Abstract
Background. Oral lichen planus (OLP) is an inflammatory condition of mucous membranes with an immunological basis. To date, no standardized treatment protocol has been established. Another complicating factor in clinical research is the assessment of disease activity and treatment efficacy. Fractal dimension analysis offers an objective method for evaluating mucosal irregularities, making it well suited for assessing OLP lesion morphology and treatment outcomes.
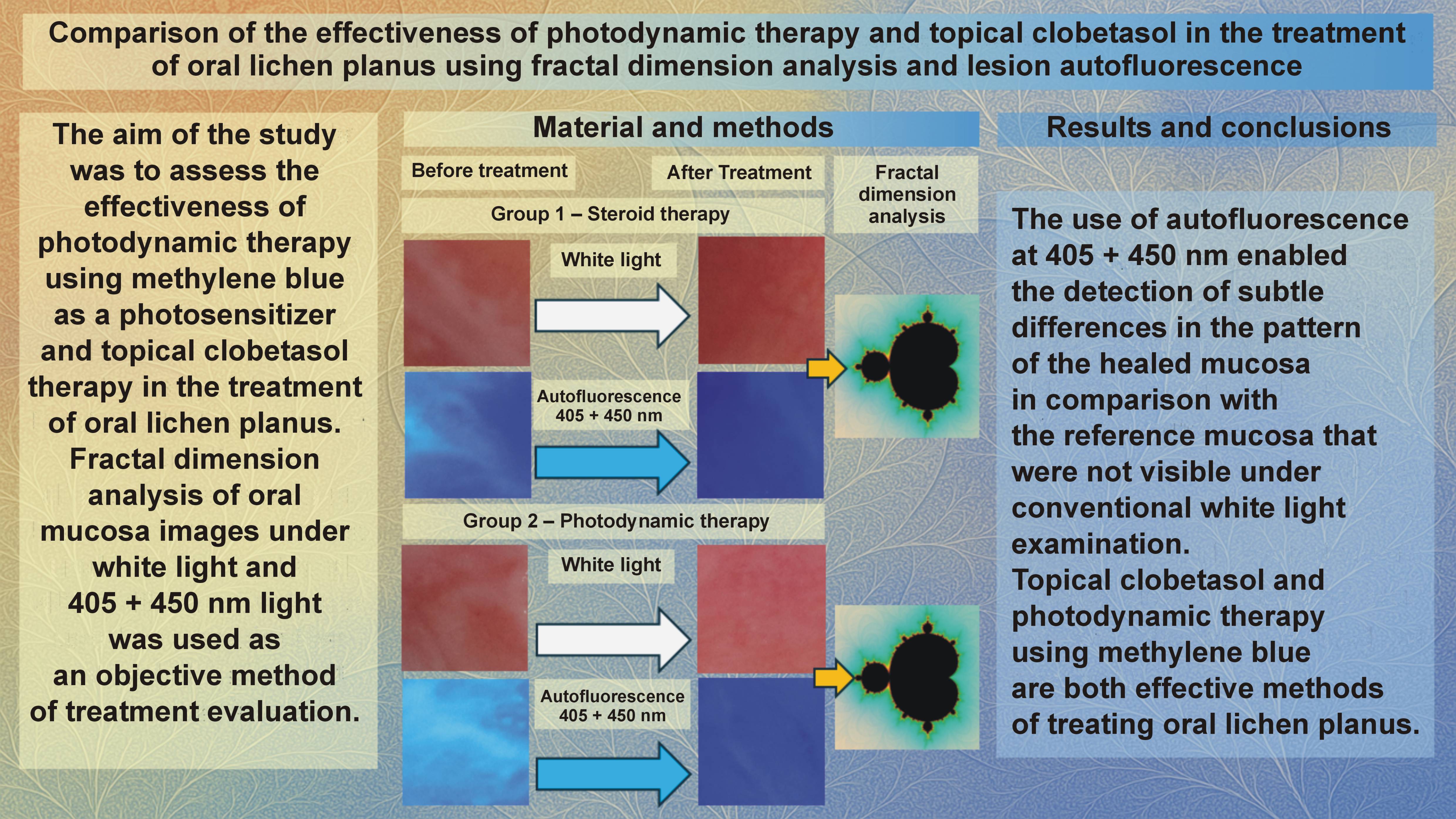
Objectives. The aim of the present study was to assess the effectiveness of photodynamic therapy (PDT) using methylene blue as a photosensitizer and topical clobetasol therapy in the treatment of OLP.
Material and methods. Fractal dimension analysis of oral mucosa images under white light and 405 + 450 nm light was used as an objective method of treatment evaluation.
