemic period (κ = 0.474).
During the pandemic, the co-occurrence of chemosensory AEs was moderate for both non-COVID-19 (κ = 0.500) and COVID-19 vaccines (κ = 0.489). Ageusia and anosmia exhibited substantial agreement for non-COVID-19 (κ = 0.721) and COVID-19 vaccines (κ = 0.710). Similarly, hypogeusia and hyposmia showed moderate agreement for non-COVID-19 (κ = 0.474) and COVID-19 vaccines (κ = 0.438) (Figure 2).
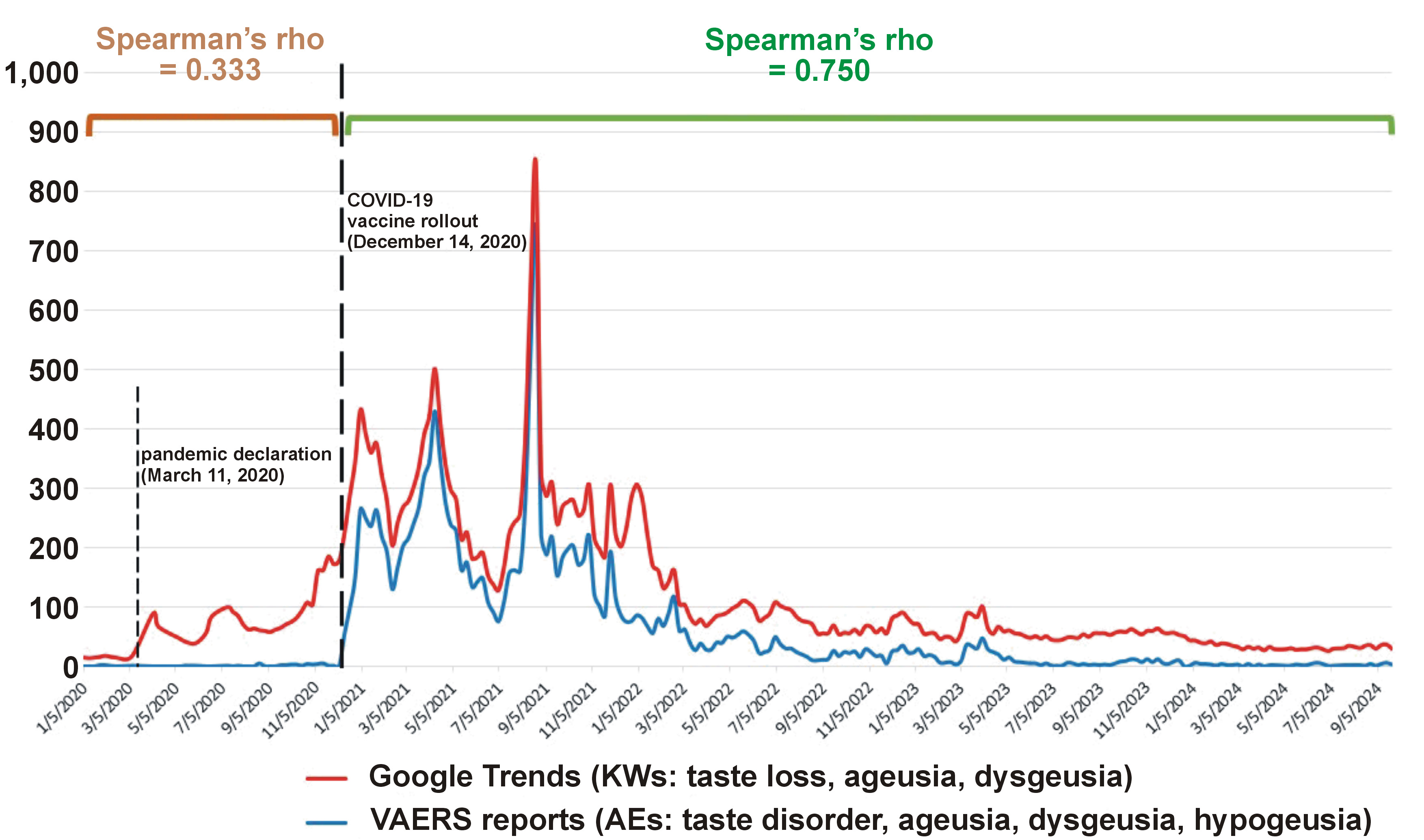
Google Trends and VAERS reports
A non-parametric correlation analysis was performed between the weekly search volume of keywords (taste loss, ageusia and dysgeusia) in Google Trends (https://trends.google.com/trends) and the weekly number of gustatory AE reports in VAERS (taste disorder, ageusia, dysgeusia, and hypogeusia). Spearman’s rho coefficient was 33.3% during the pandemic period preceding the COVID-19 vaccine rollout, and increased to 75.0% following the public administration of COVID-19 vaccines on December 14, 2020 (Figure 3).
