Abstract
Background. Intraoral appliances are widely used in dentistry. Their surface roughness may influence patient comfort, biofilm formation and durability.
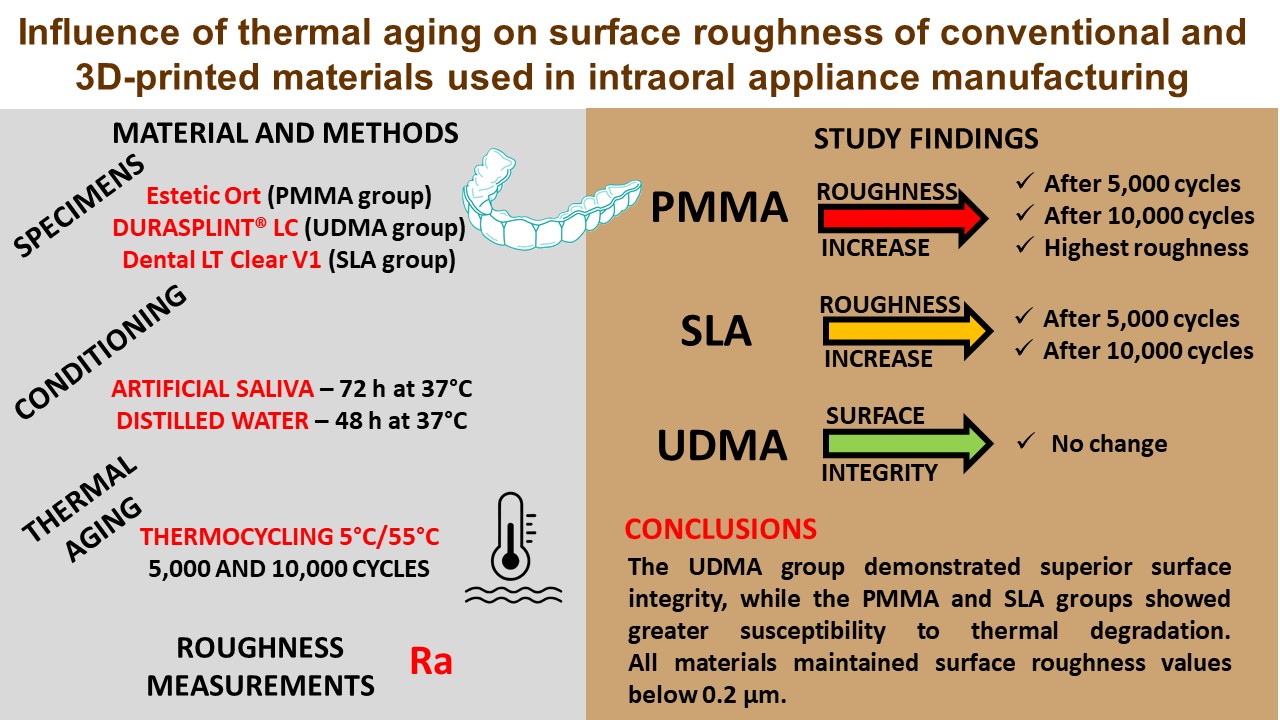
Objectives. The aim of the study was to evaluate and compare the effect of thermocycling on the surface roughness (Ra) of different materials used for the fabrication of intraoral appliances.
Material and methods. Seventy-two standardized specimens (40 mm × 10 mm × 2 mm) were fabricated from 3 materials: a self-curing poly(methyl methacrylate) (PMMA) resin (PMMA group); a light-cured urethane dimethacrylate (UDMA)-based resin (UDMA group); and a stereolithography (SLA) 3D-printed resin (SLA group). Surface roughness was measured before and after thermocycling (5,000 and 10,000 cycles between 5°C and 55°C) using a contact profilometer. Values were reported as mean (M) ±standard deviation (SD). The data was analyzed using repeated-measures or ordinary one-way analysis of variance (ANOVA), followed by Bonferroni-corrected post hoc tests (α = 0.0167).