Results. The use of autofluorescence at 405 + 450 nm enabled the detection of subtle differences in the pattern of the healed mucosa as compared to the reference mucosa, which were not visible during conventional white-light examination. Both treatment methods (topical clobetasol and PDT using methylene blue) demonstrated comparable efficacy in treating OLP.
Conclusions. The use of autofluorescence at 405 + 450 nm is an objective method of assessing healing, more accurate than observations under white light.
Keywords: photodynamic therapy, oral lichen planus, autofluorescence, fractal dimension analysis, clobetasol
Introduction
Oral lichen planus (OLP) is an inflammatory condition of mucous membranes with an immunological basis. The average prevalence of OLP in the global population ranges from 0.89% to 1.01%, varying according to the geographic region, age and gender. It occurs most frequently in Central and South America (1.74%), in individuals over the age of 40, and slightly more often in women.1, 2 The pathomechanism of the disease is not fully understood. The chronic inflammatory response in OLP is triggered by cytotoxic CD8+ and CD4+ T cells and helper T cells, leading to the apoptosis of basal keratinocytes.3 Interestingly, the proportions of Th17 and Th1 T-cell subtypes may vary depending on the clinical form of OLP, indicating complex pathogenesis.4 Histologically, a band-like inflammatory infiltrate in the basal layer of the epithelium is observed, composed predominantly of CD8+ T lymphocytes.5 In addition, the vacuolar degeneration of the basal layer is noted, and the epithelium may appear either acanthotic or atrophic.6, 7 Apart from immune dysregulation, the disease is also believed to involve genetic factors, environmental influences, and infections, particularly with hepatitis C virus (HCV).5, 8, 9, 10 Recently, a vitamin D deficiency has also been implicated in increasing the risk of developing OLP.11, 12
A diagnostic challenge arises from the existence of oral lichenoid lesions (OLLs), including lichenoid contact reactions, drug-induced lichenoid reactions, and the mucosal changes associated with graft-versus-host disease (GvHD). These entities often mimic OLP, both clinically and histologically.13, 14 Nevertheless, bilateral or multifocal distribution remains a key feature supporting the diagnosis of OLP rather than OLL.15
Oral lichen planus most commonly presents as asymptomatic, white, reticular lesions located on buccal mucosa and the lateral borders of the tongue. Less commonly observed variants include atrophic, plaque-type, papular, ulcerative, and bullous forms.16 In these types, patients typically report discomfort, a burning sensation, or pain – either spontaneous or triggered by eating, which negatively impacts quality of life (QoL).17 From a clinical standpoint, a crucial issue is the potential for malignant transformation, particularly in the erosive and atrophic forms of OLP, with reported rates ranging from 0.04% to 1.74%.16, 18, 19, 20, 21, 22, 23 According to some authors, this value may be underestimated due to the lack of standardized diagnostic criteria.24 Esthetically and functionally, the oral cavity is highly sensitive to both treatment and disease progression. Therefore, managing OLP poses a therapeutic challenge for many clinicians, particularly because treatment is symptomatic rather than curative.
To date, no standardized treatment protocol has been established. Reticular lesions usually do not require intervention and are managed through regular monitoring.25, 26 These lesions are typically asymptomatic, and patients may be unaware of their presence. In contrast, intervention is required for other forms of OLP due to the associated pain and the risk of malignant transformation. The primary therapeutic goal is to eliminate inflammation, thereby reducing pain and promoting lesion remission.25 Among the available treatment modalities, topical corticosteroids (TCSs) remain the first-line option. Systemic corticosteroids are reserved for advanced, treatment-resistant cases, particularly those involving mucocutaneous or organ involvement (e.g., the esophagus or genitals). These are often combined with other immunosuppressive or biologic agents.25
The most commonly used TCSs include triamcinolone acetonide, clobetasol propionate, betamethasone valerate, dexamethasone, and fluocinolone acetonide, which are applied in the form of creams, gels, ointments, or submucosal injections.27, 28 Oral corticosteroids include betamethasone, prednisolone and methylprednisolone.29, 30 Due to the side effects of these drugs and their incomplete efficacy, alternative therapeutic agents are continually explored. These include humanized mono-clonal antibodies (efalizumab), chemotherapeutics (dapson), interferons, retinoids (tretinoin, isotretinoin, tazarotene), calcineurin inhibitors (cyclosporine, tacrolimus, pimecrolimus, sirolimus), methotrexate, hyaluronic acid, amitriptyline, and amlexanox.31, 32, 33, 34, 35
Clinicians are increasingly turning to photodynamic therapy (PDT), which has demonstrated efficacy comparable to that of TCSs.36, 37, 38 Photodynamic therapy acts by selectively inducing apoptosis in the proliferating T lymphocytes (CD4+, CD8+, Th17) through the generation of reactive oxygen species (ROS). These ROS are produced when light is absorbed by a photosensitizer that accumulates in hyperproliferative cells. In addition to favorable clinical outcomes, PDT is easy to administer, well tolerated by patients, and exhibits high selectivity at the cellular and molecular levels – advantages not observed with steroid therapy. Furthermore, PDT leads to systemic anti-inflammatory effects, such as reduced plasma levels of CXCL10 chemokine and decreased numbers of circulating CD4+, CD137+, CD8+ cells, and IL-17-secreting T lymphocytes.39 However, some researchers still question the efficacy of PDT in treating OLP.40 From a clinical standpoint, PDT is a promising therapeutic approach. However, differences in light source selection, the photosensitizer type, the application method, and the number of treatment sessions may significantly affect outcomes. Further studies are therefore needed in this area.
