Abstract
Indian spice curcumin, which has anti-tumor, anti-inflammatory and antioxidant effects, has showed its potential as an innovative adjunct for the treatment of oral squamous cell cancer (OSCC), as well as an intriguing chemopreventive drug. The goal of this review was to consolidate the salient characteristics of curcumin and its cutting-edge nanoformulations, and to further outline the role of curcumin in the management of OSCC.
The PubMed, Scopus and Cochrane databases were used to search for evidence-based research papers on curcumin.
The current systematic review included 35 publications. There were 5 clinical studies and 30 cell line studies. The included studies employed a wide range of OSCC cell lines, with CAL-27 in 6 studies, KB in 4 studies, FaDu in 3 studies, and SCC-9 in 3 studies, being the most common. Each of the entailed study found that when cell lines were treated with curcumin, there was an overall decrease in the proliferation of cells and cell growth when measured by the MTT assay, the luciferase assay and immunofluorescence. In clinical studies, APG-157 could inhibit tumor cell death by lowering the concentrations of NF-κB-driven cytokines that induce inflammation. The WCRF International/UoB framework-recommended quality assessment of cell line studies regarded 6 studies as high-quality and 3 studies were deemed of moderate quality.
The novel formulations of curcumin have been explored for its usefulness in the management of oral cancer, with promising results.
Keywords: apoptosis, nanoformulations, curcumin, oral squamous cell carcinoma, demethoxycurcumin
Introduction
Indian spice curcumin, which has anti-tumor, anti-inflammatory and antioxidant effects, has showed its potential as an innovative adjunct for the treatment of oral squamous cell cancer (OSCC), as well as an intriguing chemopreventive drug.1 Curcumin has demonstrated its effectiveness against a variety of chronic conditions, including tumors, neurological illnesses, hepatic impairment, obesity-related disorders, and arthritic conditions. As stand-alone treatment or in conjunction with different therapies, curcumin is exceptionally effective in the management of cancer. Additionally, this organic substance has the ability to modulate a number of cellular receptors and signaling pathways connected with multiple cancers.2
Advanced-stage OSCC cells develop into tumors that are highly resistant to chemotherapeutic agents due to their ability to alter key biological processes. These adaptations can lead to therapeutic failure, including ineffective drug delivery to target sites, metabolic reprogramming, altered redox balance, and impaired DNA damage repair mechanisms. Over the past several decades, extensive research has focused on developing innovative strategies to treat cancer cells that are resistant to multiple therapies by blocking resistance pathways and employing modern anti-cancer agents. Curcumin, a naturally occurring compound, has attracted considerable attention, since structural modifications have enabled the manufacturing of analogs and derivatives with enhanced anti-cancer activity and relatively low adverse effects, contributing to the development of novel anti-tumor therapeutics.3
Curcumin exhibits a multifaceted anti-cancer mechanism of action by modulating numerous molecular pathways.4 It inhibits several components involved in cellular proliferation, including mitogen-activated protein kinases (MAPKs), c-Jun N-terminal kinases (JNKs) and cyclin-dependent kinases (CDKs), which are frequently overexpressed in tumor cells. Curcumin also reduces the expression of growth factor receptors, such as insulin-like growth factor (IGF), fibroblast growth factor (FGF) and epidermal growth factor (EGF). In addition, curcumin promotes apoptosis by upregulating the pro-apoptotic protein p53 while suppressing anti-apoptotic signaling molecules, including nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB), B-cell lymphoma 2 (Bcl-2), signal transducer and activator of transcription 3 (STAT3), and survivin. Its anti-angiogenic effects are attributed to its ability to inhibit the expression of pro-angiogenic factors, particularly hypoxia-inducible factor 1 (HIF-1), vascular endothelial growth factor (VEGF), and angiopoietin 1 (Ang-1). Curcumin also modulates the enzymes and proteins involved in tumor invasion and metastasis, such as transforming growth factor beta (TGF-β), matrix metalloproteinases (MMPs), cell adhesion molecules (CAMs), tissue plasminogen activator (tPA), and lysyl oxidase (LOX). Furthermore, curcumin can regulate the expression of microRNAs associated with tumor initiation, growth and progression. It exerts anti-inflammatory effects by decreasing the production of pro-inflammatory cytokines, including interleukin 1 (IL-1), IL-6 and tumor necrosis factor alpha (TNF-α), while increasing the anti-inflammatory cytokine IL-10, thereby helping to counteract the inflammation associated with certain malignancies. Finally, curcumin enhances immune-mediated tumor cell destruction by promoting the activity of natural killer (NK) cells and cytotoxic T lymphocytes (CTLs).5, 6, 7, 8, 9, 10, 11, 12
In addition to its biophysical and photochemical properties, the chemical structure of curcumin also contributes significantly to its therapeutic potential. In particular, the aromatic ortho-methoxyphenol groups and the α,β-diketo moiety play important roles in mediating its biological activities and associated health benefits. Two naturally occurring curcumin analogs, demethoxycurcumin and bisdemethoxycurcumin, have been widely investigated for their pharmacological properties.