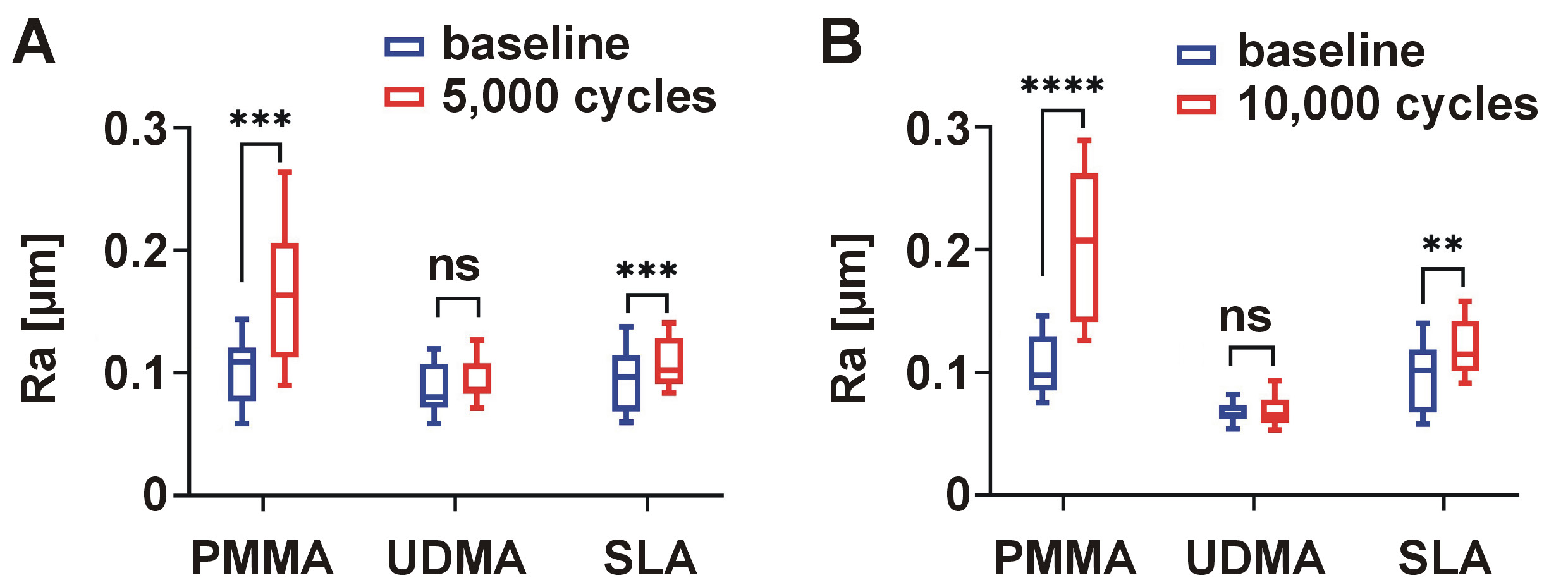
Results. The UDMA group exhibited the lowest mean initial Ra values (0.078 ±0.020 µm). Thermocycling induced changes in surface roughness. In the PMMA group, a significant increase in mean Ra was observed after 5,000 cycles (0.103 ±0.028 µm before vs. 0.167 ±0.059 µm after; p = 0.0001) and after 10,000 cycles (0.107 ±0.024 µm before vs. 0.205 ±0.060 µm after; p < 0.0001). The increase in mean Ra following thermocycling was significantly greater in the PMMA group compared to the other groups (mean ΔRa after 5,000 cycles: 0.064 ±0.035 µm; after 10,000 cycles: 0.098 ±0.046 µm; all p < 0.0001).
Conclusions. Materials used for the fabrication of intraoral appliances exhibit material-specific responses to thermal aging. The light-cured UDMA-based resin demonstrated superior surface integrity after aging, whereas conventional PMMA and the 3D-printed resin were more susceptible to surface alterations.