What is also a significant challenge in OLP management is effective drug delivery. The moist oral environment hinders the retention and therapeutic activity of topical agents. Polymer-based drug delivery systems that adhere to the mucosa and enable controlled release represent a promising innovation.
Another complicating factor in clinical research is the assessment of disease activity and treatment efficacy. Commonly used scales, such as the numerical rating scale (NRS), Thongprasom, Carrozzo and Gandolfo, or the oral lichen planus disease activity scale (OLP-DAS), are not entirely accurate due to the measurement errors associated with mucosal mobility and subjective assessments by the clinician or patient.41, 42, 43 The size of mucosal lesions is influenced by muscle tension, and the irregularity of the affected areas further complicate measurement. Additionally, image detection and evaluation depend on the light source, specifically, the wavelength of the emitted light, which was demonstrated in our previous research.44 Our evaluation of healing under white light and at 405 + 450 nm combined wavelength reveals differences between these spectral ranges. This approach complements visual assessment and aids clinicians in interpreting treatment outcomes.44
From a geometric perspective, classical Euclidean concepts are insufficient for describing the irregular morphology of OLP lesions. Fractal geometry, which allows dimensional values between 0 and 3, provides a more appropriate framework. Complex and irregular two-dimensional (2D) structures exhibit lower fractal dimension (FD) values, whereas simpler shapes approach a dimension of 2. Figure 1 illustrates the contrast between the FD of a square and the Sierpiński carpet. Fractal analysis therefore offers an objective, quantitative method for evaluating mucosal irregularities, making it well suited for assessing OLP lesion morphology and treatment outcomes.
Objectives
The objective of the present study was to compare the therapeutic effectiveness of PDT with methylene blue and topical clobetasol in the management of OLP. Treatment outcomes were evaluated using an objective assessment method based on fractal dimension analysis of oral mucosa images acquired under white light and 405 + 450 nm autofluorescence. An innovative polymer-matrix drug-delivery system was implemented, designed to reduce mucosal contact time for the photosensitizer while extending the contact duration of the corticosteroid.
The following null hypotheses were formulated:
1. There are no statistically significant differences in the lesion surface area before and after treatment.
2. The mean reduction in the lesion surface area before and after treatment is the same for both therapeutic methods.
3. The mean fractal dimension (FD) of the lesions before treatment does not differ significantly from that of healthy oral mucosa, under both white light and at 405 + 450 nm combined wavelength.
4. There are no statistically significant differences between the FD values of the lesions before and after treatment, under white light and at 405 + 450 nm combined wavelength.
5. There are no statistically significant differences in the FD values of the lesions before and after treatment with regard to the treatment method used.
6. There are statistically significant differences in the FD values of the lesions after treatment as compared to healthy oral mucosa under white light.
7. There are statistically significant differences in the FD values of the lesions after treatment as compared to healthy oral mucosa at 405 + 450 nm combined wavelength.
Material and methods
Design and patients
This project was conducted at the Oral Mucosa and Periodontal Disease Clinic of the Academic Dental Polyclinic at Wroclaw Medical University, Poland. In accordance with the Polish law, it was classified as a therapeutic medical experiment and received prior approval from the Bioethics Committee of Wroclaw Medical University (approval No. kb21/2023n). All participants provided written informed consent before enrollment, in accordance with the Declaration of Helsinki.
Methodologically, the study was designed as a split-mouth randomized controlled trial (RCT) to minimize the influence of interindividual variability on treatment outcomes. The study was retrospectively registered at ClinicalTrials.gov (NCT06752343).
The sample size was determined using McNemar’s test formula, assuming a type I error rate of 5%, a power of 0.8, a success rate of 0.75 in both groups, and a failure rate of 0.25.
