Abstract
Background. Considering the complications associated with autogenous bone grafting, the use of freeze-dried bone allograft (FDBA) granules may be considered as an alternative treatment plan.
Objectives. The aim of this study was to evaluate the effect of metformin on both the proliferation and osteogenic capability of dental pulp stem cells (DPSCs) cultured on FDBA granules.
Material and methods. First, a pilot study was conducted only on DPSCs to confirm cellular viability and the osteoinducing effect of 100 μmol/L metformin. Next, the cells were loaded on FDBA granules and treated with and without metformin. Finally, the following analyses were performed: scanning electron microscopy (SEM) (cell attachment); the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay (proliferation); and alkaline phosphatase (ALP) activity analysis (osteogenic differentiation).
Results. The SEM images revealed that metformin enhanced the adhesion of DPSCs on FDBA granules. In addition, metformin was shown to increase cell proliferation/viability from day 1 to day 7. Compared to the control, a significant difference was observed after 7 days of treatment. Metformin enhanced the osteogenic capability of FDBA in both standard and osteoinducing conditions. An increase in ALP activity was significant after 7 days of treatment. The positive effect of metformin on differentiation was significant in osteoinducing conditions.
Conclusions. Metformin can be applied as an additional osteoinductive factor in bone regeneration treatment. Moreover, scaffolds with controlled release of metformin can be considered a proper osteoinductive bone substitute that may lessen the complications related to applying allograft scaffolds alone.
Keywords: allograft, dental pulp stem cells, tissue engineering, bone regeneration, metformin
Introduction
Different types of bone materials have been used to treat major orthopedic and maxillofacial bone defects.1, 2, 3 Although their use produces fewer side effects (e.g., less morbidity, nerve damage, bleeding, infection, and loss of function) in comparison with autogenous bone grafts,4, 5 they lack essential osteoinductive factors to be utilized as bone-grafting progenitor cells.6 However, freeze-dried bone allografts (FDBAs) possess osteoinductive growth factors, such as bone morphogenic proteins.4, 6 Additionally, they have demonstrated the capability to recruit bone-forming progenitor cells and support them following differentiation.6 Despite this capability, they still have less osteogenic capacity than autogenous bone grafts, which leads to slower new bone formation in the defective site.7, 8 Hence, attempts have been made to find a solution to improve the osteogenic ability of FDBA granules.9, 10, 11 Unfortunately, they were not effective in vivo.12, 13, 14, 15
It has been proven that metformin, an insulin-inducing agent, can increase mineralization and osteogenic differentiation. In fact, metformin can activate 5’ adenosine monophosphate (AMP)-activated protein kinase (AMPK), which regulates osteoblast differentiation in specific tissues.16 Several studies have evaluated the effect of metformin, and described its positive effect on the proliferation and osteogenic differentiation of stem cells in vitro.17, 18, 19, 20 The enhancing effect of metformin on new bone formation has also been analyzed in in vivo conditions.21 It has been shown that the application of metformin helps to prevent the development of periodontitis through its osteoinductive features and its ability to regulate blood glucose.22
In this study, in order to enhance the osteogenic potential of FDBA materials, metformin, an osteogenic factor, was added to the medium. Human dental pulp stem cells (hDPSCs), a suitable cell source for regenerative treatment,23, 24, 25 were used. This study evaluated the effect of metformin on the proliferation, osteogenic differentiation and adhesion of DPSCs cultured on FDBA granules.
Material and methods
Ethical permission was obtained from the Ethics Committee of Shahid Beheshti Medical University in Tehran, Iran (Code: IR.SBMU.DRC.REC.1395.324).
Isolation and characterization
of human DPSCs
Dental pulp stem cells at passage 3 were purchased from the Dental Research Center of Shahid Beheshti University of Medical Sciences. A high expression of mesenchymal stem cell markers (CD44, CD90, CD73, and CD105; Abcam, Cambridge, UK) and a low expression of hematopoietic cell markers (CD34 and CD45; Abcam) were characterized through flow cytometry analyses. The multilinear differentiation capacity of the cells was investigated through osteogenic and adipogenic differentiation tests. The cells were cultivated in standard Dulbecco’s Modified Eagle’s Medium-high glucose (DMEM-HG) with 15% fetal bovine serum (FBS) (Invitrogen, Carlsbad, USA) and 1% penicillin/streptomycin 10,000 U/mL (Life Technologies, Carlsbad, USA) at 37°C in a humidified 5% carbon dioxide environment. When the cultures reached 90% confluence, the cells were detached using 0.25% trypsin plus ethylene diamine tetraacetic acid (EDTA) (Life Technologies), and passaged for the following experiments.