Keywords: PMMA, surface roughness, UDMA, intraoral appliance, 3D-printed resin
Introduction
Intraoral appliances, such as occlusal splints and mandibular advancement devices, are widely used in dentistry to manage temporomandibular disorders (TMD), protect dental hard tissues, reduce bruxism symptoms, and treat obstructive sleep apnea.1, 2, 3 These intraoral devices create a physical barrier between the dental arches, provide suitable occlusal contacts, redistribute occlusal forces, reduce muscle activity, protect temporomandibular joint structures, and decrease the frequency of apnea episodes.1, 2, 3, 4 Recent epidemiological studies indicate that approx. 8–12% of the adult population requires some form of occlusal therapy, highlighting the clinical relevance of these appliances.5
The efficacy of occlusal splint therapy and patient compliance depend on proper design and adjustment, as well as on the physical and mechanical properties of the materials used for fabrication.6 Traditionally, poly(methyl methacrylate) (PMMA) has been the material of choice due to its favorable mechanical properties, ease of manipulation and cost-effectiveness.7 However, advancements in dental material science have introduced alternative materials, including light-cured urethane dimethacrylate (UDMA) resins and 3D-printable resins, which are increasingly adopted in clinical practice.8, 9, 10
Surface roughness (Ra) is a critical physical property of dental materials that has an influence on biofilm formation, staining potential, wear resistance, and patient comfort.11 Quirynen et al. demonstrated that surfaces with an Ra exceeding 0.2 μm significantly increase bacterial adhesion and biofilm formation.12 Furthermore, Ionescu et al. reported that increased Ra of intraoral appliances may cause tongue irritation, altered taste perception and reduced patient compliance.13 From a longevity perspective, rougher surfaces are more susceptible to wear and chemical degradation, potentially compromising the mechanical integrity and therapeutic efficacy of occlusal splints during clinical service.14
The oral environment exposes dental materials to numerous physical and chemical challenges, including temperature fluctuations, pH variations, masticatory forces, and salivary enzymes.15 Artificial aging protocols, such as thermocycling, are employed to predict the long-term performance of dental materials in laboratory settings.16 Thermocycling simulates thermal changes in the oral cavity through repeated exposure to temperature extremes, typically ranging from 5°C to 55°C.17 This process induces structural stress within materials due to differences in thermal expansion coefficients among their components, which may affect surface characteristics and mechanical properties.18 Although previous studies have extensively investigated the effects of artificial aging on the mechanical properties of occlusal splint materials,19 evidence regarding its influence on Ra remains limited and often contradictory.20
The emergence of digital technologies in dentistry, particularly computer-aided design and computer-aided manufacturing (CAD/CAM) and 3D printing, has been hoped to revolutionize the fabrication process of occlusal splints.21 Additive manufacturing techniques such as stereolithography (SLA) offer advantages including high precision, reproducibility and time efficiency.22 However, the long-term performance of 3D-printed materials in the oral environment, especially regarding surface properties after aging, has not been sufficiently documented.23 Revilla-León et al. highlighted the need for comprehensive investigations evaluating the aging behavior of these novel materials compared with conventional alternatives.24
Furthermore, the effect of thermocycling on Ra may vary significantly among materials used for the fabrication of intraoral appliances. Factors such as polymer composition, cross-linking density, filler content, and manufacturing techniques may influence susceptibility to thermal degradation and subsequent surface alterations.25 Identifying material-specific responses to artificial aging would provide valuable guidance for clinicians in selecting the most appropriate material for individual clinical situations.26
Despite the clinical importance of Ra for the performance and longevity of intraoral appliances, standardized comparative studies evaluating the response of different contemporary materials to artificial aging protocols remain scarce. This knowledge gap is particularly evident for newer 3D-printable resins, which are increasingly incorporated into routine dental practice.23, 26, 27
Therefore, the aim of this study was to evaluate and compare the influence of artificial aging on the Ra of 3 different materials commonly used for intraoral appliance fabrication: a self-curing PMMA resin; a light-cured UDMA-based resin; and an SLA 3D-printed resin. The null hypotheses were that thermocycling would not significantly affect Ra, and that no differences would be observed between the materials in their response to thermal aging. The findings of this investigation may provide evidence-based guidance for material selection in the fabrication of intraoral appliances, potentially improving clinical performance and patient satisfaction.