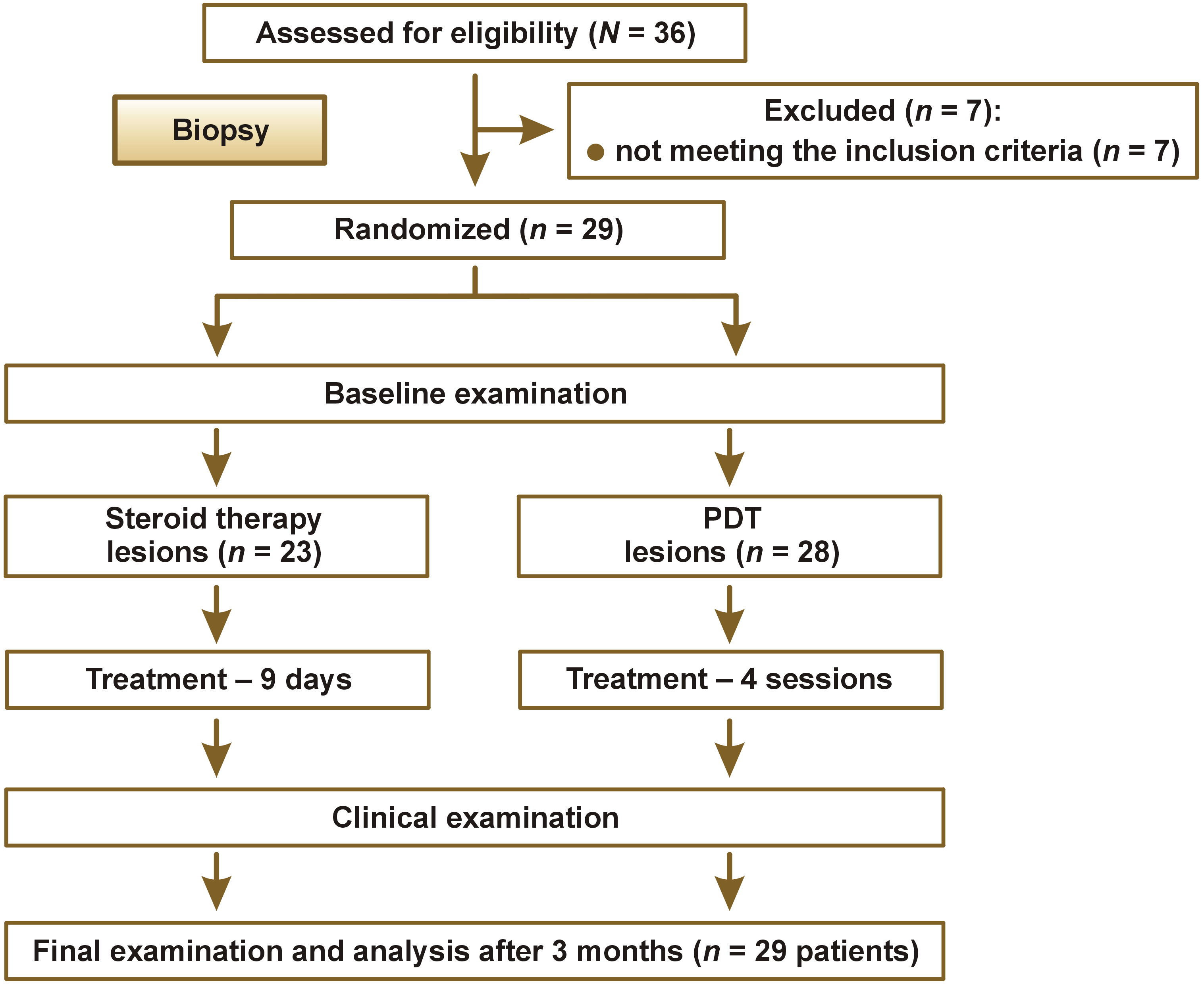
A total of 29 patients participated in the study. Patients with bilateral erythematous or erosive OLP lesions larger than 1 cm in diameter, confirmed histopathologically at the Department of Clinical Pathology of Wroclaw Medical University, were included in the study. The qualification process and assignment of numbers from 1 to 29 were conducted by the same physician (TPK). The main exclusion criterion was dysplasia identified in the histopathological examination. Other exclusion criteria included diabetes, liver diseases, smoking (including e-cigarettes with nicotine), pregnancy, or the objective determination of a lichen planus-like nature of the lesions. The clinical assessment of the evolution of OLP lesions was performed at baseline, at the end of both forms of treatment (day 12) and after 12 weeks without treatment. Patient workflow is presented in Figure 2.
Carriers with clobetasol and methylene blue
The method of preparation and the composition of carriers for the active substances used in the present study were described in the authors’ initial pilot study.45 The matrices were obtained by lyophilizing the foam formed from the dispersion of pullulan, sodium alginate, glycerol, and methylcellulose. The active substances – clobetasol (Pol-Aura, Zabrze, Poland) or methylene blue (abcr, Karlsruhe, Germany) – were added to the homogeneous mixtures (Table 1).