Pilot study
Considering the dose-dependent effect of metformin, 100 µmol/L metformin hydrochloride (Alborz Pharmaceutical Co., Tehran, Iran) was chosen based on a previous study.19 The positive effect of this concentration of metformin on the proliferation and differentiation of DPSCs was evaluated. In brief, the cells at a density of 104 were seeded in 48-well plates with 100 μmol/L metformin hydrochloride, and either the standard medium was added for the proliferation assay or the osteogenic medium for the differentiation assay. Proliferation was evaluated with the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) (Sigma Aldrich, St. Louis, USA) assay after 1, 3 and 7 days of treatment with or without metformin. Osteogenic differentiation was evaluated using an alkaline phosphatase (ALP) activity kit (Sigma Aldrich) after 3, 7 and 14 days. Similar to the proliferation assay, DPSCs cultured in the osteogenic medium only (DMEM-low glucose (LG), 0.2 mol/L ascorbic acid 2-phosphate, 10−8 mol/L dexamethasone, and 10 mmol/L β-glycerol phosphate; Life Technologies), without metformin, were used as a control for pairwise comparisons at each time point.
Cell seeding
Particulate cortical/cancellous mineralized 1–2-millimeter FDBA granules (Tissue Regeneration Corporation, Tehran, Iran) were placed in 48-well plates and a cell suspension including 2.5 × 104 DPSCs was loaded into the wells. The plates were incubated at 37°C for 1 h. Then, according to the type of evaluation, 2 kinds of media (standard or osteogenic) were added to the wells. The osteogenic medium included DMEM-LG, 0.2 mol/L ascorbic acid 2-phosphate, 10−8 mol/L dexamethasone, and 10 mmol/L β glycerol phosphate (Life Technologies). Next, 100 μmol/L metformin hydrochloride was added to the samples. The study groups and the content of the wells are summarized in Table 1.
Cell adhesion
The adhesion of DPSCs was evaluated with a scanning electron microscope (SEM) (VEGA; Tescan, Brno, Czech Republic) at ×1,000 and ×2,000 magnifications 1 and 7 days after cell seeding. After being rinsed with phosphate-buffered sodium (PBS) (Sigma Aldrich), the cells were fixed for 2 h in 2.5% glutaraldehyde and for 1 h in 1% osmium. The samples were then dehydrated with ascending grades (30%, 50%, 70%, 90%, 95%, and 100%) of ethanol and dried in a desiccator for 24 h. Finally, they were covered with gold and analyzed using SEM imaging.
Cell proliferation
In order to evaluate cell viability, the MTT assay was performed 1, 3 and 7 days after seeding. First, the granules were transferred to new wells to avoid counting the cells adhering to the bottom of the plate. Then, MTT was added to the wells and the plates were incubated at 37°C for 4 h. Next, they were incubated with dimethyl sulfoxide (Carlo Erba Reagents, Milan, Italy) overnight. The supernatants were collected and transferred to 96-well plates, and absorbance was read using a microplate reader (Anthos Labtec Instruments, Salzburg, Austria) at a wavelength of 590 nm.
Cell differentiation
The osteogenic differentiation of the stem cells was evaluated 3, 7 and 14 days after cell seeding using an ALP activity kit. This test was performed on the cells seeded in both the non-inducing and inducing media. The cell-scaffold constructs were rinsed twice with PBS, homogenized in lysis buffer (10 mmol/L Tris-HCl – pH 7.5, 1 mmol/L MgCl2 and 0.05% Triton X-100) and centrifuged at 12,000 rpm for 10 min at 4°C. The cell lysate was mixed and incubated with the ALP assay reagent at 37°C for 30 min, and the absorbance of the resulting ALP activity was quantified by calculating the optical density values at 405 nm. It must be mentioned that the reaction was stopped using 0.5 N sodium hydroxide.
Statistical analysis
The Mann–Whitney U test was used to compare the MTT values. The Kruskal–Wallis test was performed for the multiple group comparison. Data was analyzed using the PASW Statistics for Windows computer software, v. 18.0 (SPSS Inc., Chicago, USA), at a significance level of 0.05.