Material and methods
Materials
This in vitro study evaluated 3 materials commonly used for the fabrication of intraoral appliances. These materials were selected based on their widespread clinical application, distinct chemical compositions and different manufacturing methodologies, as reported in contemporary dental literature.28 Additionally, the selection reflected the authors’ extensive clinical experience with these materials.
The first material tested was a classic self-curing PMMA resin (Estetic Ort; Wiedent, Łódź, Poland) (PMMA group). This material represents the conventional approach to intraoral appliance fabrication and consists of separate powder and liquid components. It served as the control and reference material for intermaterial comparisons.
The second material evaluated was a light-cured UDMA-based resin (DURASPLINT® LC; Scheu Dental, Iserlohn, Germany) (UDMA group). This product is supplied as preformed bars and consists primarily of a UDMA matrix with minor additions of acrylic resin, photoinitiators and cross-linking agents.
The third material investigated was a biocompatible photopolymer specifically designed for SLA 3D printing (Dental LT Clear V1; Vertex Dental, Soesterberg, Netherlands) (SLA group). This product is classified as a Class IIa biocompatible resin composed mainly of methacrylate oligomers, methacrylate monomers and photoinitiators. Dental LT Clear is approved for the fabrication of orthodontic and dental devices, including occlusal splints.
Specimen preparation
A total of 72 standardized rectangular specimens were fabricated (n = 24 per group). The specimen dimensions (40 mm × 10 mm × 2 mm) were selected following the recommendations reported in similar studies.29, 30
The PMMA specimens were prepared using a standardized compression molding technique with metal molds to ensure dimensional accuracy.29, 30, 31 The material was mixed according to the manufacturer’s instructions, with a standardized powder-to-liquid ratio of 3:1 by weight. The mixture was packed into the molds at the dough-like stage, compressed between glass plates under 2.0 bar pressure, and allowed to polymerize for 20 min at room temperature (23 ±1°C), followed by 30 min in a pressure vessel (Polyclav®; Dentaurum, Ispringen, Germany) at 55°C and 2.5 bar pressure.30
To ensure dimensional accuracy, the UDMA-based specimens were prepared using calibrated silicone molds and a compression technique. Polymerization was performed in a laboratory light-curing unit (LC-6 Light Oven; Scheu Dental) for 2 cycles of 10 min each. The specimens were inverted halfway through the process to ensure complete polymerization of both surfaces.32
The SLA specimens were designed using Meshmixer v. 3.5.474 (https://meshmixer.org; Autodesk, San Francisco, USA). Subsequently, the PreForm® software v. 3.28.1 (https://formlabs.com/software/preform; Formlabs Inc., Somerville, USA) was used to generate supports and define printing parameters. The specimens were printed using a 3D printer (Form 2; Formlabs Inc.) at a resolution of 100 μm, with layers oriented at 90° to the building platform. After printing, the specimens were washed twice for 10 min in 99% isopropanol (STANLAB, Lublin, Poland) and air-dried at room temperature for 30 min. Subsequently, they were post-cured in a Form Cure (Formlabs Inc.) at 80°C for 20 min. Finally, the supports were removed by cutting and grinding.32
Following fabrication, all specimens were inspected for defects such as porosities, inclusions or dimensional irregularities. Specimens exhibiting any visible defects were excluded and replaced. To remove processing artifacts, specimen edges were polished with 1,000-grit silicon carbide paper (P.S. Trading, Ołtarzew, Poland). Water sandpaper (grit P500, P1000, P1200; P.S. Trading) and 0.6-mm pumice stone powder (Everall7, Warsaw, Poland) were used to finish the specimens. The upper surface of each specimen was polished for 1 min with a polishing paste for resin and metals (Everall7) using POLIRET MINI (REITEL Feinwerktechnik GmbH, Bad Essen, Germany). The procedure was conducted in accordance with the manufacturer’s instructions and available scientific literature regarding chosen materials.32, 33 All polishing procedures were performed by the same experienced operator to minimize variability. Finally, each specimen was rinsed under water and underwent ultrasonic cleaning (Elmasonic S30H; Elma Schmidbauer GmbH, Singen, Germany) in distilled water with a mild detergent (CLEAN and CLEVER; IGEFA Handelsgesellschaft mbH & Co. KG, Ahrensfelde, Germany) at 30°C for 10 min.