Despite its promising biological effects, curcumin has several limitations, including poor bioavailability, low stability, and limited aqueous solubility, which restrict its broader clinical application. Consequently, numerous structural analogs and derivative compounds have been developed over time to overcome these drawbacks and enhance its therapeutic efficacy.13, 14
Nanotechnology-based approaches provide strategies to prolong drug dissolution, enable targeted drug delivery and enhance transport across biological barriers. By carefully modifying the chemical composition, size and structural characteristics that regulate nanoparticle activity, a wide range of nanoformulations has been developed using organic, inorganic, lipid, and polypeptide-based materials, typically within the size range of 1–100 nm. These nanomaterials have been widely utilized as carriers for various anti-cancer drugs.15, 16, 17, 18, 19, 20, 21
The goal of this review was to consolidate the salient characteristics of curcumin and its cutting-edge nanoformulations, and to further outline the role of curcumin in the management of OSCC.
Material and methods
Study protocol
This systematic review was performed in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines for study selection, synthesis and reporting of results.22 The study protocol was registered in the International Prospective Register of Systematic Reviews (PROSPERO) under the registration number CRD42023483768.
Focused question
The focus of this literature review was to investigate the inhibitory effects of curcumin and its nanoformulations on cellular proliferation in OSCC at the molecular level. The review examined studies that explore the molecular mechanisms underlying the anti-cancer activity of curcumin and its nanoformulations. In addition, it analyzes the outcomes of in vitro and in vivo studies that have evaluated the effects of curcumin and its nanoformulations on the proliferation of OSCC cells.
Information sources and search strategy
The PubMed, Scopus and Cochrane Library databases were used to identify evidence-based research articles investigating the cytotoxic effects of curcumin and its nanoformulations on OSCC, published between January 2012 and August 2023. Following an initial study of the available literature, the following Medical Subject Headings (MeSH) phrase combination was used: (“Curcumin” OR “Curcuminoids” OR “Nanocurcumin” OR “Curcumin Analog” OR “Curcumin Microemulsions” OR “Desmethoxycurcumin” OR “Bisdemethoxycurcumin” OR “Curcumin Nanomicelles”) AND (“Cisplatin-Resistant Human Oral Cancer Cells” OR “Cell Culture” OR “Cell Lines”) AND (“Oral Squamous Cell Carcinoma” OR “Head and Neck Squamous Cell Carcinoma” OR “Oropharyngeal Carcinoma”) AND (“MTA” OR “Flow Cytometry” OR “Annexin Assay” OR “Western Blot” OR “RT-PCR”) AND (“Apoptosis” OR “Cytotoxicity” OR “Interleukin” OR “Signaling Pathway” OR “Photodynamic” OR “Epithelial–Mesenchymal Transition”). A manual check of the references for each of the included publications was executed to find studies that were not found in the online databases. The search was done by two reviewers independently, and screening was completed using the pre-established parameters.
Eligibility criteria
The eligibility criteria utilized in the present systematic review were established based on the PICOS criteria (Population, Intervention, Comparison, Outcome, and Study Design).
Population: Patients with OSCC aged 18 years and above.
Intervention: Free curcumin, curcuminoids or its nanoformulations.
Comparison: Placebo control group, or the administration of other synthetic chemotherapeutic drugs or radiotherapy.
Outcome: In vitro cellular proliferation, cytotoxicity or in vivo tumor dimensions, or tumor incidence, apoptosis, cell cycle arrest, and/or the evaluation of protein expression.
Study design: In vitro or clinical studies to evaluate the cytotoxicity effects of curcumin and its nanoformulations on OSCC.
Systematic reviews, questionnaire-based surveys, commentaries, case reports or case series, pilot studies, animal studies, and expert opinions were excluded. Articles not published in English or those that did not focus on the anti-tumor effects of curcuminoids or their nanoformulations were also excluded. The literature search, data extraction from the included studies, and quality assessment were independently performed by two expert reviewers. Any disagreement was resolved through discussion, and when necessary, a third reviewer was consulted to reach a consensus.
Study selection
The titles and abstracts of all the identified studies were independently screened by two reviewers. Publications that did not meet the eligibility criteria were excluded. The full texts of the selected articles were then independently evaluated by the same reviewers according to the predefined eligibility criteria. In addition, the reference lists of the included studies were manually searched to identify any further relevant publications. Any disagreement between the reviewers was resolved through discussion. If consensus could not be reached, a third reviewer was consulted to make the final decision. The final selection of studies was determined through agreement among all three reviewers.