The developed carriers containing clobetasol and methylene blue demonstrated high mechanical strength, smear resistance, and effective release of active substances during in vitro tests. The carrier was precisely fitted to the size of the lesion treated on the mucosa, allowing the accurate determination of the active substance amount in mg/cm2 in the form of a dry carrier.45
Laser device
In our study, we used a 405-nm, 450-nm and 640-nm laser device. This device was invented and manufactured at the Institute of High Pressure Physics, Polish Academy of Sciences, Warsaw, Poland. A reflector in the form of a regular pyramid couples laser diodes into a multi-mode fiber. This device is able to couple into one optic fiber 2 or 3 wavelengths together.
Photodynamic therapy procedure
The treated lesion was entirely covered for 10 min with the carrier containing methylene blue, and then irradiated with a diode laser with a wavelength of 640 nm, at a dose of 120 J/cm2 and a power of 520 mW, a spot diameter of 8 mm, power density of 1.034 W/cm2, and irradiation time 227 s. Photodynamic therapy was repeated in 4 sessions, at 3-day intervals. Each time, the procedure was conducted by the same physician (KJ).
Image acquisition
In previous studies, we demonstrated that the highest level of image detail can be captured from photographs taken using the autofluorescence of the lesions excited with a combined wavelength of 405 + 450 nm.44 All photographs were taken using a Canon EOS 77D camera, with a Canon 60 mm f/2.8 EF-S USM Macro lens (Canon Inc., Ota City, Tokyo, Japan), with Metz 15 MS-1 ring light (Metz, Markham, Canada). The same distance (45 cm) was set to take all photos. The optical axis of the camera was kept perpendicular to the surface of the lesion. The focus plane was locked to 45 cm to obtain repeatability. During laser irradiation, photographs were taken in the same way as under white light. Laser parameters were set to 1.6 W for 405 + 450 nm. The optic fiber was equipped with a special diffuser of light, enabling the uniform irradiation of the surface. To achieve similar histogram filling, we used the following parameters of photographs: 405 + 450 nm – ISO3200, f/9, 1/500 s; white light – ISO 3200, f/9, 1/250s. The regions of interest (ROIs) were 300 × 300 pixels in size. They were selected from both healthy mucosa and the pathologically altered areas, each time from the same anatomical structures. For example, if the pathological lesion was located on buccal mucosa, the reference ROI was taken from the unaffected area of buccal mucosa. The ROIs before and after therapy were selected from the same locations by simultaneously analyzing pre- and post-treatment images.
Fractal dimension analysis
The ImageJ software, v. 1.53e (National Institutes of Health (NIH), Bethesda, USA, https://imagej.net/ij), and the FracLac plugin, v. 2.5 (Charles Sturt University, Bathurst, Australia, public domain license) were used to conduct fractal dimension analysis. The procedure of FD calculation was described in detail in our previous study.44
Statistical analysis
Statistical analysis was performed using Statistica, v. 13.3 (StatSoft, Krakow, Poland), and 0.05 was set as the statistically significant level. The normality of distribution was determined with the Shapiro–Wilk test. The homogeneity of variance was checked using Levene’s test. In case of normal distribution, we performed parametric tests. We applied the paired and unpaired Student’s t test. If the distribution was not normal, we turned to non-parametric tests – the Wilcoxon and Mann–Whitney U tests. The lack of normal distribution was observed for the perimeter (and the associated values, for example the area, etc.) and surface FD.
Results
Table 2 presents the lesion surface area values before and after treatment, categorized by the therapeutic method. Notably, the standard deviation (SD) values for the lesion surface area were high both before and after treatment, as well as for the difference between the 2 time points. In the latter case, the SD values significantly exceeded the mean (M) values. The mean reduction in the lesion surface area was 112.7 ±236.0 mm2 for steroid therapy and 199.1 ±240.2 mm2 for PDT. Null hypothesis 1 was rejected.
No statistically significant differences were observed in terms of reduction in the lesion surface area between the treatment methods (Table 3). Null hypothesis 2 was retained.
Table 4 presents the FD of lesions before treatment in comparison with the reference oral mucosa under white light and at a wavelength of 405 + 450 nm. Statistically significant differences were observed for both white light and autofluorescence induced by 405 + 450 nm light. Null hypothesis 3 was rejected.
Table 5 presents the FD values of the lesions before treatment as compared to their FD after treatment for both therapeutic methods. Statistically significant differences in the image structure of the lesions before and after treatment were observed for both topical steroid application and PDT. Null hypothesis 4 was rejected.
No statistically significant differences in the FD of the lesions before and after treatment were found in relation to the applied treatment method, which highlights their comparable effectiveness (Table 6). Null hypothesis 5 was sustained.