Conditioning
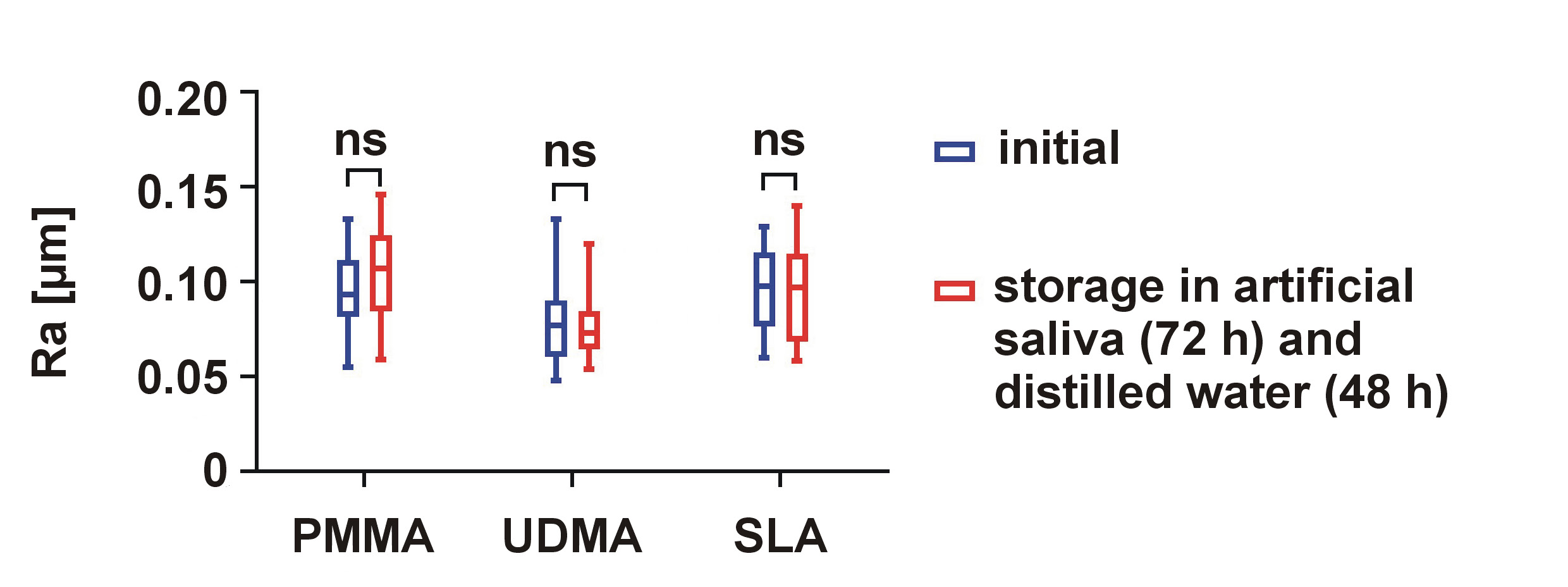
All specimens were stored in artificial saliva (UKD saliva solution; University Pharmacy, Dresden, Germany) for 72 h at 37°C to simulate the intraoral environment and allow water sorption equilibrium.29, 32, 33, 34, 35
Subsequently, the specimens were transferred to distilled water for an additional 48 h at 37°C.36 All specimens underwent ultrasonic cleaning under the conditions described above to remove surface contaminants.37, 38
Thermal aging
Specimens from each group were randomly divided into 2 equal subgroups (n = 12 per subgroup) according to thermocycling exposure (5,000 or 10,000 cycles). Thermal aging was performed using a thermocycler (THE-1200; SD Mechatronik GmbH, Feldkirchen-Westerham, Germany). The specimens were alternately immersed in distilled water at 5°C and 55°C. Each cycle lasted 77 s, with 27 s in each liquid container and 9-s drip time.29, 39 A total of 5,000 and 10,000 cycles were performed to simulate 6 and 12 months of clinical use, respectively.29, 40 However, it should be noted that the cycle-to-time correlation was an estimate for comparative purposes only.