Data extraction
The following information was extracted using a standardized data collection template: first author’s name; study title; journal name; year of publication; country where the study was conducted; study design; assays and cell lines used; source of the cell lines; sample size and baseline characteristics; drug dosage; and key study findings. To minimize errors, data extraction was performed independently by two reviewers. When additional information was required, the corresponding author was contacted for clarification.
Quality assessment
The quality assessment of the included clinical studies was conducted using the Cochrane Collaboration Risk of Bias tool (RoB),23 which categorizes the risk of bias as high, low, or unclear. The tool evaluates 7 domains: sequence generation; allocation concealment; blinding of participants and personnel; blinding of outcome assessment; incomplete outcome data; selective reporting; and other potential sources of bias. A low risk of bias was assigned when all criteria were fulfilled. A moderate risk of bias was designated when one domain was not adequately met. A high risk of bias was assigned when more than one domain was not satisfied.
The quality of the cell culture studies was evaluated using the World Cancer Research Fund International/University of Bristol (WCRF/UoB) framework,24 which provides a scoring range from 0 to 6. A score of 0 was assigned to parameters that were not met or not reported. Studies were classified as low quality if they scored 0–2, moderate quality for scores of 3–4, and high quality for scores of 5–6. These scores reflected the overall study quality and were used to assess the degree of risk of bias for each cell culture study.
Results
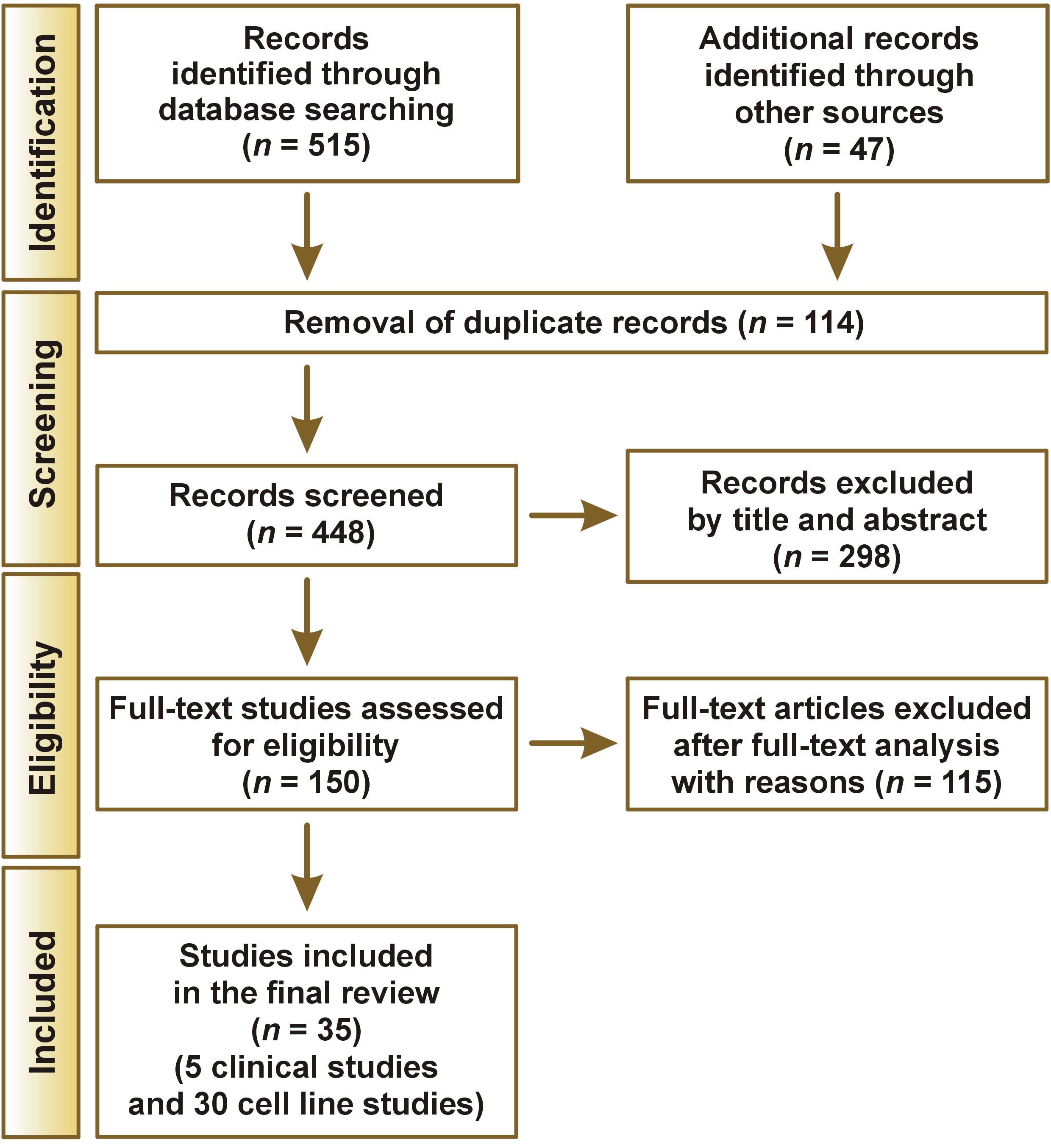
Figure 1 illustrates the stepwise procedure for study selection. A total of 515 studies meeting the inclusion criteria were initially identified from the aforementioned databases, and an additional 47 articles were retrieved through manual search. After removing 114 duplicate records, 448 publications remained for title and abstract screening, resulting in the exclusion of 298 articles. Consequently, 150 full-text articles were assessed for eligibility. During full-text review, 115 studies were excluded due to deviations from the predefined criteria. Ultimately, 35 publications were included in the present systematic review, as depicted in the PRISMA flow chart.25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59
Five clinical studies28, 29, 41, 51, 52 and 30 cell line investigations25, 26, 27, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 42, 43, 44, 45, 46, 47, 48, 49, 50, 53, 54, 55, 56, 57, 58, 59 were selected (Table 1, Table 2). Two of the 5 clinical trials were double-blind, randomized, placebo-controlled trials,29, 52 and one was single blinded.28 The included studies employed a wide range of OSCC cell lines, with CAL-27 in 6 studies,31, 33, 42, 43, 46, 55 KB in 4 studies,36, 39, 45, 49 FaDu in 3 studies,33, 42, 55 and SCC-9 in 3 studies35, 47, 56 being the most common (Table 2).
General characteristics