Results
Pilot study
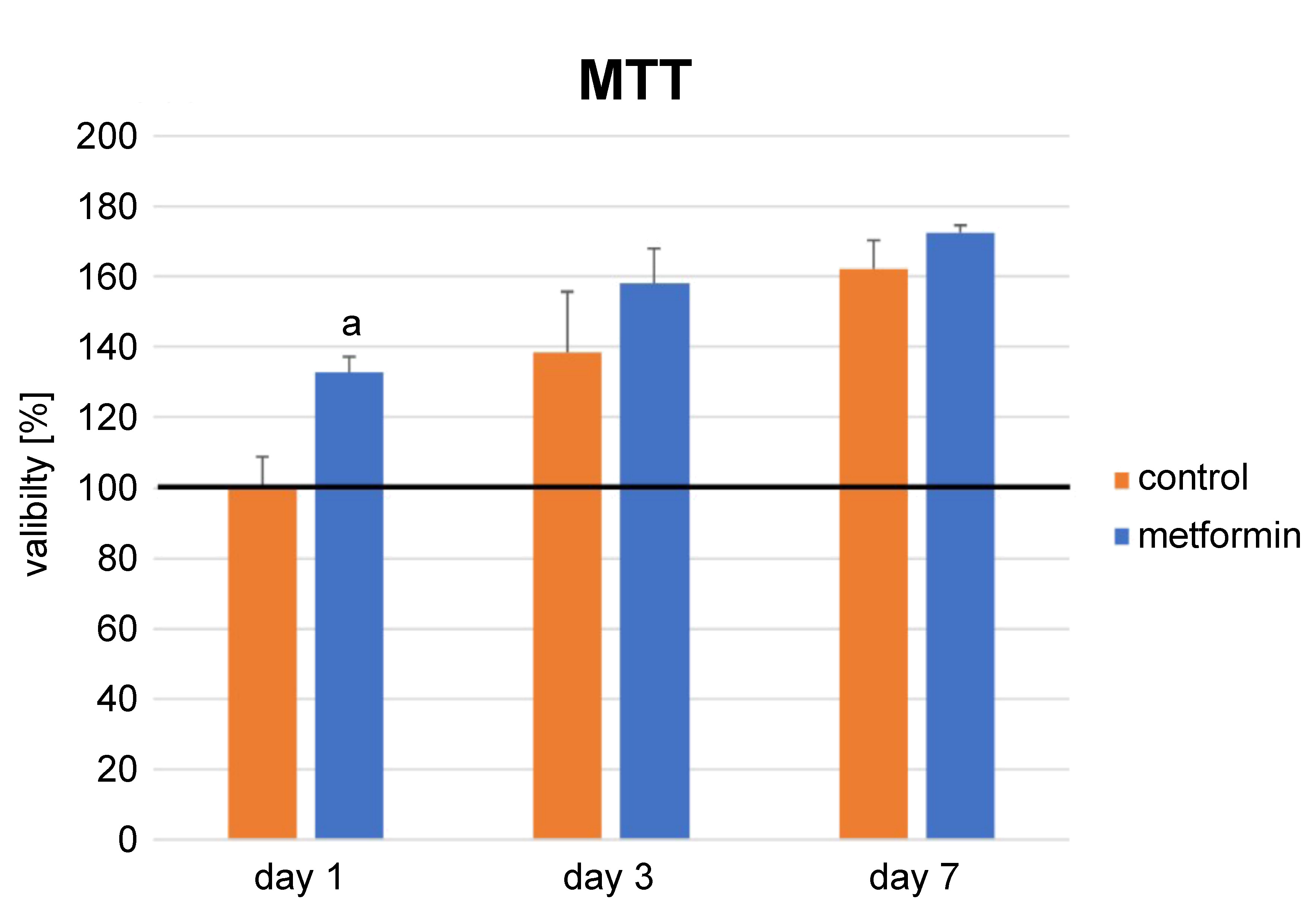
The positive effect of metformin on the proliferation and differentiation of DPSCs was evaluated in this phase of the study. The proliferation of DPSCs in the metformin group was higher than that in the control group at each time point, but the difference was significant only at day 1 (p = 0.00) (Figure 1). An increase in the number of cells from day 1 to day 7 demonstrated the biocompatibility of this concentration of metformin (p = 0.00). In addition, just as with proliferation, the ALP activity of the metformin group was higher as compared to the control group in the osteogenic medium (p = 0.00), but the difference between the groups was not significant in the standard medium (Figure 2). Moreover, it was shown that the osteogenic medium increased the differentiation of both the control and metformin groups (p = 0.00) (Figure 2).