Surface roughness measurements
Surface roughness measurements were performed after specimen preparation (initial), after conditioning (baseline) and after thermal aging (5,000 or 10,000 cycles). Surface roughness was measured using a contact profilometer (Hommel-Etamic W20; JENOPTIK Industrial Metrology Germany GmbH, Villingen-Schwenningen, Germany) with the stylus method in accordance with the VDE standard VDI/VDE 2602 Part 2, 2018-04: Surface Measurement.41 For each specimen, 6 measurements were taken: 3 longitudinal and 3 transverse, positioned perpendicular to each other. The angle of measurement for the diagonal printing path was 45°. The measurements were conducted with a stylus tip radius of 2 µm and a tip angle of 90°. The feed mechanism moved horizontally along the polished surface at a constant speed of 0.5 mm/s and a measuring force of approx. 0.8 mN. The measuring length was 4.8 mm, with a cut-off wavelength of 0.8 mm. Subsequently, the arithmetic mean Ra value was calculated from the 6 measurements for each specimen at each time point (initial, baseline, and 5,000 or 10,000 cycles) within the thermal aging process.29, 30 The change in Ra (ΔRa) was calculated as the difference between baseline Ra and Ra after thermal aging (5,000 or 10,000 cycles).
Statistical analysis
The results were analyzed using the GraphPad Prism 9.1.2. software (GraphPad Software, Boston, USA). Descriptive statistics were presented as box plots showing the five-number summary of data (minimum, first quartile, median, third quartile, and maximum) (n = 12 for each group) and as mean (M) ±standard deviation (SD) in the text. Normality of data distribution was assessed using the Shapiro–Wilk test, which confirmed normal distribution. For within-material comparison, differences in Ra across time points were analyzed using repeated measures one-way analysis of variance (ANOVA) followed by Bonferroni-corrected post hoc tests (α = 0.0167). For between-material comparison, ΔRa values after 5,000 and 10,000 thermocycles were analyzed separately using one-way ANOVA followed by Bonferroni-corrected post hoc tests (α = 0.0167).
Results
The descriptive data for the initial Ra values and Ra after conditioning (baseline) is shown in Figure 1. The UDMA group exhibited the lowest mean initial Ra values (0.078 ±0.020 µm), whereas the SLA group showed the highest mean Ra values (0.096 ±0.020 µm). The coefficient of variation for mean Ra within each material group ranged from 13.314% (UDMA) to 28.992% (SLA). After storing the specimens in artificial saliva and distilled water, no significant changes in Ra were observed for any of the tested materials (all p > 0.05) (Figure 1). The coefficient of variation after conditioning ranged from 15.385% (UDMA) to 23.736% (PMMA). A comparison of ΔRa values after storage in artificial saliva and water revealed no statistically significant differences between the materials (Table 1).
The effects of thermocycling on Ra are presented in Figure 2 and Table 2. An increase in mean Ra was observed for all materials after thermocycling; however, statistically significant changes were detected only in selected groups. After 5,000 thermocycles, significant increases in Ra were observed for PMMA (0.103 ±0.028 µm before and 0.167 ±0.059 µm after 5,000 cycles; p = 0.0001) and SLA (0.095 ±0.026 µm before and 0.110 ±0.021 µm after 5,000 cycles; p = 0.0006). No significant difference was found for UDMA (p > 0.05). The coefficient of variation of the mean Ra ranged from 18.232% (SLA) to 34.735% (PMMA). A comparison of non-aged specimens with specimens subjected to 10,000 thermocycles demonstrated significant differences for PMMA (Ra = 0.107 ±0.024 µm before and Ra = 0.205 ±0.060 µm after 10,000 cycles; p < 0.0001) and SLA (Ra = 0.096 ±0.028 µm before and Ra = 0.121 ±0.022 µm after 10,000 cycles; p = 0.002), whereas no significant changes were detected for UDMA (p > 0.05) (Figure 2). The coefficient of variation ranged from 17.465% (UDMA) to 29.452% (PMMA). Between-material comparison using one-way ANOVA demonstrated statistically significant differences in ΔRa values after thermocycling (p < 0.0001). Post hoc multiple comparisons revealed that the increase in the mean Ra after both 5,000 and 10,000 cycles was significantly higher in the PMMA group than in the UDMA and SLA groups (all p < 0.0001). No significant differences in Ra were observed between the UDMA and SLA groups after 5,000 cycles (p = 0.954) or 10,000 cycles (p = 0.202) (Table 2).