The selected studies were carried out in several countries, including India,28, 34, 39, 48, 49, 53, 54, 59 China,42, 43, 45, 46, 55, 56, 57, 58 the USA,29, 35, 41, 52, 55 South Korea,25, 36, 37 Taiwan,31, 40, 44 Brazil,33, 47 Germany,38, 51 Saudi Arabia,26 Italy,27 Spain,30 Egypt,32 and Japan.50 All of the studies were published in English between 2012 and 2023. The MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) assay, the Annexin assay for determining apoptosis, flow cytometry, the proliferation assay, Western blot, Northern blot, reactive oxygen species (ROS), real-time reverse transcriptase polymerase chain reaction (RT-PCR), the enzyme-linked immunosorbent assay (ELISA), and immune histochemistry (IHC) were employed in various studies. The curcumin doses utilized in the reviewed studies varied from 0 to 100 μm. Each of the entailed study found that when cell lines were treated with curcumin, there was an overall decrease in the proliferation of cells and cell growth when measured by the MTT assay, the luciferase assay and immunofluorescence. All of the in vitro studies had been conducted on human OSCC cell lines.
Role of curcumin in OSCC management
Curcumin has been shown to inhibit regulatory T cell (Treg) migration and induce S-phase cell cycle arrest. It has also been reported to reverse epithelial-mesenchymal transition (EMT), a process associated with tumor invasion and metastasis. Mishra et al. demonstrated that curcumin reduced cell viability and induced morphological changes in human papillomavirus (HPV)-positive oral cancer (OC) cell lines.48 Northern blot analysis indicated that curcumin suppressed the E6-oncogene-mediated degradation of p53 in these HPV-positive cancer cells. At a concentration of 100 μM, curcumin inhibited the expression of E6, NF-κB and activator protein 1 (AP-1) in HPV-positive cell lines.48 Additionally, Xiao et al. reported that curcumin enhanced the expression of miR-9, which reduces the proliferation of SCC-9 cells and suppresses the Wnt/β-catenin signaling pathway.56
Zhen et al. conducted an in vitro study showing that curcumin reduced the proliferation of SCC-25 cells and induced G2/M phase cell cycle arrest in a dose-dependent manner.57 The study also demonstrated that curcumin suppressed the expression of MMP-2 and MMP-9, as well as urokinase-type plasminogen activator (uPA) and its receptor (uPAR). Curcumin was found to modulate phosphorylated epidermal growth factor receptor (p-EGFR) and downstream signaling mediators, including protein kinase B (Akt), extracellular signal-regulated kinases 1 and 2 (ERK1/2) and STAT3. Additionally, curcumin inhibited EGF-induced EGFR phosphorylation and reduced EGF-stimulated SCC-25 cell invasion.57
Both Almalki et al.26 and Zhen et al.57 reported G2/M phase arrest in curcuminoid-treated cells after 6 h of treatment and demonstrated the ability of curcumin to induce DNA damage in these cells. According to Lu et al., curcumin exhibited improved solubility and stability when encapsulated in folate-linked liposomes.45 Chang et al. showed that curcumin-loaded nanoparticles induced apoptosis and significantly inhibited proliferation in OSCC cell lines while demonstrating lower cytotoxicity in normal cells, highlighting the specificity of nano-based drug delivery.31 Conversely, Mazzarino et al. observed that free curcumin (IC50 (half-maximal inhibitory concentration) = 93.4 µM) was more cytotoxic than curcumin nanoformulations (IC50 = 271.5 µM) after 24 h of treatment.47
Role of nanocurcumins in OSCC management