Cell adhesion
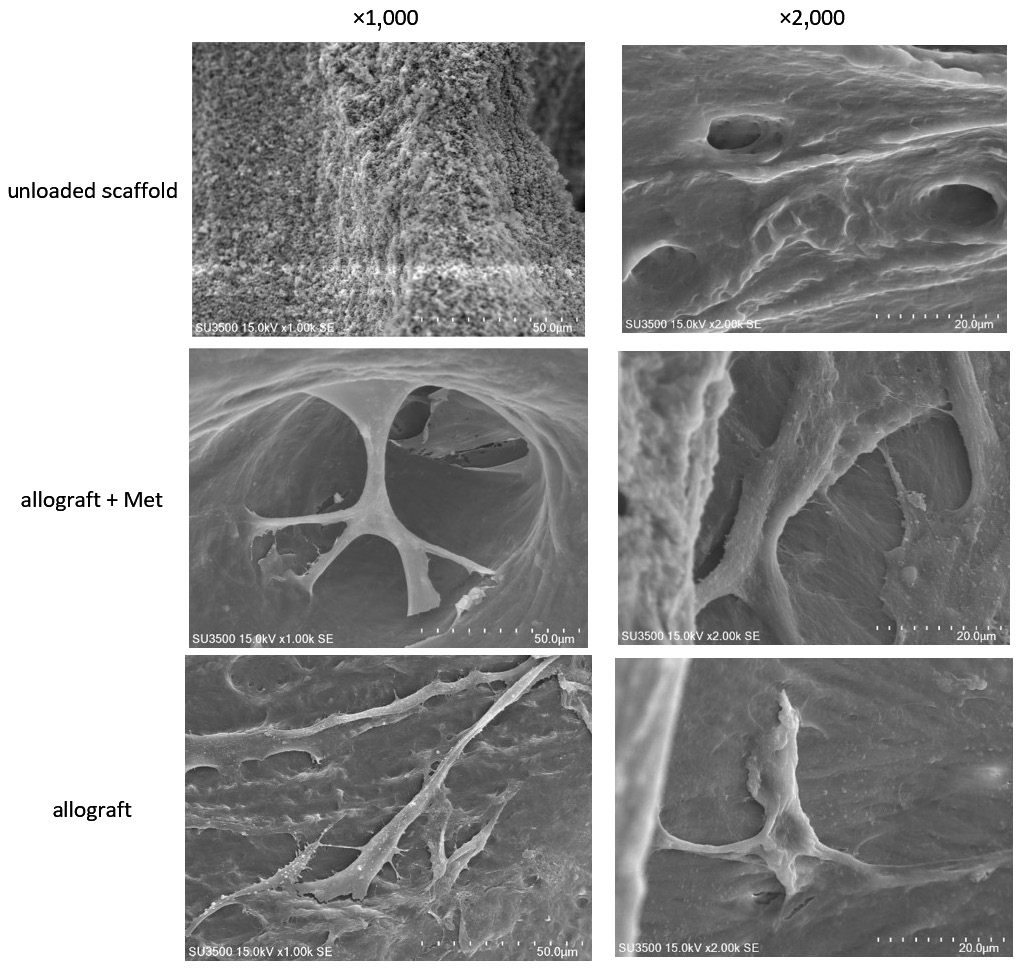
The micro-images of the unloaded scaffolds are depicted in Figure 3. Freeze-dried bone allograft granules had a lamellar texture and a pore size of 50–500 µm. As can be observed in Figure 3, SEM confirmed the strong attachment of DPSCs to FDBA granules in the presence of metformin.