Discussion
The present study investigated the influence of artificial aging on Ra of 3 materials commonly used for the fabrication of intraoral appliances: a self-curing PMMA resin; a light-cured UDMA-based resin; and an SLA 3D-printed resin. The results demonstrated material-specific responses to thermal aging, leading to the rejection of both null hypotheses.
Initial measurements revealed significant differences in Ra among the tested materials, with the light-cured UDMA-based resin exhibiting the lowest Ra values. This finding is consistent with the results reported by Mahross et al., who demonstrated that light-cured UDMA resins typically present smoother surfaces than conventional PMMA, potentially due to differences in manufacturing processes and monomer composition.42 The photopolymerization mechanism in UDMA-based materials may allow for more homogeneous polymerization with fewer internal defects, potentially contributing to improved surface characteristics.43, 44 Furthermore, the preformed bar presentation of light-cured UDMA-based resin eliminates the need for mixing components, reducing the risk of incorporating air bubbles or contaminants that may compromise surface integrity. On the other hand, Weżgowiec et al., in a study comparing the biocompatibility of 3D-printed, thermoformed and heat-cured intraoral appliances, reported no statistically significant differences in Ra between polished self-curing PMMA, light-cured UDMA-based, and SLA 3D-printed resins.33
Storage in artificial saliva followed by distilled water did not significantly influence Ra of tested materials in the present study. However, previous research has demonstrated the hydrophilic nature of PMMA and its relatively high water sorption capacity.45, 46, 47 Conventional PMMA undergoes hydrolytic degradation when exposed to aqueous environments, leading to monomer leaching and disruption of the polymer network.48, 49 Bettencourt et al. reported significant surface alterations in PMMA after storage in artificial saliva, attributing these changes to the combined effect of water sorption and the action of saliva on residual monomers.50 With regard to the SLA 3D-printed resin, the findings reported in this study are consistent with other reports indicating that similar storage conditions did not significantly affect the Ra of Dental LT Clear.51
Thermocycling induced material-specific changes in Ra, with the self-curing PMMA resin exhibiting the most pronounced increase after both 5,000 and 10,000 cycles. This deterioration in surface quality can be attributed to several factors. First, PMMA has a relatively high coefficient of thermal expansion compared to other dental polymers, making it more susceptible to dimensional changes under temperature fluctuations.52, 53 Second, the powder–liquid mixing process in self-curing PMMA may result in heterogeneous polymerization, residual monomer content and porosities, creating weak points vulnerable to thermal stress.54, 55
Similarly to PMMA, the SLA 3D-printed resin demonstrated significant increases in Ra after both 5,000 and 10,000 cycles. The layer-by-layer manufacturing process inherent to SLA technology may create microscopic interfaces between polymerized layers that become weak points during thermal cycling.56 This finding is particularly relevant considering the growing popularity of 3D printing in dental applications. Time-dependent degradation in 3D-printed dental resins can be attributed to polymerization boundaries between layers, which become more evident over time.57 It is worth emphasizing that printing parameters, such as orientation, have been shown to influence roughness and gloss of 3D-printed resins, suggesting that optimization of these parameters may enhance surface integrity.58 Artificial aging has also been reported to affect other mechanical properties of Dental LT Clear, including compressive and tensile strength.59 However, polishing procedures appear to mitigate aging-related damage.59 Moreover, flexural strength and dimensional stability of other 3D-printed resins used in occlusal splint manufacturing have been reported to depend on printing and post-polymerization protocols.60 Collectively, these findings underscore the need for further research to optimize protocols for 3D-printed resins.
