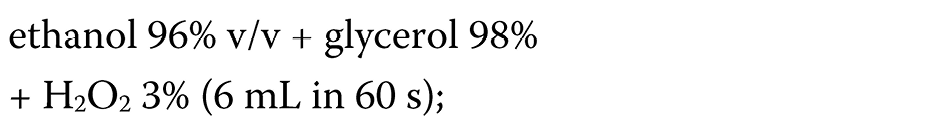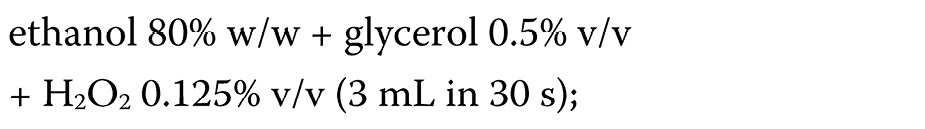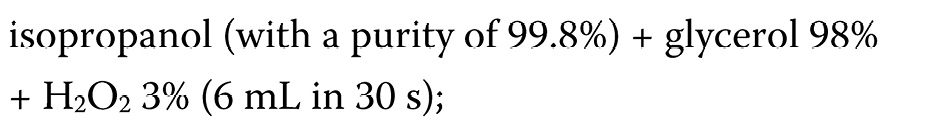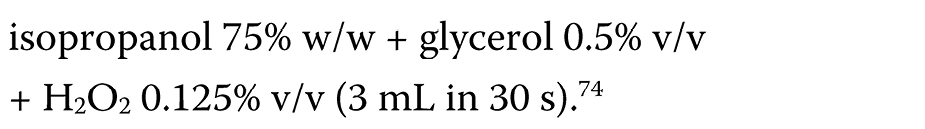Abstract
Healthcare-associated infections are well-studied in the literature, but remain a significant challenge for healthcare workers (HCWs) in dental practice. This type of infection is strongly correlated with the hand route of transmission of infectious agents. Thus, hand hygiene can be considered a crucial element in the prevention and control of infections.
The coronavirus disease 2019 (COVID-19) outbreak is an experience of a new human coronavirus infection that has been difficult for HCWs, such as dentists and dental assistants, to control. Handwashing (HW) is a keystone method for the prevention and control of spreading severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2).
In healthcare settings, contrary to the general consideration of HW as a simple practice, HW is regarded as a specific procedure that should be clearly instructed to HCWs. Handwashing is based on using a correct method and the necessary equipment, and its role should be continually emphasized to reinforce compliance. Proper HW might contribute to avoiding possible cross-infection during healthcare activities, particularly in the pandemic situation.
The aim of this article was to report on different HW techniques in medical and dental practice, and appropriate HW equipment to perform this simple but important procedure to prevent cross-infection, particularly during the current COVID-19 outbreak.
Keywords: hand hygiene, handwashing, cross-infection, COVID-19 outbreak
Introduction
Handwashing (HW) is an effective measure for the prevention of cross-infection in healthcare settings. While providing dental care, cross-infection may occur directly from person to person, or indirectly via soiled instruments, clothes or hands. Pittet et al. reported that the hands could be the principal route of transmission of microorganisms.1
Hand skin exhibits endogenous and exogenous microflora. The endogenous flora grows on the deeper layers of the skin and on hair follicles. It is residential, commensal and specific for each individual. It renews rapidly, and it is almost impossible to remove it completely; it may act as a biological barrier. By contrast, the exogenous flora is transient, colonizes the superficial skin layers and is mostly acquired via environmental routes. This flora mainly consists of saprophytes, commensal bacteria and fungi of the oropharyngeal sphere, and digestive microorganisms that are transferred from patients. The exogenous flora is harmful and pathogenic, but can be easily removed by means of clinical HW procedures.2, 3 In absence of correct hand hygiene, hand skin microflora, especially the exogenous flora, is frequently a source of the healthcare-related infections acquired while performing care activities.2, 3, 4 Hence, it is crucial to emphasize the key role of this simple but important activity in preventing cross-infection, particularly during the ongoing coronavirus disease 2019 (COVID-19) pandemic. This emergent outbreak is the 6th public health emergency of international preoccupation, and the etiological agent – severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) – is the 3rd most pathogenic human coronavirus that occurred in the last 2 decades.5, 6 This new human betacoronavirus (βCoV) is an RNA virus belonging to the Coronaviridae virus family.7 It has infectious potential through the respiratory droplets spread through coughing or sneezing by an infected individual, direct physical contact (e.g., shaking contaminated hands), or non-physical contact.3, 7, 8, 9 Distant contamination can be explained by the dynamic turbulent gas model, which has shown that pathogen-loaded droplets can circulate over a long distance.10, 11 This may happen even if a patient is not showing signs of the disease (coughing or sneezing).6 According to previous studies, the moving turbulent gas can be transported over a distance of 23–27 feet, which corresponds to 7–8 m.12, 13, 14
