Abstract
Background. Periodontal disease is a condition that impacts the tissues surrounding the teeth, sparked by the build-up of bacteria leading to inflammation. These bacteria form biofilms, shielding themselves from antimicrobial treatment. Typically, mechanical cleaning disrupts these biofilms and lowers bacterial levels. However, in some cases, it might not fully address the disease due to intricate root structures and irregular lesion shapes. Local drug delivery directly to the affected area serves as a reservoir for drugs, potentially altering harmful bacteria and aiding in the healing process.
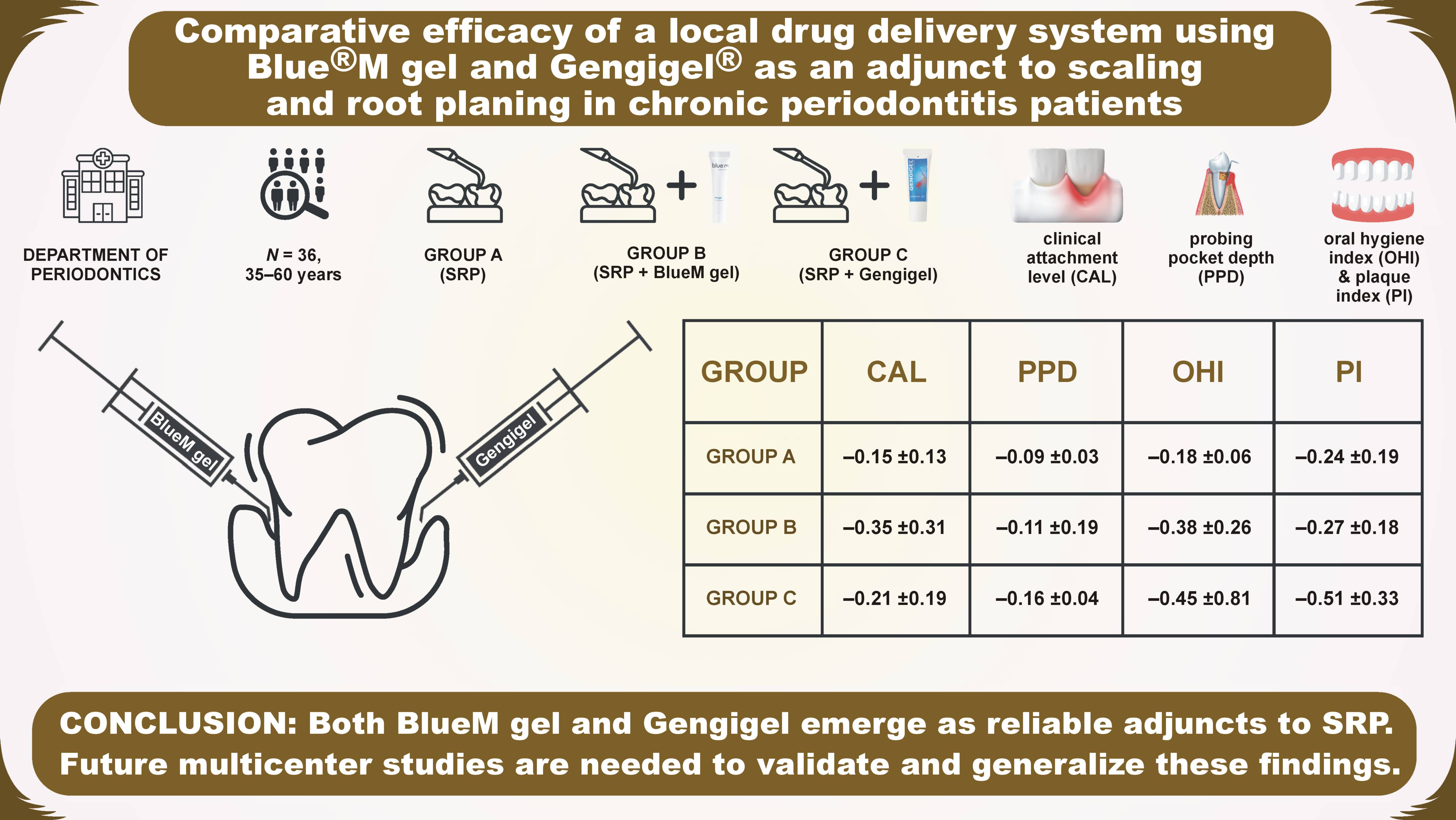
Objectives. The aim of the study was to evaluate and compare the effectiveness of various local drug delivery systems, specifically Blue®M gel along with scaling and root planing (SRP), Gengigel® with SRP, and SRP alone, in managing patients with chronic periodontitis.