In Table 7, the post-treatment FD values of the lesions are compared with the FD values of the reference (healthy) mucosa. Null hypothesis 6 was rejected. Statistically significant differences were observed between the FD of the lesions after treatment and the FD of the reference mucosa when analyzing the lesion images acquired via autofluorescence. Null hypothesis 7 was sustained.
Discussion
With regard to the local treatment of OLP, the key issue is applying the active substance directly onto oral mucosa. Before the introduction of polymeric matrices, both research outcomes and treatment efficacy assessments were uncertain. The use of medications in the form of ointments, rinses, or sterile gauze pads soaked and applied to lesions introduces a significant confounding factor in clinical trials. Such approaches do not guarantee an adequate drug concentration in the pathological tissue, are burdensome for patients, may result in drug overdosage, and can cause a range of side effects. Consequently, comparing studies that employ different application methods for active substances lacks precision and may bias study results, especially in meta-analyses. Introducing polymeric matrices into clinical use eliminates these confounders and, moreover, enables conducting split-mouth RCTs. The scientific value of these studies is very high, as they eliminate issues related to the commonly used mucosal drug delivery methods and exclude unknown disrupting factors found in parallel-group randomized trials.46 Lesions are treated at similar stages, thereby avoiding period effects. Additionally, fewer patients are needed to achieve comparable treatment outcomes than in parallel-group studies.
We revealed statistically significant differences between white light and autofluorescence induced by 405 + 450 nm light. In both cases, the FD values of the examined lesions were lower as compared to the reference mucosa. By definition of fractal dimension, such results indicate a lower degree of structural organization in the lesion pattern as compared to the healthy mucosa. In the case of white-light examination, the FD values for both the lesions and the control mucosa were higher than those obtained from the images captured using autofluorescence. This leads to the conclusion that the use of autofluorescence in the mentioned wavelength range reveals more details in both the lesion image and healthy oral mucosa. For both white light and induced autofluorescence, the FD of the lesions after treatment was higher than before treatment. This observation is interpreted as an increase in the structural order of the examined lesions. However, no statistically significant differences in the FD of the lesions before and after treatment were found in relation to the applied treatment method, which highlights their comparable effectiveness. In the case of white light analysis, no statistically significant differences were observed between the images of the healed lesions and the reference mucosa. This observation was consistent for both TCS treatment and PDT, showing similar levels of significance. However, interesting results were found when analyzing the lesion images acquired via autofluorescence. In this case, statistically significant differences were observed between the FD of the lesions after treatment and the FD of the reference mucosa. This finding supports the hypothesis of the superiority of autofluorescence over conventional white-light examination. Despite the absence of differences in FD between the treated lesions and healthy mucosa under white light, suggesting full healing, the autofluorescence-based images revealed enough subtle structural characteristics to distinguish them from healthy tissue.
Split-mouth RCTs employing the precise application of active substances onto oral mucosa in treating OLP are extremely rare in the literature. Most typically, they involve the submucosal injection of various active substances, rather than precise mucosal delivery as in our study.47, 48, 49 To our knowledge, only two projects have used adhesive membranes as drug carriers. In one, a biodegradable Rivelin®-CLO membrane was used for local clobetasol delivery; it can adhere to moist oral tissues for an average of 90 min.50 According to the authors, this solution led to a significant reduction in the OLP lesion size, and patients were able to follow the treatment regimen easily. Since the RCTs compared different steroid concentrations with placebo, it is difficult for us to refer to those results.50 In the other project, the properties of an electrospun, bilayered mucoadhesive system were reported.51 It comprises an outer hydrophobic backing layer of polycaprolactone (PCL), and an inner mucoadhesive component made by electrospinning polyvinylpyrrolidone (PVP) and Eudragit® RS100 as fiber-forming polymers. Once loaded with drugs, the system promises enhanced therapeutic efficacy through localized and controlled drug delivery to oral mucosa. The authors suggest this solution is a promising option for treating OLP or recurrent aphthous stomatitis (RAS).51