The viability of SARS-CoV-2 depends on its capacity to be stable on surfaces. Chin et al. reported that in vitro, the viral stability was up to 3 h on paper, 2 days on wood and fabrics, 4 days on glass and banknotes, and 7 days on stainless steel and plastic.15 Similarly, van Doremalen et al. reported that the viral stability of SARS-CoV-2 was higher on plastic and stainless steel than on copper and cardboard.16 Santarpia et al. reported significant environmental contamination in rooms where patients positive for SARS-CoV-2 were taken care of.17 Indeed, contamination was detected in all types of samples: air samples; personal items; room surfaces; and toilets. The presence of viral replication in the cell culture for some of the samples confirms the potential infectious nature of this virus.17 Smither et al. suggested in an experimental study that when the virus was transmitted within small-particle aerosols, it might remain viable for at least 90 min.18 However, it is worth mentioning that SARS-CoV-2 is sensitive to standard disinfection methods, even though it has high stability in favorable environment.15
Thus, proper measures are needed to prevent this deadly virus. Handwashing is considered one of the essential control measures for preventing the spread of this infection by healthcare workers (HCWs) until effective antiviral therapy and vaccination are discovered.5, 8, 19
The present review is a general overview of the importance of HW during the COVID-19 outbreak in dental practice. This review summarizes the mechanism of transmission of SARS-CoV-2, the HW procedure and the appropriate equipment needed to perform HW effectively in safe conditions. This work was designed as follows: the selection of databases was based on the principal electronic databases, i.e., MEDLINE/PubMed, Scopus (Elsevier), Science Direct Journals (Elsevier), and Google Scholar; the Medical Subject Headings (MeSH) terms used to search the articles were: “coronavirus”, “coronavirus disease 2019”, “COVID-19”, “COVID-19 outbreak”, “2019-nCoV”, “SARS-CoV-2”, “dental COVID-19”, “dental COVID-19 outbreak”, “dentistry COVID-19”, “handwashing COVID-19”, “hand hygiene”, “cross-infection”, and “healthcare workers”, with the use of “AND” and “OR” between the MeSH terms. The retained articles were studies, reviews, and reports from international organizations focusing on HW during the COVID-19 outbreak in dental practice. Some earlier publications were included to understand the HW procedures and to overcome the lack of recent scientific studies regarding the issues.
Compliance with handwashing
in medical practice
As cross-infection may occur in medical practice through HCWs’ contaminated hands, HW is highly recommended. However, compliance with correct HW practice may vary among HCWs. Before the 1990s, it did not exceed 40% because of several factors.20, 21 Joshi et al. reported that differences in HW practice among various medical staff members were due to such factors as workload, accessibility to soap dispensers, hand irritation, hand dryness due to chemical irritants, and the level of awareness regarding hand hygiene.22 This study concluded that even though HCWs understood the importance of HW, they washed their hands selectively, depending on the situation. The majority of HCWs washed their hands after patient care rather than before.22 This raises the need to emphasize the importance of HW in medical practice, particularly in dental practice, where HW compliance is relatively low and the risk of infection is high.9 Thus, continuing education and training programs in medical settings could increase HCWs’ HW compliance and the effectiveness of the procedures intended to prevent infectious diseases.23, 24, 25, 26
During the outbreak of severe acute respiratory syndrome (SARS) in 2003 and Middle East respiratory syndrome (MERS) in 2012, numerous studies recognized the impact of these occurrences on raising HW awareness and compliance in community settings in the affected regions. However, applicable procedures generally failed to be properly implemented.23, 24, 25 Fung and Cairncross reported that a high fatality rate for SARS was a helpful factor to remind the individuals living in Hong Kong about the importance of HW, and that HW compliance increased and remained high for nearly 2 years after the outbreak.25 Therefore, the preventative measures applied during those previous health emergencies may be used as a guide for HCWs during the current COVID-19 outbreak, but also after this situation to prevent possible cross-infection and to constantly review HCWs’ attitudes regarding protection against infections.