Material and methods. The study population comprised 36 patients of both sexes in the age group of 35–60 years, with supra-bony pockets of 4–6 mm in any quadrant. The participants were randomly divided into 3 groups according to the treatment provided: group A – SRP alone; group B – SRP with BlueM gel; and group C – SRP with Gengigel. The gels were applied into the pockets. The follow-up period was 6 months.
Results. In a study involving 36 patients, predominantly males, with an average age of 42.03 years, Gengigel demonstrated a slightly greater reduction in the probing pocket depth (PPD) (−0.16 ±0.04 mm), the oral hygiene index (OHI) (−0.45 ±0.81) and the plaque index (PI) (−0.51 ±0.33) as compared to the other 2 groups. BlueM gel exhibited a superior reduction in the clinical attachment level (CAL) when compared to the control and Gengigel groups (−0.35 ±0.31 mm).
Conclusions. Both BlueM Gel and Gengigel have proven to be dependable choices or adjuncts to SRP. However, additional studies are needed to further explore their effectiveness.
Keywords: periodontitis, local drug delivery, scaling and root planing, Blue®M gel, Gengigel®
Introduction
Periodontitis is a persistent inflammatory condition that undermines the structural integrity of the tissues supporting the teeth. These tissues include the gingiva, the periodontal ligament and the alveolar bone, collectively called the periodontium.1 Chronic inflammation associated with periodontal disease compromises the integrity of the periodontal tissues, which is a crucial barrier against microbial invasion. This loss of integrity has adverse effects on the underlying periodontal tissues.2 The primary cause of periodontal disease is plaque on the surface of the tooth, causing the inflammation of marginal tissues; a condition known as gingivitis, which, if left untreated, may progress to periodontitis.3, 4 Surface root debridement, accepted as the most effective approach for treating periodontitis, involves removing the biofilm and bacterial products on the surfaces of the afflicted roots surfaces.5, 6 Despite its efficacy, scaling and root planing (SRP) might be technically challenging and inconsistent in eradicating all periodontal bacteria or reducing inflammation in periodontitis.2 Therefore, systemic and local antimicrobial therapies are often used as adjuncts to mechanical surface root debridement to enhance clinical outcomes.
Local drug therapy is frequently employed in managing periodontal disease, offering site-specific delivery of medications precisely where needed for an adequate duration. This approach ensures effective treatment without subjecting the entire body to therapeutic agents.7
Oxygen is a vital nutrient in cellular metabolism, particularly in energy generation. It is closely implicated in several physiological processes, including oxidative bacterial clearance, tissue re-epithelialization, angiogenesis, and collagen formation.8 Under controlled concentrations, reactive oxygen species (ROS) can transform into free radicals, promoting the growth of fibroblasts and epithelial cells. However, elevated concentrations of these free radicals may lead to tissue injury.9 Dr. Peter Blijdorp from the Netherlands10 developed a product named Blue®M based on active oxygen, comprising sodium perborate, glucose oxidase enzyme derived from honey, xylitol, and lactoferrin.11 These components are thoughtfully combined to harness the benefits of active oxygen while ensuring a balanced and controlled application, potentially supporting wound healing and tissue regeneration. Research by Koul et al.,9 Deliberador et al.11 and Asha et al.12 suggests that BlueM gel could be considered a viable alternative or adjunct to SRP in managing periodontal disease.