Most RCTs comparing steroid therapy with PDT use ointments or rinses for active ingredient delivery. This approach is impractical for oral use: saliva washes away the active substance, making it difficult to achieve an adequate drug concentration in the tissue. Due to the rich vascularization of oral mucosa, there is also a risk of uncontrolled systemic absorption and general effects. Using polymeric matrices in our study eliminates this issue, giving us full control over drug delivery. Another factor affecting PDT outcomes is the light source, specifically its power, wavelength and energy density, as well as the irradiation time, and the number of sessions. It is important to remember that light in the visible and near-infrared spectrum penetrates deeper with longer wavelengths: red light penetrates about 0.5 cm at 630 nm and up to 1.5 cm at 700 nm. For OLP, this depth is less significant, as lesions are superficial. However, selecting an appropriate photosensitizer sensitive to a specific wavelength and achieving a sufficient tissue concentration is crucial. Common photosensitizers used in OLP therapy include 5% methylene blue, toluidine blue (TB), methyl-5-aminolevulinate (MAL), and 5-aminolevulinic acid (5-ALA). Considering the many variables in PDT itself, comparing treatment outcomes without standardization is difficult. Nevertheless, systematic reviews unanimously confirm the efficacy of PDT in symptomatic OLP treatment. Additionally, some studies show the superiority of PDT over TCSs in symptom reduction.38, 52, 53, 54 However, the limited number of studies – especially with long follow-up – and the methodological differences in PDT justify the need for further research. A meta-analysis by He et al. indicates higher efficacy of topical ALA vs. methylene blue rinses.53 This difference likely stems from the short rinse time for methylene blue, typically 5 min, as compared to 30–120 min for ALA, which significantly increases the tissue concentration. The authors of the meta-analysis report a PDT recurrence rate of 1.94%, although with follow-up periods varying widely across the studies (3–48 months).53 Thus, long-term PDT outcomes in OLP treatment remain not fully elucidated, underscoring the urgent need for prolonged follow-up. In our case, patients remain in the program and continue follow-up visits for oncologic vigilance. When comparing the effectiveness of TCSs and PDT in our study, we found both methods to be equivalent. The meta-analysis by Hu et al. aligns with our findings.53 Another meta-analysis also reported similar efficacy of PDT and TCSs in symptomatic OLP treatment.55 The analyzed studies used 5-ALA, TB and methylene blue as photosensitizers, and dexamethasone, betamethasone and Kenalog as steroids. Only one study showed TCSs slightly outperforming PDT, though both methods were deemed effective.56 That meta-analysis included different light sources (diode lasers and LED lamps), and showed significant variation in the irradiation time (5–30 min) and device parameters. Despite this heterogeneity, the meta-analysis suggests PDT is effective in OLP treatment.56
The results of our study concur with meta-analyses. Regarding the reduction of the lesion surface area, we found no statistically significant difference between the two methods. We noted high SD values for the surface lesion area, both before and after treatment. Similar conclusions emerged from He et al.’s meta-analysis.53 The FD of the lesions before and after treatment, for both therapeutic options, also showed significant statistical changes, confirming comparable efficacy (with no statistical difference). Likewise, the FD values captured in white light as well as at 405 + 450 nm before and after treatment support the abovementioned similarity. Notably, in white light, the post-treatment FD values and those for the reference mucosa were higher than those obtained at 405 + 450 nm. Thus, the 405 + 450 nm light reveals more detail, greatly enhancing treatment outcome assessment. Consequently, visual evaluation via autofluorescence is superior to white-light imaging.
Autofluorescence enables the analysis of biochemical changes in pathological cells via fluorophores like tryptophan, collagen and porphyrin, which emit fluorescence under appropriate excitation.57 Autofluorescence is used to detect oral cancer, particularly early-stage, and potentially malignant lesions. Light-based detection systems exploit the metabolic changes in pathological cells affecting absorption or fluorescence.58 In autofluorescence, the diseased tissues lose fluorescence due to metabolic disruption and altered fluorophore distribution, appearing darker. The commercial VELscope™ system achieves sensitivity for detecting malignant or potentially malignant lesions at 22–100%, and specificity at 16–100%.58 However, the literature lacks strong evidence to endorse it as a screening tool for oral cancer.59 To date, no specific spectral signature has been presented for OLP. A few studies suggest that 320 nm light is optimal for assessing the disrupted collagen variability in OLP.60, 61 This data and our findings suggest that developing OLP diagnostics using autofluorescence is worthwhile, and may be useful in monitoring treatment progression.