On March 11, 2020, the World Health Organization (WHO) announced that the COVID-19 outbreak was a pandemic phenomenon, and outlined numerous recommendations and guidelines to face the situation.27 The WHO’s multimodal hand hygiene improvement strategy (MHHIS) in saving lives from COVID-19 global pandemic provides the evidence and the recommendations retained from the 2009 guidelines on hand hygiene in healthcare to support healthcare facilities. The following 5 key steps formed this approach28, 29, 30:
– a change in the system that consists in providing permanent access to hand hygiene products (alcohol-based hand rubs (ABHR), water supply, soap, and towels);
– regular training and education for HCWs;
– the evaluation of HW and feedback (monitoring practices and knowledge);
– reminders in the workplace; and
– the promotion of an institutional safety climate with the active participation of all HCWs.
Several studies reported that the implementation of MHHIS brought overall good results all over the world with regard to staff education.16, 17, 31
How and when to practice handwashing in dental practice?
‘Hand hygiene’ is a general term referring to any action of hand cleansing involving HW, antiseptic HW, antiseptic hand rubbing, or surgical hand antisepsis.30, 32, 33 Handwashing is a “procedure of washing hands with plain or antimicrobial soap and water”; it can be performed by means of different methods, depending on the risk of infection from healthcare activities.30, 34 Three methods of HW can be practiced: routine HW; hygienic or antiseptic washing; and surgical washing (Table 1).
While HW can be considered a simple practice performed by everybody on an everyday basis, as a professional exercise, it needs to meet some specific requirements with respect to procedure and time (Figure 1)35, 38:
– the time required to achieve proper HW is 40–60 s;
– all jewelry or other accessories should be removed before HW;
– the hands should be held upward after rinsing;
– the hands should be dried carefully with a single-use towel; the last towel should be used to turn the faucet off, if no automatic source is available, before throwing it into a pedal bin;
– regarding surgical HW, the procedure is finished by brushing the fingernails and the subungual areas, where the concentrations of microorganisms are high, with a sterilized brush with or without antiseptic solution; the brush should be used only for the fingernails and not for the skin, as it could create micro-cuts or erosions, which might lead to the exposure of the underlying cutaneous layers to microorganisms and possibly enable infection.2
Independently of healthcare activities and procedures, HW is an imperative procedure for any person who comes into contact with patients, principally HCWs, such as dentists and dental assistants, who are at particularly high risk of the SARS-CoV-2 infection during the present COVID-19 pandemic.39, 40
Handwashing is indicated once the hands get soiled or contaminated.41, 42, 43 Peng et al. defined specific indications with regard to HW for oral health professionals during the COVID-19 pandemic as follows: before and after routine dental examinations or procedures; after touching non-disinfected surroundings and equipment; and after touching the oral mucosa, damaged skin or wounds, blood, body fluids, secretions, and excreta.9 Ding et al. consider toilets as high-risk areas in hospitals with COVID-19 patients, and emphasize the strong need for hand and environmental hygiene as an intervention against the transmission of COVID-19.44 Indeed, contaminated aerosols may come from 3 possible sources: the exhaled release from patients when using the bathroom; toilet-generated aerosols when the toilets are flushed of feces and urine; and the import of airborne particles from the stalls. This proves that the use of toilets in healthcare areas might result in vector transmission, especially hand transmission.44, 45
During the COVID-19 pandemic, HW is a crucial procedure, especially given the persistence of SARS-CoV-2. Indeed, this virus can be present on surfaces for a few hours up to several days, which exposes HCWs to a high risk of infection. There is no sufficient data concerning the viral load of coronaviruses on inanimate surfaces or the hands, and its transmissibility from contaminated surfaces to the hands in healthcare settings in an outbreak situation.46
Therefore, surface disinfection, especially of the frequently touched surfaces with the highest viral load, is important for preventing hand contamination. To ensure environmental cleaning and disinfection, the WHO recommends the consistent, correct and thorough cleaning of environmental surfaces with water and detergent as well as applying the commonly used hospital-level disinfectants, such as sodium hypochlorite, as effective and sufficient procedures.46, 47 Several studies investigated which disinfectant agents should be used for surfaces and the hands to protect against SARS-CoV-2. Many formulations were tested to assess their rapidity, spectrum of microbicidal activity, accessibility, and safety (Table 2). Furthermore, to limit the risk of airborne contamination, different air purifiers are relevant during this pandemic (Table 3).