The antibacterial and anti-inflammatory properties of hyaluronic acid (HA) have positioned it as a valuable local chemotherapeutic agent in treating periodontal disease.13 Beyond its antibacterial efficacy, HA is recognized for its antifungal effects, contributing to its versatility in combating microbial infections. Additionally, it exhibits angiogenic and osteoinductive properties, further enhancing the healing process within the periodontal tissues.14 Gengigel®, which contains HA, is crucial in promoting the formation of blood vessels, the granulation tissue and osteoblasts.15 The role of HA is pivotal in modulating the fibrin network, the granulation tissue and the provisional matrix, which are critical to periodontal regeneration. A collective body of research, including studies by Mohammad et al.,5 Suchetha et al.,16 Sahayata et al.,17 Kalra et al.,18 and Sandhu et al.,19 has evidenced the therapeutic benefits of using HA. Notably, Rodríguez-Aranda et al.’s systematic review of 9 studies corroborated the efficacy of HA in improving clinical periodontal indices – the clinical attachment level (CAL), the probing pocket depth (PPD) and bleeding on probing (BoP) – both as standalone treatment and as a complement to surgical interventions like flap debridement, bone grafting, and the use of growth factors.14
Our study ventured beyond the individual assessments of BlueM gel and Gengigel, engaging in a direct comparison between the two. We evaluated their impact on the key periodontal parameters – PPD, CAL, the oral hygiene index (OHI), and the plaque index (PI). This study is the first to assess the efficacy of BlueM gel and Gengigel when each is combined with SRP, as well as SRP alone. The study aimed to provide insights into the potential benefits of these interventions in enhancing the outcomes of periodontal therapy.
Material and methods
Preliminary plan and ethics statement
This study was a prospective, randomized controlled trial (RCT), conducted at MNR Dental College and Hospital, Sangareddy, India, with the approval of the institutional review board (IRB) (MNR EC-BHR-07/23). The participants were fully briefed about the study and consented in writing. Employing randomization minimized variability and bias, ensuring standardized test site selection across the groups from the outset and enhancing the reliability of the results.
Inclusion and exclusion criteria
Patients were selected from the ambulatory care unit of the Department of Periodontology at MNR Dental College and Hospital in accordance with the guidelines of the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions.20 The inclusion criteria comprised individuals aged 35–60 yars of both genders, diagnosed with chronic periodontitis (stage II grade B periodontitis). Eligible patients presented with at least one site with PPD of 4–6 mm and CAL of 4–6 mm, along with horizontal bone loss, necessitating non-surgical treatment. The study excluded individuals with systemic diseases, those on antibiotics or anti-inflammatory medications, smokers, tobacco or alcohol users, immunocompromised persons, pregnant or lactating women, non-compliant or physically challenged individuals, and those unable to maintain good oral hygiene or attend follow-ups.
Sample size calculation
Sample size calculation was based on a study by Manjunatha et al.21 A one-way analysis of variance (ANOVA) with fixed effects was employed to assess the data. An a priori analysis was conducted to calculate the necessary sample size, using an effect size of 0.55, an alpha error probability of 0.05, and a power of 0.80 across 3 groups. This analysis yielded a required total sample size of 36, divided evenly into 12 participants per group. The output indicated a non-centrality parameter (λ) of 10.89, a critical F-value of 3.285, numerator degrees of freedom (dfn) of 2, denominator degrees of freedom (dfd) of 33, and an actual power of 0.813.
Estimation of clinical parameters
After the initial screening, all patients underwent supragingival full-mouth SRP. A total of 36 patients meeting the inclusion and exclusion criteria were chosen. The PI,22 OHI,23 PPD (measured with the UNC-15 periodontal probe), and CAL values were recorded at baseline and 6 months post-treatment.
Randomization
The allocation of 36 sites from 36 patients was randomized using the Research Randomizer software, v. 2.0 (https://www.randomizer.org).
Gel composition
A qualified clinician administered the allocated treatment plan to the selected sites for each group. The control group received treatment with SRP using UNC-15. In contrast, the experimental group sites were treated with SRP and either BlueM gel or Gengigel.
BlueM gel (Bluemcare India; New Delhi, India) composition: aqua; alcohol; glycerin; silica; sodium saccharin; sodium perborate; citric acid; PEG-32; sodium gluconate; lactoferrin; xanthan gum; cellulose gum.
Gengigel (Ricerfarma, Milan, Italy) composition: aqua; xylitol; cellulose gum; alcohol; PEG-40 hydrogenated castor oil; poly(vinyl) alcohol (PVA); polycarbophil; dichlorobenzyl alcohol; aroma (CITROMINT 1/074600); sodium hydroxide; Acid Blue 9 (CI 42090).