Cell proliferation
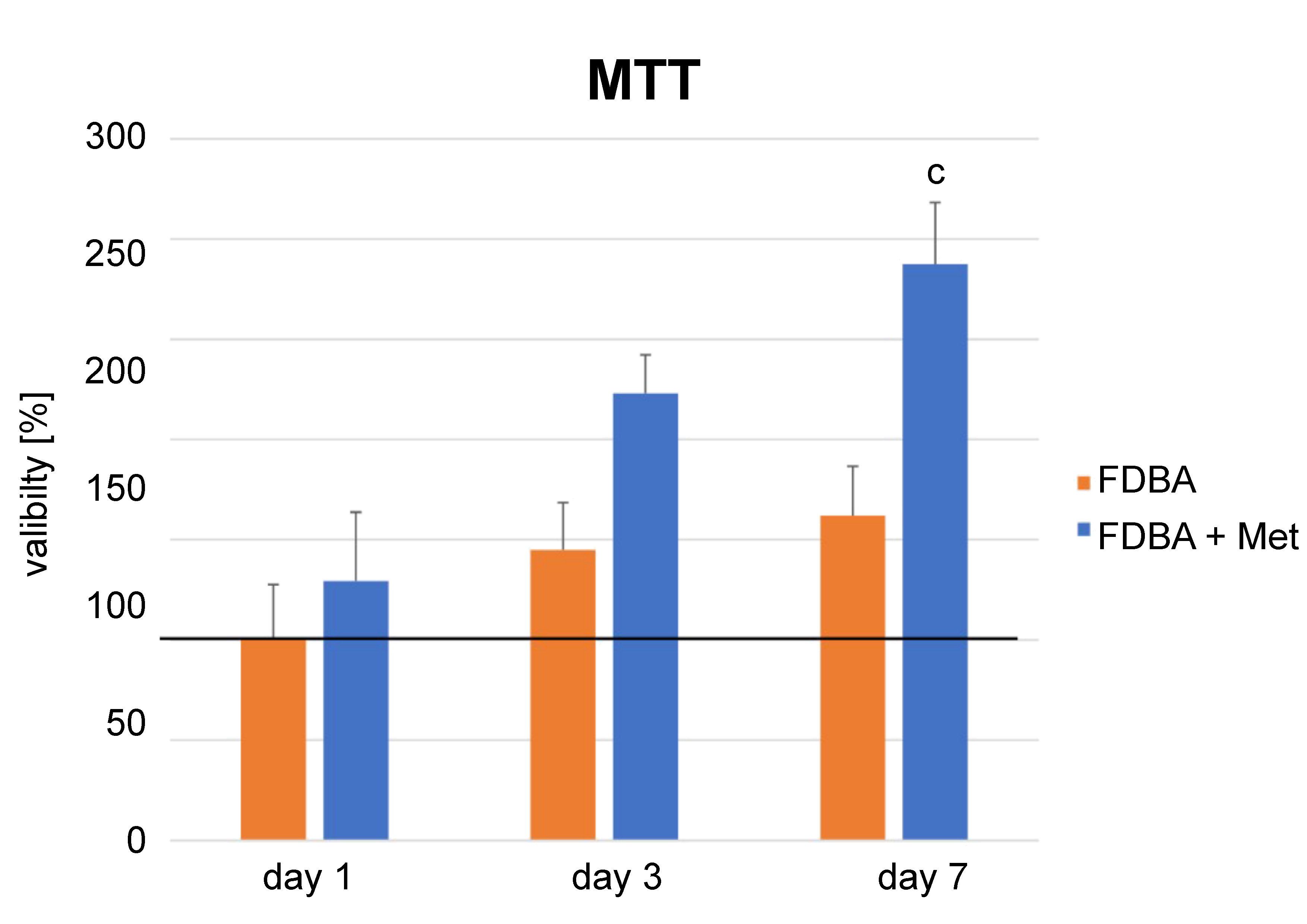
Cell viability increased from day 1 to day 7 in all groups (p = 0.00) (Figure 4). The FDBA + metformin (Met) group showed higher proliferation at each time point. However, the difference was significant only at day 7 following cell seeding (p = 0.004).