In contrast, the light-cured UDMA-based resin demonstrated remarkable stability after thermal aging, with no significant changes in Ra even after 10,000 cycles. This enhanced resistance to thermal degradation can be explained by the high degree of conversion achieved through controlled light polymerization, resulting in a more densely cross-linked and stable polymer network.43, 44 Additionally, the absence of residual monomer, which typically acts as a plasticizer in self-curing resins, contributes to improved thermal stability of UDMA-based materials.61
From a clinical perspective, a Ra threshold of 0.2 μm has been established as critical for bacterial adhesion, with rougher surfaces promoting microbial colonization and biofilm formation.62 In the present study, all materials maintained mean Ra values below this threshold even after simulating 1 year of use (10,000 thermocycles), suggesting acceptable clinical performance regarding microbial resistance. However, the significant increase in roughness observed for self-curing PMMA raises concerns about its long-term performance without additional polishing, particularly in patients with compromised oral hygiene or increased susceptibility to candidiasis.63
The different responses to artificial aging observed among the tested materials have important implications for clinical material selection. Particularly, the significant increase in Ra observed for PMMA and the 3D-printed resin during simulated long-term use suggests that these materials may require more frequent polishing compared with UDMA-based resins. In our previous study, polishing significantly reduced surface roughness parameters (both Ra and roughness depth (Rz)) in conventional PMMA and 3D-printed resins.33 However, clinical decision-making should balance surface characteristics with other relevant material properties, such as mechanical strength and wear resistance.
Several limitations of the present study should be acknowledged. Firstly, although thermocycling is a widely accepted method for simulating thermal aging, it does not fully replicate the complex aging processes of dental materials in the oral environment, including masticatory forces, pH fluctuations and enzymatic activities. Moreover, the cycle-to-time correlation is an estimate for comparative purposes only, not a definitive predictor of clinical longevity. Secondly, Ra reflects only one aspect of material performance. Additional parameters, such as hardness, tensile strength and wear resistance, would allow for a more comprehensive evaluation of aging behavior.59, 60, 64, 65 Furthermore, the study evaluated only 3 commercially available materials, which may limit the generalizability of the findings across broader material categories. Also, the specimen geometry used in this study (flat rectangles) differs from the complex three-dimensional structure of actual occlusal splints, potentially influencing aging patterns.
Despite these limitations and considering the limited available literature as well as the rapidly developing fields of TMD management and dental sleep medicine, where both traditional and digitally manufactured intraoral appliances are increasingly used, the present study provides a significant contribution to the field and a foundation for further research.
Future research should address these limitations by incorporating additional aging parameters. Studies evaluating the combined effect of thermal aging and mechanical stress would be particularly valuable, as occlusal splints are subjected to considerable forces during clinical use. In this context, it would also be interesting to investigate the mechanical surface wear of splint materials, taking into consideration chewing forces, and to determine the abrasion of microplastic particles. Comparative studies in a chewing simulator could provide results regarding microplastic particles that are currently the focus of attention due to their harmful effects on general health. Furthermore, exploring the relationship between surface changes and other clinically relevant outcomes, such as bacterial adhesion, staining susceptibility and patient comfort, would provide more comprehensive guidance for material selection. Despite the broad use and popularity of the materials selected for the purpose of this study, future studies should include a broader range of commercially available materials within each category to better assess in-group differences and improve the representativeness of results.
Long-term clinical trials comparing the performance of different splint materials would be the gold standard for validating laboratory findings and establishing evidence-based recommendations. Additionally, the development and evaluation of novel surface treatments or modifications aimed at improving the aging resistance of conventional PMMA and 3D-printed resins represent promising directions for future research.
Conclusions
The materials investigated for intraoral appliance fabrication exhibited significantly different responses to thermal aging. The light-cured UDMA-based resin demonstrated superior surface integrity, even after 10,000 thermal cycles. At the same time, the conventional self-curing PMMA and SLA 3D-printed resins showed greater susceptibility to thermal degradation, as reflected by significant increases in Ra. Despite these differences, all materials maintained Ra values below the clinically relevant threshold for bacterial adhesion (0.2 μm).
Ethics approval and consent to participate
Not applicable.
Data availability
The additional datasets supporting the findings of the current study are available from the corresponding author on reasonable request.
Consent for publication
Not applicable.
Use of AI and AI-assisted technologies
Not applicable.