Intracrevicular administration of BlueM gel and Gengigel
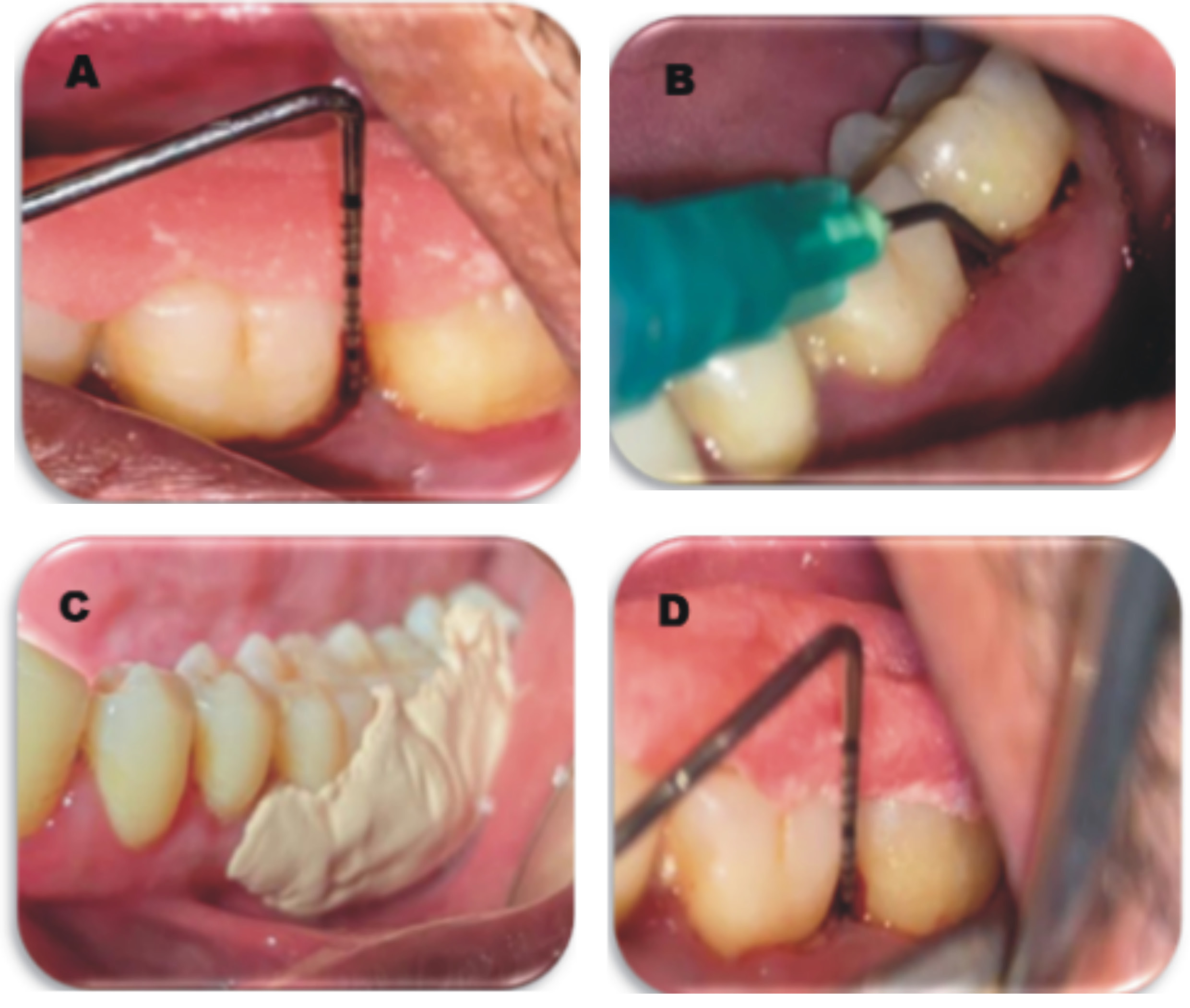
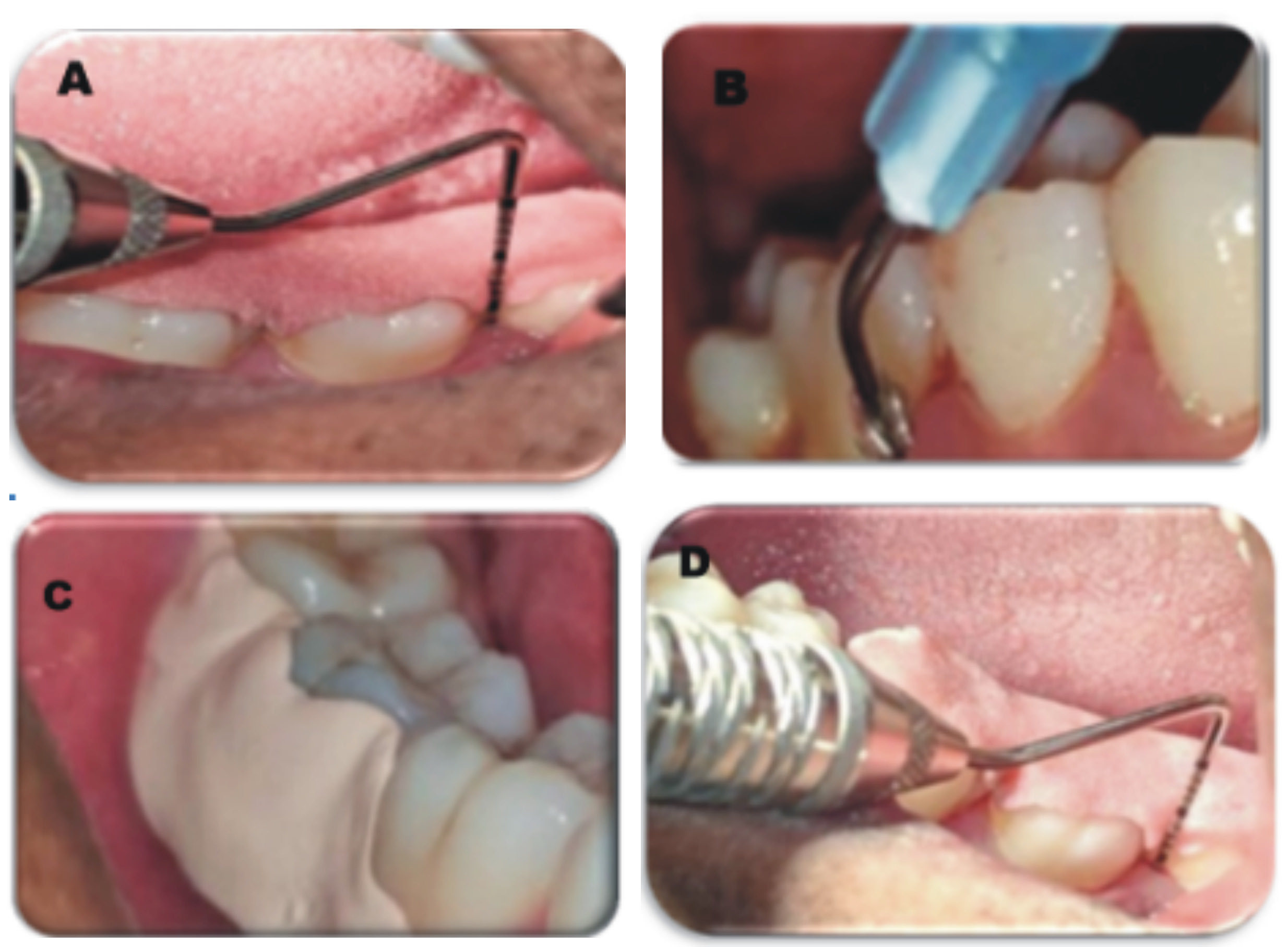
The study utilized BlueM gel and Gengigel as the study materials. The dental arches were divided into 4 quadrants, each receiving a different treatment modality: group A – SRP alone; group B – SRP with BlueM gel; and group C – SRP with Gengigel. All patients in the 3 groups underwent a pre-surgical protocol that included the collection of pretreatment records, such as detailed medical and dental histories, thorough clinical examinations, clinical photographs, and essential laboratory investigations. The gels were loaded into a syringe and placed intra-sulcularly in the selected area. The site was then covered with a periodontal Coe-Pack, and the patients were instructed to refrain from eating coarse foods, drinking, brushing, or rinsing the treated area for 24 h following application (Figure 1 and Figure 2).