Several innovative strategies have been developed to design curcumin-based nanomaterials as targeted drug-delivery systems. Polymer nanoparticles are particularly effective due to their excellent biocompatibility and prolonged circulation in the bloodstream. Srivastava et al. reported that curcumin nanoparticles exhibited chemoprotective effects in OC cells, mitigating the adverse reactions typically induced by 5-fluorouracil.59 Nanocurcumin also demonstrated antioxidant properties and modulated the expression of apoptotic proteins, including Bcl-2 and Bcl-2-associated X protein (Bax), in the treated cells.59
Another study investigated the anti-tumor efficacy of gefitinib and curcumin-loaded nanoparticles in human OC SAS cells in vitro and in SAS xenograft tumors in vivo.40 The results showed that γ-polyglutamic acid (γ-PGA)-coated gefitinib/curcumin nanoparticles were effectively internalized by SAS cells, significantly reducing viable cell counts. Both free Gefitinib/curcumin and γ-PGA-gefitinib/nanocurcumin induced apoptotic cell death via caspase-3, caspase-9 and mitochondria-mediated pathways. In vivo experiments further demonstrated that γ-PGA-gefitinib/nanocurcumin significantly inhibited tumor growth as compared to the untreated or free gefitinib/curcumin-treated groups.40
Essawy et al. investigated the effects of curcumin nanoparticles on OC cells, using a simple and cost-effective solvent–antisolvent precipitation method.32 Unlike doxorubicin, which primarily induced necrosis in these cell lines, curcumin nanoparticles triggered cytotoxicity predominantly through apoptosis. The study also highlighted the intrinsic luminescence of curcumin nanoparticles, suggesting their potential as dual-function theranostic agents.32
Another study reported that the combined treatment with 200-nm nanocurcumin and cetuximab enhanced cytotoxicity as compared to cetuximab monotherapy, indicating the potential chemo-adjuvant role of curcumin nanomaterials in OC therapy.49
De Souza Ferreira et al. developed nanostructured gel formulations, incorporating curcumin for OC treatment.33 These formulations facilitated the rapid absorption and localization of curcumin in the hydrophobic core of nano-sized polymeric micelles, resulting in prolonged retention in the oral cavity.39 Cytotoxicity assays revealed that these nanoformulations selectively targeted tumor cells while sparing normal cells.33 Such strategies improve the physicochemical properties of curcumin by enhancing its solubility and tissue penetration, thereby augmenting its anti-cancer activity. Given these promising findings, further preclinical and clinical studies are warranted to evaluate the therapeutic potential of novel curcumin formulations.
Lin et al. conducted a study to evaluate the effects of curcumin microemulsions on OC cell lines.44 The results demonstrated that short-term treatment with curcumin in microemulsion form exerted significant cytotoxic effects on cancer cells. In SCC-25 cells, these effects were further enhanced when ultrasound was applied. This enhancement was likely due to the improved cellular delivery of curcumin, facilitated by the interaction of microemulsion particles with cell membranes or by overcoming transport barriers through ultrasound-induced membrane permeabilization and localized heating.44 When combined with ultrasound, these biodegradable microemulsions may provide controlled drug release and improved tissue-targeting capabilities. Additionally, curcumin nanoparticles have been shown to possess significantly higher bioavailability and improved aqueous solubility as compared to conventional curcumin, which may enhance their therapeutic potential.