During the COVID-19 pandemic, hand-rub products are widely used in healthcare settings as one of the infection control tools. In routine medical practice, the ABHR formulations (ethanol or isopropanol agents) are commonly applied, as recommended by the WHO.30 However, their effectiveness appears to be limited and does not meet the European standards.72, 73 Thus, modified formulations have been suggested. Suchomel et al. proposed the following modified formulations:
– the WHO I formulation (Equation 1):
– the modified WHO I formulation (Equation 2):
– the WHO II formulation (Equation 3):
– and the modified WHO II formulation (Equation 4):
In light of the ongoing circumstances, several reports from international organizations40, 75, 76, 77, 78 recommend the use of medical gloves as part of the personal protective equipment (PPE) to strengthen protection against potentially infectious biologic secretions.39 Ye et al. reported that gloves were the PPE most contaminated with SARS-CoV-2 at 15.4%.79 Hence, wearing gloves should not be an alternative to HW in healthcare practice, mainly because of rapid bacterial proliferation due to the humidity of the hands under the surface of gloves and the deterioration of gloves.4, 80
Additionally, HCWs should observe further precautions concerning the route of transmission of microorganisms by avoiding touching their face, eyes, mouth, and nose.9 Macias et al. reported that during the influenza A (H1N1) pandemic, face touching behavior was commonly observed on average 3.3 times per hour in the community.81 It was also reported that face touching behavior among medical students with their own hands was observed on average 23 times per hour, with contact mostly to the skin (56%), followed by mouth (36%), nose (31%), and eyes (31%).82 These results cannot be generalized to all HCW categories because of several factors, such as the duration of the experiment and the degree of awareness. However, they illustrate the higher risk of infection HCWs face as compared to the rest of the community, which makes HW procedures an effective and inexpensive preventative method to break the colonization and transmission cycle from the autoinoculation route, and to minimize the spread of infection.82 Furthermore, to avoid potential nosocomial infection, it is recommended to have short fingernails, no nail polish and no jewelry; otherwise, contamination can frequently occur even with proper HW.83, 84, 85, 86, 87
To summarize the timings of HW, the WHO recommends “My Five Moments for Hand Hygiene” approach as guidelines for dental practice as follows: before touching a patient; before clean/aseptic procedure; after body fluid exposure risk; after touching a patient; and after touching patient surroundings.86
Professional handwashing materials
In medical areas, a well-planned HW equipment set-up (washbasins, liquid soap dispensers, and hand wiping and drying systems) can protect HCWs from potential nosocomial infection and ensure correct HW. Indeed, Coleman et al. reported that contaminated washbasins and sink drain outlets were associated directly or indirectly with the hospital outbreaks of infection.6 Ye et al. reported that hand sanitizer dispensers accounted for about 20.3% of contaminated objects.79 Therefore, HW materials (washbasins, liquid soap dispensers, etc.) can be considered a possible source of microbial biofilm proliferation, and thus require good maintenance, regular cleaning and exclusive use for HW by HCWs.6, 79
With regard to HW agents, it is recommended to apply those indicated for professional medical use.87 These agents generally have the following features: are not perfumed; rarely induce allergic reactions; and are suitable for everyday use. Professional HW is based on the treatment of the hands with regular liquid soap or an antimicrobial product (soap, gel or solution), with the spectrum of activity targeted at microorganisms that are part of the cutaneous flora to prevent their transmission.76 Such products contain emollients, e.g., glycerine or aloe vera, that promote hand health and comfort. These components have a softening effect that prevents the occurrence of dermatitis, which may result from frequent HW, and also helps to ensure HW compliance.58 However, the specificity of the products indicated for antiseptic or surgical washing, as compared to simple wash products, consists in their net, rapid and persistent bactericidal