Regression analyses
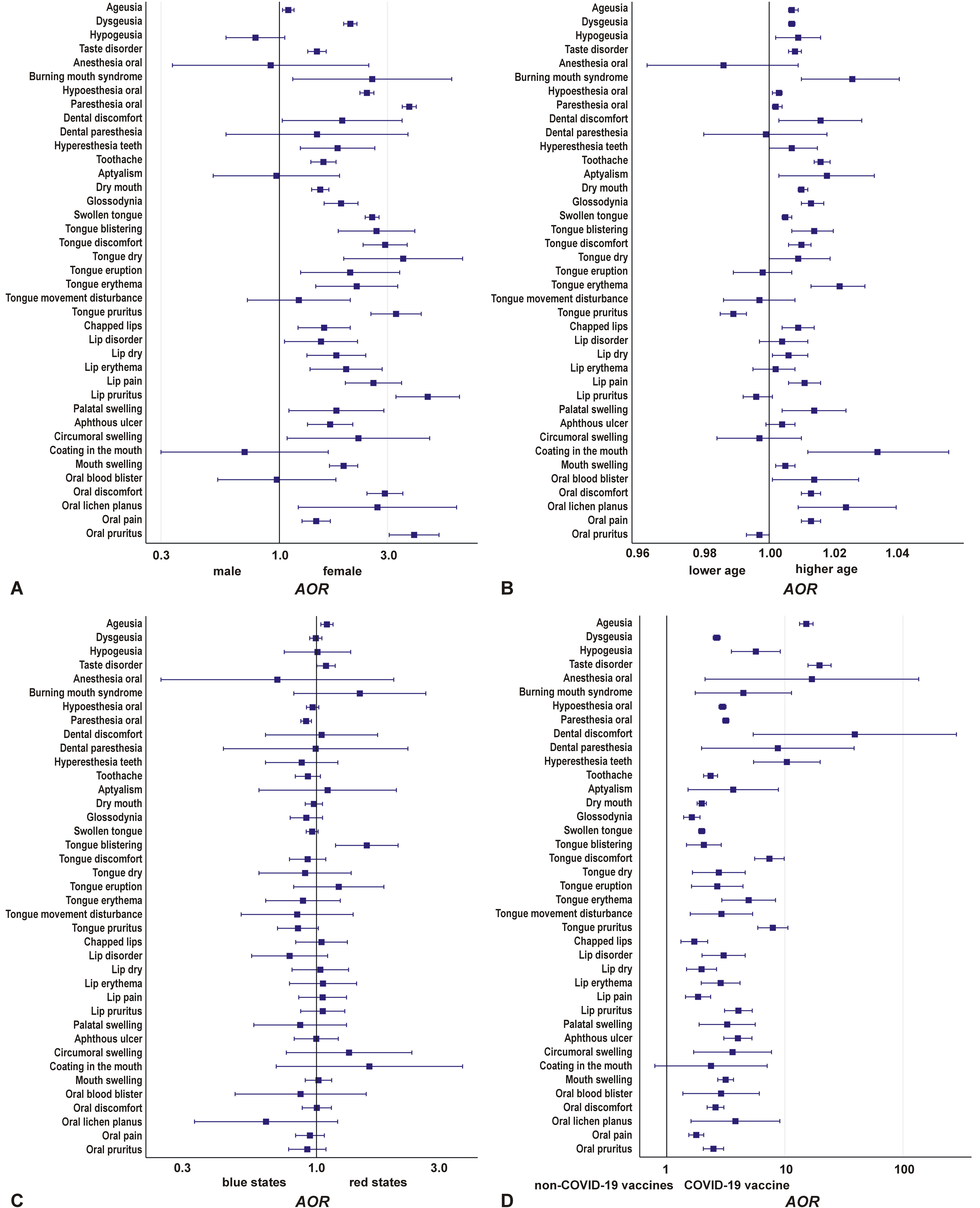
Binary logistic regression models were constructed for each true SDR, using sex (female vs. male), age (per year increase), state (red vs. blue), and vaccine type (COVID-19 vaccines vs. non-COVID-19 vaccines) as predictors to calculate adjusted odds ratios (AORs) with 95% CIs (Table 3).