Statistical analysis
All statistical calculations were performed using IBM SPSS Statistics for Windows, v. 21.0 (IBM Corp., Armonk, USA). All values were presented as mean and standard deviation (M ±SD). ANOVA and paired t tests were used for the comparative analysis of the data.
Results
Among the 36 patients, 19 (52.78%) were male and 17 (47.22%) were female, with the average age of 42.03 ±8.70 years. The PPD depth measurement was conducted at the drug delivery site, with the initial value recorded during the baseline visit, on the day of drug delivery. Subsequently, another PPD measurement was taken and recorded during the reassessment visit (Figure 1 and Table 2). A paired t test with a 95% confidence interval (CI) was performed to compare the mean difference in PPD between the BlueM gel and the Gengigel groups. The results indicated a statistically significant p-value for the mean difference between baseline and the follow-up at 6 months with regard to PPD (Table 1), CAL (Table 2), OHI (Table 3), and PI (Table 4).
All 3 groups showed a significant reduction in PPD at 6 months post-treatment, with group C (Gengigel) demonstrating a slightly greater reduction than the other 2 groups. The mean reduction in the Gengigel group was −0.16 ±0.04 mm, followed by −0.11 ±0.19 mm in the BlueM gel group, and −0.09 ±0.03 in controls.
After the 6-month period, all 3 groups demonstrated a noteworthy reduction in CAL. Group B (BlueM gel) exhibited a slightly superior reduction than the other 2 groups. Specifically, the mean reduction in group B was −0.35 ±0.31 mm, group C recorded −0.21 ±0.19 mm, and group A displayed −0.15 ±0.13 mm.
At the reassessment visit, all 3 groups exhibited a substantial reduction in OHI. The Gengigel group (C) notably demonstrated a slightly superior reduction than the BlueM gel and control groups. The mean reduction in group C was −0.45 ±0.81, group B showed −0.38 ±0.26, and group A exhibited −0.18 ±0.06.
Upon completing the 6-month period, a substantial reduction in PI was observed in all 3 groups. Group C (Gengigel) showed a slightly superior reduction than the other 2 groups. The mean reduction in group C was −0.51 ±0.33, while group B showed −0.27 ±0.18, and group A exhibited −0.24 ±0.19.
Discussion
The foundation of periodontal therapy lies in non-surgical mechanical treatment, which represents the primary and recommended approach for controlling periodontal infections. Additionally, the local subgingival application of chemotherapeutic agents can augment the clinical and microbiological benefits of mechanical debridement. Oral hygiene products supplemented with chemotherapeutic agents, offering diverse antimicrobial actions, have been shown to be effective adjuncts when used alongside mechanical plaque control.24
In the present study, all 3 treatment modalities resulted in a significant reduction in the clinical parameters. Clinical studies have demonstrated the osteoinductive properties of hyaluronan, as it stimulates osteoprogenitor cells within the defect. This stimulation leads to successive differentiation into osteoblasts and the formation of new bone.5, 25 A systematic review by Rodríguez et al., which included 9 studies, reported that the adjunctive application of HA yielded moderate but favorable clinical and radiographic outcomes in the surgical treatment of periodontal defects.14 Suchetha et al. reported that the use of Gengigel significantly reduced the PI scores (p < 0.001), from baseline (2.19 ±0.41) to 1 month (1.33 ±0.17) and 3 months (1.10 ±0.07),16 while in the present study, Gengigel showed a reduction of −0.51 ±0.33. The PPD (7.70 ±1.07 mm vs. 5.35 ±0.75 mm) and CAL (8.00 ±1.01 mm vs. 5.85 ±0.81 mm) scores observed by Suchetha et al. at baseline and a 90-day follow-up, respectively,16 were higher than in the present trial. A significant reduction in the mean PPD between baseline (4.38 ±0.62 mm) and 6 weeks (3.19 ±0.75 mm), and then 3 months (2.81 ±0.66 mm) was noted in a study by Shergill et al.26 In the current study, the mean difference in PPD from baseline to follow-up after the application of Gengigel was −0.16 ±0.04 mm. In our study, Gengigel also showed significant improvement in PI (−0.51 ±0.33), while a study conducted by Sahayata et al. reported non-significant improvement (3.16 ±0.42 vs. 2.30 ±0.36) when compared to placebo and scaling.17 Kalra et al.18 and Sandhu et al.19 used Gengigel along with the amniotic membrane and platelet-rich fibrin, respectively, showing promising results. A study by Gupta et al. reported a mean reduction in PPD of −1.33 ±0.50 mm from baseline to 6 months, along with a reduction in PI and a mean gain in relative attachment level (RAL) of 1.78 ±0.83.27 An RCT conducted on 14 patients by Fawzy El-Sayed et al. in 2012 showed that the application of hyaluronan gel in conjunction with periodontal surgery brought improvement in the PI, CAL (5.50 mm vs. 2 mm) and PPD (5 mm vs. 2 mm) values over time.28 The outcomes observed in the present study might stem from the reduction of gingival inflammation, which led to the contraction and reconnection of connective tissue fibers, as well as a decrease in inflammatory mediators.