One cannot ignore parallel-group RCTs comparing PDT and TCSs, which are far more common than split-mouth RCTs. Yet, to our knowledge, no trials compare clobetasol with PDT in OLP treatment. Additionally, active substance applications usually involve rinsing with a photosensitizer solution or pipetting it on, while steroids are often applied via ointments or rinses – methods diametrically opposite to ours. However, the absence of studies like ours necessitates comparison to other designs. Mostafa et al. compared Kenalog with PDT using methylene blue.37 The steroid was applied as a thin layer of ointment (Kenacort-A Orabase) thrice daily for 30 min, while 5% methylene blue rinse was followed by 660 nm diode laser irradiation (100–130 mW/cm2) for 70 s, repeated weekly for 2 months. The results indicated PDT was a better option than topical steroids, with greater pain reduction. Yet, their study had significantly more female subjects and a younger average age than ours.37 Jajarm et al. compared PDT with TB and dexamethasone in OLP treatment.56 They applied a TB solution (1 mg/mL) by pipette, irradiated after 10 min with a GaAlAs 630 nm laser (10 mW/cm2 continuous wave, fluence 1.5 J/cm2), twice weekly for a month. A steroid rinse (5 mg/5 mL) was administered for 5 min, 4 times daily for a month. The authors reported that both methods were effective, though the mean pain improvement and treatment efficacy favored steroids.56 In our approach, we used similar wavelengths, but significantly higher power and fluence, with fewer sessions, making comparisons difficult due to methodological differences. Bakhtiari et al. compared PDT with methylene blue and dexamethasone.62 They used a 630 nm LED lamp with 7.2–14.4 J/cm2 fluence, irradiating for 30–120 s in sessions on days 1, 4, 7, and 14. Patients rinsed their mouths with 5% methylene blue for 10 min; a steroid rinse (0.5 mg/5 mL) was used for 2 min, 4 times daily for 2 weeks. They found no significant difference between the methods regarding efficacy. Beyond the different PDT protocol, they included erosive and reticular OLP, which is significantly methodologically different from our study.62 Mirza et al. conducted a parallel-group RCT comparing PDT, low-level laser therapy (LLLT) and topical steroid therapy.36 The PDT protocol involved TB (1 mg/mL) pipetted, and irradiated after 10 min with a GaAlAs 630 nm laser at 10 W/cm2 continuous wave, twice weekly for a month. A dexamethasone rinse (5 mg/5 mL) was applied for 5 min, 4 times daily for a month. Pain reduction was higher for TCS, yet treatment efficacy was statistically greater for PDT.36 All the 4 studies featured divergent methodologies as compared to ours. Notably, frequent irradiation sessions and self-administered steroid use are burdensome for patients, and increase the risk of complications and missed doses, which may significantly bias results.
A database analysis clearly indicates that PDT is an effective alternative to topical steroid therapy. Given the heterogeneity of studies, it is impossible to objectively compare those described in the literature, especially considering parameter variability, like wavelength, and energy density, and drug application methods. Our proposed treatment protocol, using patented drug carriers, is more precise and paves the way for standardizing OLP therapy. Moreover, evaluating lesions under 405 + 450 nm light enhances the sensitivity of OLP lesion detection and facilitates treatment monitoring. Changes in the FD of the treated mucosa compared to the reference mucosa in the 405 + 450 nm spectrum suggest the presence of subtle post-treatment differences, not detectable under standard white-light examination.
Finally, it is worth mentioning non-pharmacological treatment approaches that utilize laser devices. One is LLLT, which shows long-term analgesic effects (>12 weeks). Interestingly, this effect is not observed in the short term (<12 weeks).63 Additionally, LLLT reduces pathological lesions, which is confirmed by biopsy. Conversely, another meta-analysis reports limited scientific evidence for the superiority of LLLT over corticosteroids.62 Nevertheless, LLLT represents a promising OLP treatment option, especially where steroid side effects are unacceptable. The range of laser devices is broad, and clinical trials also employ high-energy lasers emitting various wavelengths. Devices used in OLP treatment include Er:YAG, Nd:YAG, CO2, Er,Cr:YSGG, Nd:YAG, and diode lasers (630–808 nm).64, 65 Research outcomes for these devices show short-term efficacy, but due to the limited number of studies and differences in the irradiation parameter, caution is advised when interpreting such results.
Study limitations
Capturing reproducible intraoral photographs without surface light reflections – even on dried mucosa – remains a significant challenge. An additional limitation is the difficulty in achieving the uniform illumination of the examined area due to the complex anatomical structure of the oral cavity. The study is further limited by the relatively small number of patients evaluated and the short follow-up period after the completion of active treatment. Each case of red OLP requires careful, long-term clinical monitoring because of its potential risk of malignant transformation.
Conclusions
Both treatment methods demonstrated comparable efficacy in managing OLP lesions. Fractal dimension analysis enabled clear differentiation between the treated lesions and healthy mucosa, revealing statistically significant differences in the FD values before and after treatment under both white light and 405 + 450 nm illumination. Additionally, autofluorescence imaging at 405 + 450 nm detected subtle structural differences between the healed and reference mucosa that were not visible under conventional white-light examination, with statistically significant post-treatment differences in the FD values observed under this wavelength.
Trial registration
The study was retrospectively registered at ClinicalTrials.gov (NCT06752343).
Ethics approval and consent to participate
The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the Bioethics Committee at Wroclaw Medical University, Poland (approval No. kb21/2023n). Written informed consent was obtained from all subjects involved in the study.
Data availability
The datasets supporting the findings of the current study are available from the corresponding author on reasonable request.
Consent for publication
Not applicable.
Use of AI and AI-assisted technologies
Artificial intelligence (AI) was used for improving the English language and for preparing the highlights (ChatGPT).