Cell differentiation
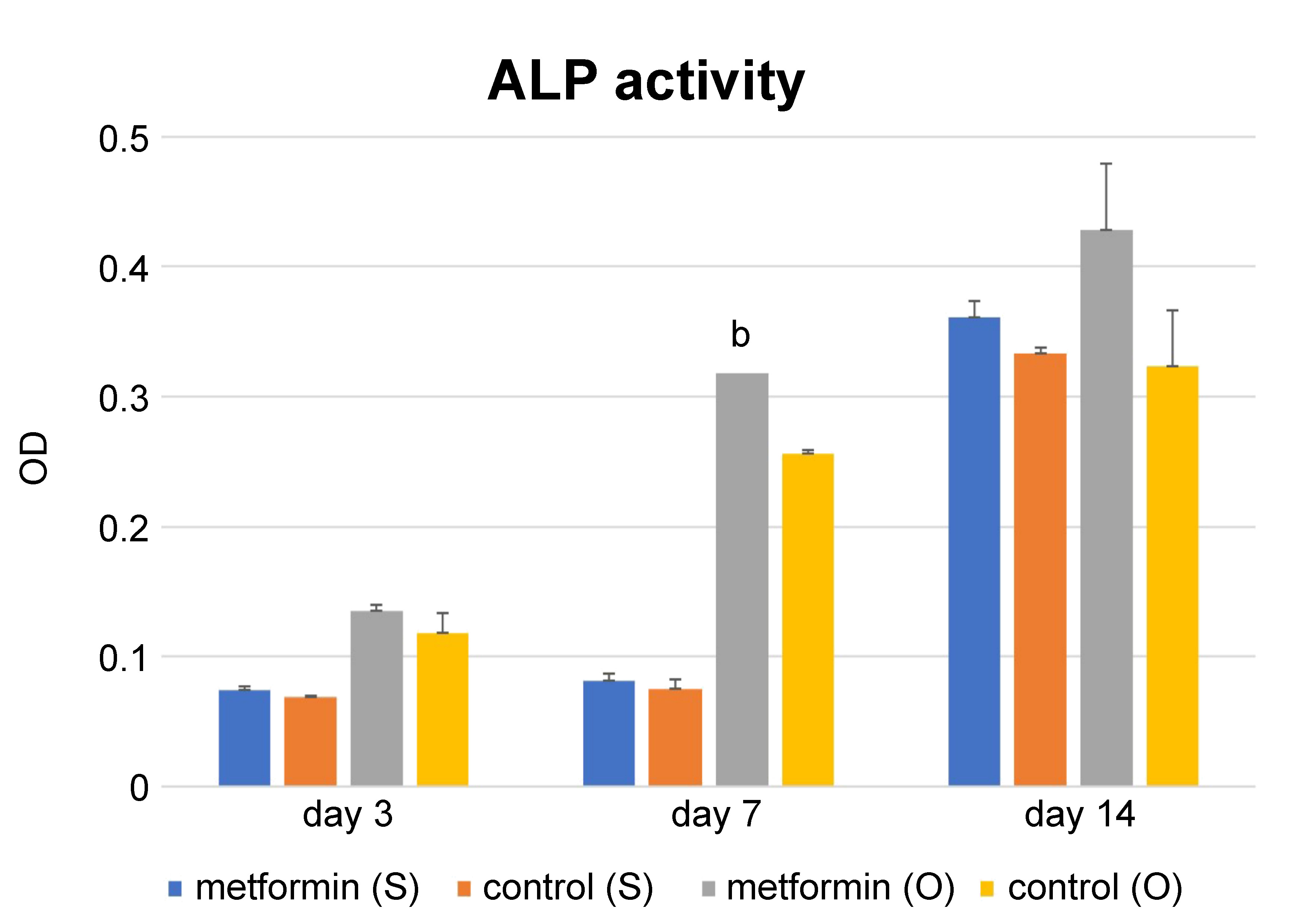
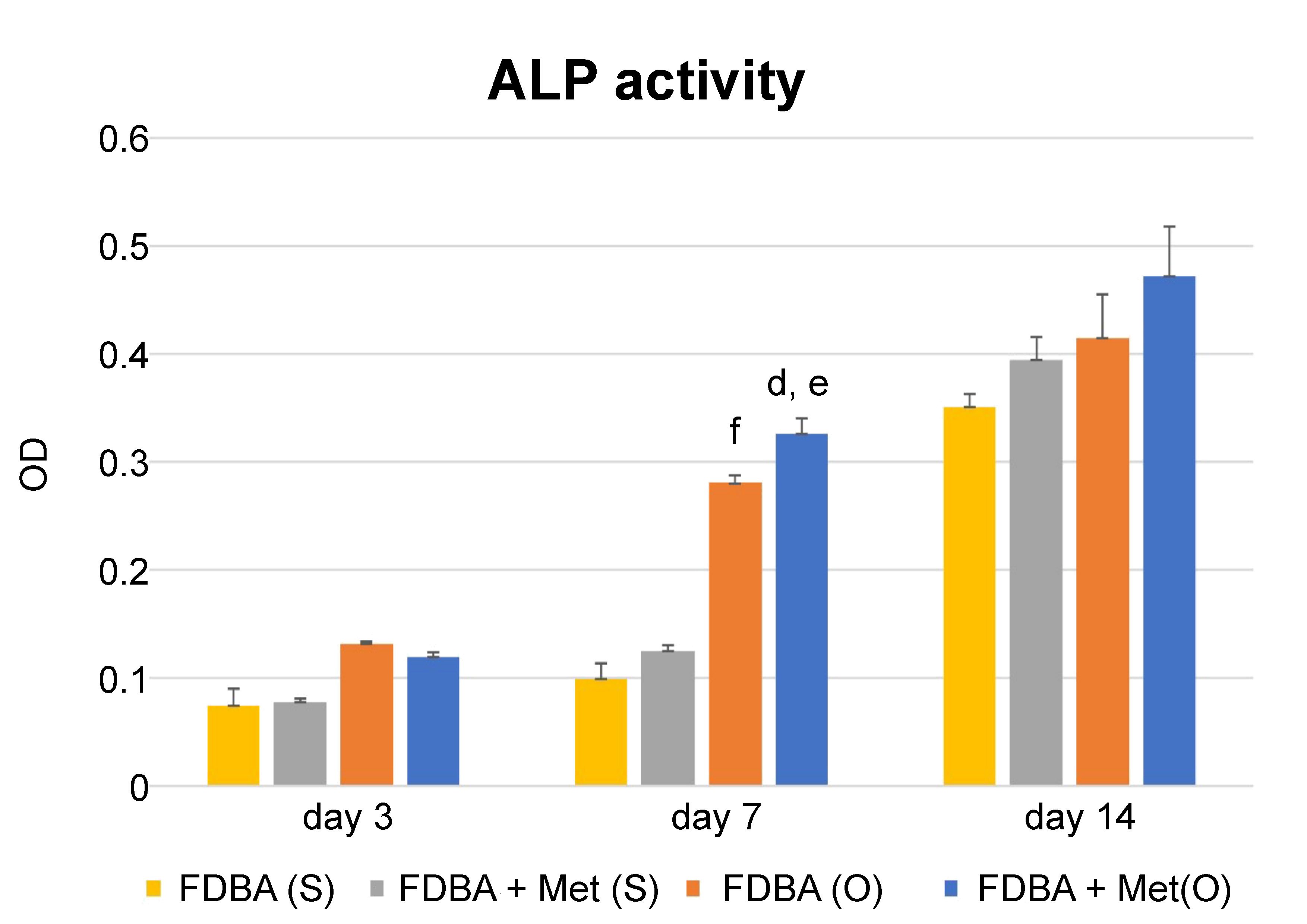
Alkaline phosphatase activity increased in all groups 7 days after cell seeding, in both the inducing and non-inducing media. After 3 days, the FDBA + Met group showed a lower ALP activity in the osteogenic medium, but the difference was not statistically significant. However, after 7 days, the FDBA + Met group showed a significantly higher ALP activity in osteogenic conditions only (p = 0.01). Using the osteogenic medium increased ALP activity significantly in both groups (p = 0.00), but 14 days following cell seeding, the difference was not significant. In addition, 14 days following cell seeding, there was no significant difference in the ALP activity rate between the FDBA and FDBA + Met groups (Figure 5). Thus, it was shown that the positive effect of metformin on differentiation was significant in osteoinducing conditions.
Discussion
The success of regeneration treatment depends on providing a suitable environment for the attachment, proliferation and differentiation of stem cells. Proper interactions between stem cells, growth factors and scaffolds are essential to reach this goal. Freeze-dried bone allograft granules have less osteogenic capacity in comparison with autogenous bone grafts. Thus, several studies have been conducted to enhance the osteogenic ability of these granules. The aim of this study was to evaluate the effect of metformin on the adhesion, proliferation and differentiation of DPSCs cultured on FDBA granules. The results showed that metformin could enhance the adhesion, proliferation and differentiation of DPSCs on FDBA granules in inducing conditions.
Houshmand et al. evaluated the effect of metformin on the proliferation, differentiation and adhesion of DPSCs on macroporous biphasic calcium phosphate granules.19 They performed a pilot study to determine the proper concentration of metformin. They stated that 100 µmol/L increased the proliferation of DPSCs the most. In addition, they analyzed ALP activity and used the MTT assay to evaluate differentiation and proliferation, respectively.19 As the present study found, metformin increased proliferation in the presence or absence of the scaffold, and had no significant effect on the differentiation of DPSCs cultured in the non-inducing medium. However, in the inducing medium, metformin increased differentiation.
Several studies have been performed to identify a proper growth factor for enhancing the osteogenic capacity of FDBA granules. Markopoulou et al. noted that recombinant human transforming growth factor-beta 1 (rhTGF-β1) enhanced the osteogenic differentiation of human periodontal ligament cells (hPDLCs) cultured on FDBA granules.10 In addition, Cenni et al. found that platelet-rich plasma (PRP) had a positive effect in in vitro conditions.9 However, according to in vivo results, PRP had no significant positive effect on the osteogenesis of bone mesenchymal stem cells (BMSCs) cultured on FDBA granules.15 Thus, our claim that metformin has a positive effect on osteogenesis in vitro should be analyzed in vivo as well.