and virucidal effect for several hours on the cutaneous flora of the hands.88 The products commonly used in medical practice are based on chlorhexidine from 2% up to 4%, alcohols between 60% and 70%, or iodine-based compounds.2, 4, 32 Regarding the soap form, bar soap should be avoided in hospitals to prevent creating a microbial niche that is consequently a source of contamination, unless it is intended for single use.37 According to several studies, bar soap can be a reservoir of various microorganisms and using it in hospital practice can transform HW into a vehicle for spreading infections, such as gastrointestinal infection, respiratory infection or cutaneous infection, including the SARS-CoV-2 respiratory infection.22, 89, 90 Thus, liquid soap is highly recommended instead of bar soap. Nevertheless, the risk related to the soap dispenser system must be controlled. The soap refill is screwed into the system and the liquid flows by gravity. To avoid contamination, the last drop should not be sucked into the cartridge, and the valves should be operated with the elbow to avoid direct contact with the soap dispenser.
In addition, the manual water faucets commonly installed in healthcare settings should be avoided because of the risk of infection and contamination. To avoid manual contact, the faucet should be controlled by a pedal or the elbow. Ideally, an automatic faucet (electro-sensitive) with a sufficiently large and deep washbasin to avoid splashing contamined water should be used for HW. Notably, the area reserved for HW should be independent of the instrument disinfection area.37 For simple washing and antiseptic washing, warm water is more recommended than water of extreme temperature (hot or cold) to avoid cutaneous irritation and dryness.58 Regarding surgical HW, using pure water is practiced; according to the microbiological quality control standards, ‘clean water’ is of level 1, which is obtained by the chlorination (0.1 mg/L) chemical treatment of the water supply.91
After HW, meticulous hand drying is the final and necessary step to prevent the transmission of microorganisms, which is caused more often by wet hands than by dry hands due to the nature of the clinical practice, frequently exposing HCWs’ hands to wetness. In contrast, hand drying may decrease the number of microorganisms translocated through touch.91, 92 For wiping hands, single-use hand towels are recommended after simple and hygienic HW, and sterile hand towels after surgical HW. In general, the following characteristics of hand towels are recommended for safe hand drying: they should be absorbent; flexible to fit the hand’s anatomy; and resistant to tearing. The hand drying system has an influence on the protection against cross-infection.
Apart from being slow and noisy, electric hand dryers are to be avoided in healthcare settings because, according to some studies, the waterborne microorganisms dispersed by hand dryers can become aerosols.2 Kimmitt and Redway studied hand drying methods by comparing paper towels, a warm air dryer and a jet air dryer for their potential to disperse viruses and contaminate the immediate environment during use.93 They found that the use of a jet air dryer led to a significantly greater and further dispersal of microorganisms from artificially contaminated hands as compared to paper towels and a warm air dryer.93 Hence, it is necessary to make a careful choice of the hand drying system in healthcare settings, where the risk of cross-infection is high, especially during critical situations like the present outbreak, to safeguard patients and HCWs.92, 93 It is worth noting that before wearing gloves, HCWs should dry their hands completely after hand disinfection, as residual humidity, such as that from ABHR, may increase the risk of glove perforation during daily dental care.94 Lastly, for collecting used hand towels, a pedal bin installation is required near the HW set-up to avoid touching the bin, and thus to limit the risk of contamination of HCWs’ hands.
Conclusions
In medical practice, HW is a basic measure for preventing the transmission of germs and the spread of infection related to healthcare activities. The occurrence of more or less serious nosocomial infection is a real risk in dental practice, and prevention concerns are paramount. Hence, HW is a very important and simple – but indispensable – procedure to prevent the manual transmission of germs.