Addressing the global crisis of antibiotic resistance primarily involves minimizing antibiotic usage. Addressing the global crisis of antibiotic resistance primarily involves minimizing antibiotic use. Reactive oxygen species represent a promising alternative, as they can be administered directly to the affected area through topical application, thereby reducing the reliance on systemic antibiotics.11 The notable reduction in PPD observed at the sites treated with BlueM gel is attributed to the increased release of active oxygen, which promotes rapid and progressive healing. Juliana and Tarek’s research found that BlueM oxygen gel accelerated re-epithelialization, likely due to its role in promoting neovascularization, enhancing new blood vessel formation, and stimulating stem cell activity that supports fibroblast proliferation.29 In a study by Koul et al., the reported reduction in PPD after using BlueM gel was 2–3 mm at 30 days,9 which is less than in our study (−0.11 ±0.19). In the current investigation, BlueM gel demonstrated more significant efficacy in reducing CAL than the other 2 interventions. In a study conducted by Niveda and Kaarthikeyan,30 the decrease in PPD observed from baseline (7.2 ±0.42 mm) to follow-up (4.7 ±0.57 mm) was greater than in the present investigation, where the mean reduction was −0.11 ±0.19. Research by Rajkhowa et al. showed that BlueM gel helped reduce both PI and OHI, and thus could be safely used to reduce the microbial load.31 In a case report by Asha et al., BlueM gel was locally delivered for 7 days to treat periodontal pockets.12 The patients were evaluated after 30 days, revealing a moderate reduction in PPD.12
Limitations
The study was conducted over a 6-month period with a relatively small sample size. This duration may be insufficient to comprehensively evaluate the effects of periodontal therapy, especially those involving regenerative techniques. Therefore, further research with a longer follow-up period and a larger sample size is warranted to assess the long-term stability of the outcomes.
Conclusions
The present study demonstrated that both BlueM gel and Gengigel are reliable adjuncts to SRP. Both agents effectively enhance periodontal health when used in combination with SRP, with Gengigel showing greater reductions in PPD, OHI and PI, while BlueM gel achieved superior improvement in CAL. Notably, this study is the first to assess the efficacy of both gels in this context. Future multicenter studies are needed to confirm and extend these findings across diverse populations and clinical settings. Additionally, comparative evaluations of this protocol against other established and clinically proven agents are warranted for a comprehensive assessment.
Ethics approval and consent to participate
The study was approved by the institutional review board (IRB) at MNR Dental College and Hospital, Sangareddy, India (MNR EC-BHR-07/23). The participants were fully briefed about the study and consented in writing.
Data availability
The datasets supporting the findings of the current study are available from the corresponding author on reasonable request.
Consent for publication
Not applicable.
Use of AI and AI-assisted technologies
The authors of this manuscript declare that in the writing process of this work, no generative artificial intelligence (AI) or AI-assisted technologies were used to generate content, ideas or theories. We utilized Grammarly for the purpose of enhancing readability and refining language. This use was under strict human oversight and control. After the application of AI technologies, the authors carefully reviewed and edited the manuscript to ensure its accuracy and coherence.