Besides in vitro studies, several studies have been conducted to improve the osteogenic capacity of FDBA granules in vivo. Borie et al. evaluated the effect of autogenous particles and FDBA granules on osteogenesis, using a rabbit calvarial defect model.14 They found that FDBA granules caused lamellar bone formation after 90 days, while a mixture of autologous particles and FDBA granules caused woven bone formation over the same period of time.14 In addition, Dallari et al. mentioned in their study that PRP did not enhance the osteogenic capacity of FDBA granules in rabbit trabecular defects.15 Choukroun et al. found that PRP had no positive effect on the osteogenic capacity of FDBA granules in sinus lift procedures.12 In addition, Samandari et al. mentioned no positive effect for plasma-rich growth factor on the osteoinductive ability of FDBA during socket preservation.13 Thus, more in vivo studies are required to determine a proper material for improving the osteogenic capacity of FDBA granules.
The time- and concentration-dependent effects of metformin on the behavior of stem cells have been proven.26 However, a particular concentration of metformin does not seem to have the same effect on different types of stem cells. For instance, Śmieszek et al. stated that metformin at concentrations of 100 µM and 500 µM did not affect the proliferation of BMSCs significantly, while 10 µM metformin decreased proliferation.26 However, Wang et al. found that 10 µM metformin had no significant effect on the viability of human-induced pluripotent stem cell-derived mesenchymal stem cells (hiPSC-MSCs).20 Zhang et al. mentioned that the 10 µM/L and 50 µM/L concentrations of metformin had no significant effect on the proliferation of periodontal ligament stem cells (PDLSCs), but 100 µM/L metformin increased proliferation.17 In fact, due to the saturation of ligands, higher concentrations of metformin cannot promote its effects.27 Besides, higher concentrations of metformin may have adverse effects on the energy metabolism of cells, and result in a decrease in proliferation and differentiation.17, 28
The exact molecular mechanism of metformin is still unclear. Mu et al. proposed that metformin could enhance differentiation and proliferation through AMPK-mammalian target of rapamycin Complex 2 (AMPK-mTORC2) and Akt-mTOR Complex 1 (Akt-mTORC1) signaling pathways.29 However, Ma et al. stated that metformin increased the osteogenic differentiation of BMSCs by inhibiting glycogen synthase kinase 3 beta (GSK3β) and Wnt signaling pathways.18 In addition, they mentioned that AMPK played a crucial role in the abovementioned procedure.18 Several other studies have mentioned that metformin induces osteogenic differentiation through AMPK and mitogen-activated protein kinase (MAPK) pathways.20, 21, 29, 30 Thus, there is still controversy over the intracellular pathways regarding the effect of metformin.
In this study, the effect of metformin on the behavior of DPSCs was evaluated. Several other studies have been conducted to assess the reaction of other types of cells to metformin. The positive effect of metformin on the proliferation and differentiation of murine pre-osteoblasts,29 PDLSCs,31 BMSCs,32 and induced pluripotent stem cells20 has been described. In addition, Qin et al. showed that besides osteogenic differentiation, metformin could enhance the odontogenic differentiation of DPSCs.33, 34
Considering the limitations of applying autogenous bone grafts, a wide range of natural and synthetic scaffolds has been used in bone regeneration treatment.2 However, due to the lower osteogenic capacity of these scaffolds in comparison with autogenous grafts, applying them by themselves has yet to be considered.6 The addition of growth factors, one of the fundamental components of tissue engineering besides stem cells and scaffolds, can be deemed a possible method for enhancing the osteoinductivity of bone substitutes. In this study, a significant positive effect of metformin was shown in vitro. Therefore, this factor may improve the efficacy of both natural and synthetic scaffolds in vivo.
Since this study was performed in vitro, factors related to the recipient features, including the mechanical features of the tissue, possible inflammation, a heterogeneous tissue environment, and interactions between different cell types, were not considered. Therefore, for generalizing the results to in vivo conditions, performing animal and clinical studies is necessary.
Conclusions
The positive effect of metformin on cell proliferation and differentiation, in conjunction with FDBA granules, would be useful for its future application in fabricating FDBA granules with controlled release of this osteoinductive factor for bone tissue engineering. Besides, metformin can be applied in bone regeneration treatment to enhance osteogenesis.