Females had significantly higher odds for 32 SDRs, while no SDRs were more prevalent in males. Notably, elevated risks in females were observed for dysgeusia (AOR = 2.056 (1.921–2.199)), ageusia (AOR = 1.094 (1.032–1.160)), taste disorder (AOR = 1.464 (1.333–1.609)), oral paresthesia (AOR = 3.742 (3.485–4.019)), oral hypoesthesia (AOR = 2.433 (2.266–2.612)), dry mouth (AOR = 1.514 (1.387–1.653)), aphthous ulcer (AOR = 1.674 (1.330–2.108)), and oral lichen planus (AOR = 2.708 (1.211–6.055)), among others (Figure 4A).
Increasing age was associated with higher odds for 27 SDRs. For each additional year of age, the probability of developing coating in the mouth increased by 3.4% (1.2–5.7%), burning mouth syndrome by 2.6% (1.0–4.1%), oral lichen planus by 2.4% (0.9–4.0%), and tongue erythema by 2.2% (1.3–3.0%). These findings suggest a cumulative effect of age, where the likelihood of developing these SDRs progressively increases over time (Figure 4B).
State-level political inclination was not statistically significant for 36 SDRs. However, 3 SDRs showed significant variation: tongue blistering and ageusia were more prevalent in red states, whereas oral paresthesia was more common in blue states (Figure 4C).
For 38 SDRs, odds were significantly higher for COVID-19 vaccines compared with other vaccines, except for coating in the mouth (Figure 4D).
After adding time period to the models, sex-, age- or state-based associations remained stable, while some vaccine-based associations lost statistical significance, including oral anesthesia, burning mouth syndrome and oral lichen planus, among others. The pandemic period was associated with increased odds for 19 SDRs, including taste disorder, ageusia, oral paresthesia, and aphthous ulcer (Table 4).
Discussion
Key findings
The disproportionality analysis demonstrated that oral AEs were reported 2.40 times more frequently following COVID-19 vaccination than after all other vaccines in the VAERS database. Females and older adults exhibited higher PRRs, whereas severe, hospitalized and fatal cases showed substantially lower PRRs. Among the 39 true SDRs, taste-related AEs showed the highest PRRs, including taste disorder (23.96), ageusia (17.79), hypogeusia (6.25), and dysgeusia (3.22), alongside other sensory SDRs such as oral paresthesia (3.72) and oral hypoesthesia (3.50).
Sensitivity analyses confirmed that females exhibited a greater number of SDRs (32/39) than males (21/39). Middle-aged adults showed the highest number of true SDRs, whereas children (<5 years) and the elderly (≥80 years) exhibited the fewest. Non-severe (30/39), non-hospitalized (34/39) and non-fatal (36/39) cases accounted for substantially more SDRs than severe (8/39), hospitalized (2/39) and fatal cases (0/39). The mRNA-based (35/39) and viral vector-based (25/39) vaccines were associated with more SDRs than protein subunit COVID-19 vaccines (6/39) and influenza vaccines (0/39). Co-occurrence analyses demonstrated moderate agreement between gustatory and olfactory AEs (κ = 0.459), which increased for non-COVID-19 vaccines during the pandemic (κ = 0.500) compared with the pre-pandemic period (κ = 0.115). This pattern suggests the presence of notoriety bias rather than a causal association with COVID-19 vaccines.