Chang et al. demonstrated that water-soluble poly(lactic-co-glycolic acid) (PLGA)-based nanocurcumin enhanced therapeutic efficacy in CAL-27 cisplatin-resistant (CAR) OC cells.31 The nanocurcumin formulation promoted the generation of ROS and increased the expression of apoptosis-related molecules, including cleaved caspase-3/caspase-9, apoptotic protease activating factor 1 (Apaf-1), cytochrome c, and Bax, while reducing the expression of the anti-apoptotic protein Bcl-2. These effects collectively activated the intrinsic apoptotic pathway in CAR cells.31 Additionally, nanocurcumin modulates the structure and activity of multidrug-resistance proteins while enhancing ROS production, thereby contributing to increased cancer cell susceptibility to treatment.53 Organically modified silica nanomaterials used as carriers for curcumin delivery in OC cells demonstrated improved cellular uptake of curcumin and enhanced phototoxic effects in cancer cells, highlighting the potential of silica-based nanocarriers to improve the anti-cancer activity of curcumin.25, 53
Mazzarino et al. evaluated the therapeutic potential of mucoadhesive polycaprolactone curcumin nanostructures coated with chitosan for the treatment of OC.47 Ghosh et al. developed a multifunctional nanoconjugate by integrating the surface of gold nanorods (GNRs) with a cytotoxic nucleoside, 5-fluoro-2’-deoxyuridine (FdU), incorporated into a DNA hairpin structure along with a hydrophobic curcumin complex.34 The study demonstrated that curcumin could be successfully incorporated into small DNA hairpins to improve drug delivery efficiency. When combined with FdU nucleotides, this system significantly enhanced cytotoxicity in SCC-131 cells, suggesting its potential application as a therapeutic strategy for advanced-stage cancers.34
Clinical trials on curcumin in OSCC
The present review reported elevated levels of cytokines in the salivary supernatant of cancer patients, with reduced concentrations observed following treatment with APG-157, particularly for the pro-inflammatory cytokines IL-1 and IL-8. These findings suggest that APG-157 may suppress tumor-associated inflammation by reducing levels of NF-κB–regulated cytokines that contribute to inflammatory responses.29
Reiter et al. also demonstrated significant DNA damage caused by smoke condensate, primarily as a result of oxidative stress.51 In this context, curcumin and celecoxib were shown to provide protective effects against such DNA damage.51
In the study conducted by Arun et al., 21 patients with OSCC were randomly allocated into 2 groups – group A, comprising 12 patients, and group B, comprising 9 patients.28 The baseline tumor dimensions were assessed using contrast-enhanced computed tomography (CECT). After 3 months of treatment, patients were re-evaluated using CECT scans, and treatment responses were assessed according to the Response Evaluation Criteria in Solid Tumors (RECIST), which categorize outcomes as complete response, partial response, progressive disease, or stable disease.28
Latimer et al. reported a significant increase in the serum levels of microgranular curcumin administered via the transmucosal route within 30 min of ingestion in both cancer patients and healthy volunteers.41
Quality assessment of the studies reviewed
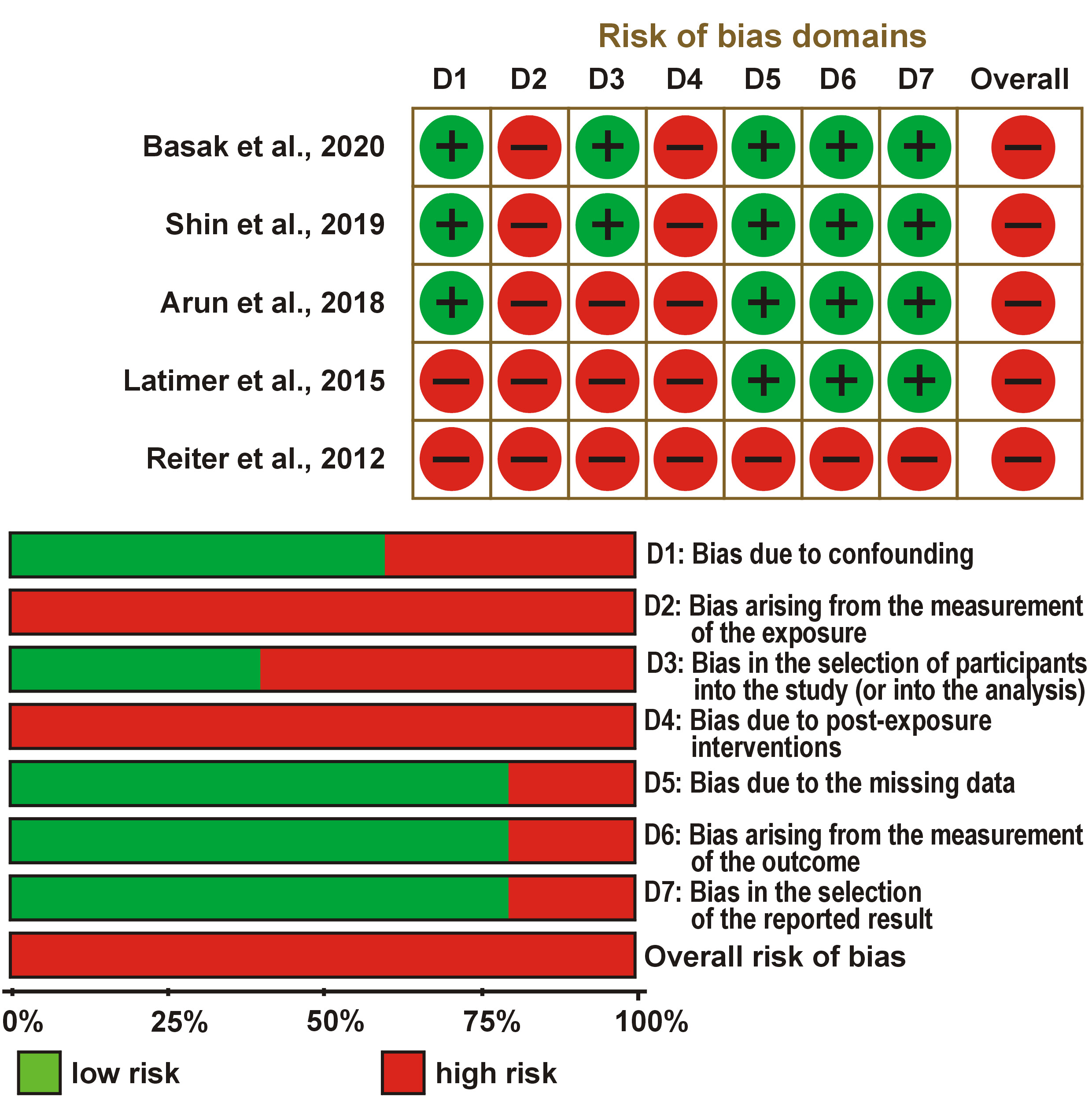
When evaluated using the RoB, all the included clinical trials were considered to have a high risk of bias (Figure 2). This classification was primarily due to the incomplete reporting of the key methodological aspects, particularly allocation concealment and blinding procedures, which are essential components of rigorous quality assessment.
Using the WCRF/UoB framework for evaluating cell line studies, 25 studies were rated as high quality, achieving scores between 5 and 6, while 5 studies were categorized as moderate quality with scores ranging from 3 to 4 (Table 3).
Discussion
Curcumin exhibits antimicrobial, antifungal, antiviral, anti-inflammatory, and pro-apoptotic properties. Despite significant advancements in chemotherapy and an improved understanding of the molecular pathophysiology of OC, the survival rates have remained relatively low, ranging from approx. 50% to 59% in recent years.60 Curcumin has been extensively investigated in numerous in vivo and in vitro models of OSCC worldwide. Studies have reported that curcumin reduces the proliferation of OSCC cell lines in a dose-dependent manner in both in vitro and in vivo settings.61
Curcumin modulates multiple signaling pathways in cancer cells, reduces the expression of the proteins associated with therapeutic resistance and enhances the efficacy of anti-cancer drugs at various stages of treatment. It may also inhibit drug resistance mechanisms by increasing the sensitivity of chemotherapy-resistant cells.62 Nanotechnology offers promising opportunities for developing medications with nanoscale dimensions, and nanocarrier-based drug delivery systems are being explored to improve the physicochemical properties and therapeutic efficacy of the drugs administered to these patients.60
Curcumin has been shown to suppress cellular proliferation and tumor metastasis while promoting apoptosis in OC cell lines through the modulation of multiple signaling pathways.7 The synergistic effects of curcumin in combination with the established chemotherapeutic agents, radiotherapy, the emerging therapeutic strategies, or other plant-derived compounds have been widely investigated across different tumor types. These studies have reported enhanced therapeutic benefits as compared to the conventional kinds of treatment alone.
Similar findings have been obtained in OSCC cell lines and experimental animal models when curcumin was combined with epigallocatechin gallate (EGCG), resveratrol and photodynamic therapy (PDT). Moreover, the interaction of curcumin with radiotherapy or chemotherapeutic agents such as cisplatin and 5-fluorouracil – commonly used in OSCC treatment – has been shown to significantly enhance anti-cancer efficacy. These outcomes are consistent with the conclusions of a previous systematic review.63
The development of innovative photodynamic nanotechnologies incorporating precise anti-cancer nanotheranostic systems has attracted considerable interest over recent decades. A novel strategy has been developed using tumor microenvironment–responsive, near-infrared–sensitive GNR drug conjugates to enable integrated plasmonic photothermal therapy for cancer treatment.64
Multiple preclinical studies have demonstrated the anti-cancer potential of curcumin in various cancers. Curcumin has been shown to induce G2/M phase cell cycle arrest, trigger apoptosis and autophagy, and disrupt the key molecular signaling pathways involved in tumor progression. It also interferes with tumor cell invasion and metastasis, thereby enhancing the therapeutic efficacy of currently used chemotherapeutic agents. Furthermore, the suppression of pro-inflammatory cytokines by curcumin may significantly reduce the inflammation associated with cancer progression. Curcumin also participates in additional signaling mechanisms, including the induction of neutrophil activation and degranulation through increased cell surface expression of markers such as CD35, CD66b and CD63.65
Several strategies related to drug formulation and delivery systems have been developed to overcome the limited oral absorption of curcumin caused by its poor solubility. In this review, we analyzed studies that investigated free curcumin, curcumin in combination with other agents, curcumin nanoformulations, and curcumin analogs. Among the included studies, 11 investigations evaluated free curcumin (37%), 10 examined curcumin nanoformulations (33%), 7 explored curcumin in combination with other agents (23%), and 2 investigated curcumin analogs (7%). The findings indicate that curcumin exhibits biological activity even in its unmodified form.