Regression analyses confirmed that female sex was associated with significantly higher odds for 32 true SDRs, and that increasing age was associated with a higher probability of developing 27 SDRs. State-level political inclination had no significant influence on SDR reporting, except for 3 SDRs. The inclusion of time period as a predictor did not alter sex-, age- or state-based associations, indicating that notoriety bias did not substantially affect these patterns. However, adjustment for time attenuated the associations for several SDRs, particularly burning mouth syndrome, oral anesthesia and oral lichen planus, suggesting that reporting trends influenced the strength of association for certain AEs over time.
Sex- and age-related disparities
The higher susceptibility of females to reporting oral AEs following COVID-19 vaccination aligns with previous pharmacovigilance studies involving other organ systems.24 Disproportionality analyses using the WHO VigiBase database have consistently shown that females are at least twice as likely to report autoimmune,25 neurologic26, 27 and cutaneous AEs28, 29 following COVID-19 vaccination.25, 26, 27, 28, 29 This sex disparity may be attributed to both biological and behavioral factors.30 Sex hormones, particularly estrogen, enhance immune responses and contribute to greater reactogenicity, while chromosomal differences further modulate immune function.31 Additionally, higher baseline prevalence of certain conditions in females, coupled with increased health-seeking and reporting behaviors, may amplify these disparities.30
The observed age-related patterns are consistent with prior analyses indicating that adults, especially middle-aged individuals, are more susceptible to AEs following COVID-19 vaccination than minors (≤18 years) and seniors (≥64 years).25, 32, 33 Increase in AE reporting with age has been documented for conditions such as herpes zoster, myocardial infarction and circulatory collapse.28, 34 In contrast, some studies have identified a reversed pattern, where certain AEs, including myocarditis/pericarditis, thrombosis/thrombocytopenia, and anaphylaxis were more frequently reported in younger populations, while others found no significant age-related differences.35, 36, 37, 38, 39, 40
State-level political inclination
Political preferences have been shown to influence perceptions of vaccine safety.3, 41, 42, 43 A recent US cross-sectional study reported that while vaccinated individuals across political groups experienced similar AEs, Republicans were more likely to perceive these events as severe, report higher rates of negative reactions among their social circles, and discourage others from vaccination.41 The study highlights the role of political ideology in shaping vaccine perceptions even among the vaccinated.41 Consistently, Asch et al. performed a disproportionality analysis of COVID-19 vaccine AEs in VAERS, revealing that a 10% increase in Republican voting was associated with a 5% rise in overall AE reporting, a 25% increase in severe AE reports, and a 21% higher likelihood that any reported AE was classified as severe.42 Moreover, partisan divisions over COVID-19 vaccines are speculated to erode public support for other vaccination programs, including childhood immunization mandates.43 Media politicization may further reinforce these trends, as right-leaning outlets were more likely to emphasize the risks of COVID-19 vaccine-associated myocarditis (CVM) without contextualizing its rarity or comparing it to the risks of myocarditis from SARS-CoV-2 infection, whereas left-leaning media were highlighting the benefits of vaccination and provided numerical risk estimates to contextualize CVM concerns.3 In the present analysis, state-level political inclination was used as a proxy for political orientation, as previously described.42 No strong association was noted between political affiliation and oral AE reporting patterns.42
Vaccine schedule and type
Primary vaccination doses were associated with higher AE reporting rates than booster doses, which is consistent with previous evidence of greater reactogenicity among dose-naïve individuals.17, 44 The mRNA vaccines exhibited more SDRs than vector-based or protein subunit vaccines, which aligns with previous reports highlighting greater reactogenicity of mRNA platforms.45, 46
Are taste-related AEs a manifestation of notoriety bias?