Although numerous novel curcumin nanoformulations have demonstrated promising antineoplastic, theranostic and chemo-adjuvant properties, the field of nanotechnology remains relatively new and still faces several limitations. One of the major technical challenges in drug development is controlling the size of curcumin nanoparticles for effective drug delivery applications. In addition, these technologies are often expensive and have not yet been widely implemented. Nevertheless, nanoparticle-based delivery systems have the potential to overcome the limitations of the low bioavailability of curcumin by improving its stability, enhancing cellular uptake and enabling controlled drug release. Therefore, such approaches may offer greater therapeutic promise than the use of free curcumin alone.66
Extensive research has been conducted to develop improved curcumin formulations. Various delivery systems, including liposomes, nanoemulsions, micelles and nanostructured lipid carriers, have been explored to enhance curcumin bioavailability. However, the surfactants and organic solvents used in the preparation of these formulations may introduce potential toxicological concerns, which should be carefully considered in future development.67
The U.S. Food and Drug Administration (FDA) (2013) classified curcumin as generally safe, with no significant adverse effects reported. Some isolated cases of transient allergic dermatitis have been documented.68 Dose-escalation studies have indicated that daily consumption of curcumin at doses up to 12 g does not produce serious toxic effects.69 Furthermore, the clinical investigations involving patients with solid tumors reported no significant adverse events following the oral administration of curcumin at a dose of 900 mg/day for 8 weeks, apart from mild gastrointestinal discomfort.70
A major limitation of the present review is that a meta-analysis could not be performed due to the heterogeneity of the available data.
Recommendations for the future
Although nanotechnology-based formulations have shown promise in overcoming the limited bioavailability of curcumin, these systems often lack adequate cellular specificity. While many innovative curcumin nanoformulations demonstrate notable anti-tumor and chemo-adjuvant properties, several technological challenges remain in their development. One of the primary challenges is the precise control of nanoparticle size to achieve effective targeted drug delivery. In addition, these technologies are costly and have not yet been widely commercialized.
Polymeric nanoparticles and liposomal systems have been shown to improve curcumin bioavailability. However, the potential risk of off-target toxicities has not been sufficiently investigated. Curcumin has been reported to exert cytotoxic effects on cancer cells while displaying cytoprotective effects on normal cells at different dosages and concentrations. These observations should be carefully considered in future preclinical and clinical studies evaluating novel curcumin formulations.
Furthermore, heterogeneity in study designs, formulation types, sample sizes, and routes of administration in human cancer studies makes it difficult to determine which formulation possesses the most favorable pharmaceutical characteristics.66 Many of these promising formulations have yet to undergo extensive clinical trials and require the development of cost-effective manufacturing methods. Therefore, further comprehensive in vivo studies and well-designed clinical trials are necessary to establish their potential therapeutic applications.
Conclusions
The combination therapies incorporating nanoformulations have gained considerable attention as promising drug delivery strategies that may overcome several limitations of the conventional treatment. These systems can enhance intracellular drug concentrations and improve synergistic therapeutic efficacy in cancer management. Novel curcumin formulations have been explored for their potential role in the treatment of OC, with encouraging outcomes.
Curcumin has been shown to exert beneficial effects on OSCC cell lines by reducing cell growth and viability, primarily through the induction of apoptosis via the intrinsic pathway and by causing cell cycle arrest at the G2/M phase. Furthermore, clinical studies have indicated that curcumin may reduce tumor size and exhibit anti-inflammatory and cytotoxic effects at the cellular level.
However, further research is needed to better understand how advanced delivery systems, such as liposomes and polymeric nanoparticles, influence the therapeutic efficacy of curcumin. In addition, comprehensive investigations into the bio-interactions between curcumin nanoparticles and cellular components are required to clarify the mechanisms responsible for their selective cellular uptake and activity.
Ethics approval and consent to participate
Not applicable.
Data availability
The datasets supporting the findings of the current study are available from the corresponding author on reasonable request.
Consent for publication
Not applicable.
Use of AI and AI-assisted technologies
During the preparation of this manuscript, the authors used DeepL Translate for language checking/grammar correction. After its use, the authors thoroughly reviewed, verified and revised all content to ensure accuracy and originality. The authors take full responsibility for the integrity and final content of the published article.