The strong association between chemosensory disorders, particularly gustatory and olfactory dysfunction, and the SARS-CoV-2 infection may have increased public vigilance of these symptoms due to their diagnostic and prognostic relevance.13, 14, 15 As a result, sporadic cases of gustatory and olfactory dysfunction following COVID-19 vaccination have been reported.16, 47 In response to this heightened suspicion, active surveillance studies funded by the European Medicines Agency (EMA), including SAFETY–VAC, COVID-19 Vaccine Monitor (CVM), and Early COVID-19 Vaccine Monitor (ECVM), included anosmia and ageusia among their AEs of interest.48, 49 Surprisingly, evidence from these cohort studies indicated that the observed rates of anosmia and ageusia decreased following COVID-19 vaccination compared to the background rates, which had been severely elevated during the COVID-19 pandemic.50, 51, 52
In passive surveillance databases, chemosensory AEs were increasingly reported following COVID-19 vaccination, raising alternative hypotheses beyond a direct causal link to the vaccine, including coincidental or breakthrough infection.7, 8, 9 Both scenarios may be influenced by notoriety bias. Our findings partially support the notoriety bias hypotheses, as we observed a weak co-occurrence of gustatory and olfactory AEs before COVID-19, which became more pronounced during the pandemic for both COVID-19 and non-COVID-19 vaccines. Also, the crude reporting rate of taste-related AEs increased several-fold during the pandemic.7 Furthermore, the strengthened correlation between Google search activity and VAERS reporting after vaccine rollout was observed.
Strengths
The present study has several strengths. The hybrid analytical approach employed is strongly recommended, as no single data-mining method is inherently superior; therefore, combining multiple approaches is essential for identifying the most reliable SDRs.4, 53 Additionally, comprehensive stratification and sensitivity analyses across sex, age, clinical course, political inclination, vaccine type, and time period offer deeper insights into oral AE patterns and help identify potential reporting biases.4 The suspected notoriety bias associated with taste-related AEs was investigated using an innovative framework that incorporated stratified co-occurrence analysis and real-time digital search data. Moreover, this study is among the few to examine the role of political determinants in AE reporting.42
Limitations
This study has several limitations. First, reliance on passive surveillance data from VAERS is subject to inherent biases, including underreporting, overreporting and the inability to establish causal relationships between vaccines and reported AEs.12 As with all disproportionality analyses, causality cannot be inferred from statistical associations alone. Second, while the hybrid frequentist–Bayesian approach enhances the robustness of signal detection, disproportionality analysis alone cannot determine the clinical relevance or biological mechanisms underlying observed SDRs.5 Third, state-level political inclination was used as a proxy for individual political preferences, which may not fully capture the complexity of political influences on AE reporting.42 Finally, Google Trends data was utilized as an external indicator of notoriety bias. However, extensive media coverage and heightened public awareness, especially during the pandemic, may have contributed to the overestimation of certain SDRs, especially taste-related AEs.
Conclusions
The findings of the present study underscore the need for granular evaluation of clinically relevant oral SDRs, such as oral lichen planus, using self-controlled case-series methodologies to validate them as true safety signals.54 Given the potential role of notoriety bias in chemosensory AE reporting, future disproportionality analyses should incorporate sensitivity analyses and innovative analytical approaches to minimize its impact. Furthermore, as most oral AEs were strongly associated with favorable clinical outcomes (non-severe, non-hospitalized and non-fatal), an important public health message emerges: although oral AEs following COVID-19 vaccination may occur, they should not be perceived as life-threatening or abused by antivaccine propaganda to undermine public confidence in vaccine safety.
Ethics approval and consent to participate
Not applicable.
Data availability
Publicly available datasets were analyzed in this study. This data can be found at https://vaers.hhs.gov/data/datasets.html. The datasets supporting the findings of the current study are openly available in the Open Science Framework (OSF) at https://doi.org/10.17605/OSF.IO/3S8KW.
Consent for publication
Not applicable.
Use of AI and AI-assisted technologies
Generative AI was used in the preparation of the manuscript. Specifically, ChatGPT (v. 4; OpenAI, San Francisco, USA) was employed for proofreading and language refinement. This tool was used solely to improve grammar and readability. All substantive content was independently developed by the authors.