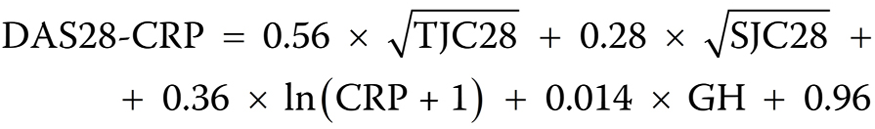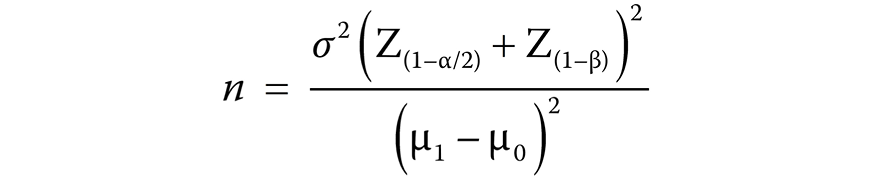Abstract
Background. The periodontal condition has a reciprocal relationship with rheumatoid arthritis (RA). Rheumatoid arthritis patients are reported to present with more serious periodontal disease (PD) as compared to non-RA patients.
Objectives. This study aimed to evaluate the effects of non-surgical periodontal treatment on Vietnamese patients with active RA and PD, where the clinical characteristics and serum indices of the patients were of interest.
Material and methods. We conducted a randomized clinical trial (RCT) on 82 RA patients with PD. The patients were randomly divided into 2 groups: the intervention group, consisting of patients who received oral hygiene instructions, scaling and root planing; and the control group, consisting of patients who received oral hygiene instructions only. Both groups received the same treatment plan for RA. The Disease Activity Score 28 based on C-reactive protein (DAS28-CRP), disease activity classification, rheumatoid factor (RF), erythrocyte sedimentation rate (ESR), anti-citrullinated protein autoantibodies (ACPAs), and C-reactive protein (CRP) were monitored, with the measurements taken at 3 months and 6 months following the treatment.
Results. The 2 groups exhibited similar parameters at baseline. In the intervention group, DAS28-CRP and disease activity classification were significantly reduced at 3 months after treatment as compared to the baseline data. At 6 months following the treatment there was a significant decrease in ESR, ACPAs and DAS28-CRP in the intervention group, while the control group showed a decrease only in ACPAs. Further, when comparing the intervention and control groups at 6 months following the treatment, there were no differences between the groups in the ACPAs, RF and CRP serum levels.
Conclusions. Non-surgical periodontal treatment can significantly reduce DAS28-CRP, disease activity classification, ESR, and the ACPAs level in serum, and can be applied to reduce RA severity in RA patients with PD.
Key words: rheumatoid arthritis, inflammation, periodontitis
Introduction
Rheumatoid arthritis (RA) patients usually have periodontitis and, in comparison with those without RA, also have a worse periodontal condition.1, 2 Periodontitis is a common chronic infection, with a global incidence in adults varying from 10% to 60%, depending on different diagnostic criteria.3 Reports have indicated a high incidence of diabetes mellitus, atherosclerosis, myocardial infarction, stroke, and RA in patients with periodontal disease (PD).4, 5, 6, 7 In our recent report on the association between PD and RA in Vietnamese patients, we found that the periodontal condition of RA patients is more serious as compared to non-RA patients.8 There was an association between the periodontal condition and clinical symptoms, such as the biochemical and immunological characteristics of RA.8
Rheumatoid arthritis is a common autoimmune disease; its chronic progression includes symptoms at joints and other body parts with varying severity, which can lead to the weakening of bones and connective tissues.9 Studies have shown that patients with RA are at high risk of cardiovascular diseases and malignancy.10, 11 Rheumatoid arthritis is prevalent worldwide, with an incidence rate of 0.5–1% in adults. In Vietnam, 0.5% of the population has RA. Among hospitalized patients, the RA rate is as high as 20%. In particular, 70–80% of patients are female, and 60–70% of them are over 30 years old. The disease is hereditary in some cases.12 Previous clinical studies show different results with regard to the role of non-surgical periodontal treatment in RA. In their research, de Noronha Pinho et al. found that the relationship between the RA and PD disease activities was not clear.13 However, it is evident that due to periodontal treatment, it is possible to control inflammation and avoid tooth extraction. A recent study by Zhao et al. demonstrated that RA patients with PD who received non-surgical treatment showed improvement in the clinical outcome for RA.14 Recently, a new classification for PD has been developed based on previous classifications.15, 16 Machtei et al.17 defined the term ‘established periodontitis’ with regard to ‘periodontitis’, which was used in clinical studies,18, 19 and ‘severe periodontitis’, referred to by the Centers for Disease Control and Prevention (CDC) in partnership with the American Academy of Periodontology (AAP).20
In this study, we evaluated the effects of non-surgical periodontal treatment on the clinical characteristics and serum indices in 82 Vietnamese patients with active RA and PD, using Machtei’s classification. The Disease Activity Score 28 based on C-reactive protein (DAS28-CRP), disease activity classification, rheumatoid factor (RF), erythrocyte sedimentation rate (ESR), anti-citrullinated protein autoantibodies (ACPAs), and C-reactive protein (CRP) were measured and monitored at baseline, and at 3 and 6 months following the periodontal treatment.
Material and methods
Study design
We conducted a clinical intervention study on RA patients with periodontitis who were treated at the Department of Rheumatology, Cho Ray Hospital, Ho Chi Minh City, Vietnam, from August 2013 to March 2015. The trial registration number is ISRCTN40789708 at www.isrctn.org.
Eligibility criteria
Rheumatoid arthritis patients with PD were recruited in the present trial in the same way as in our previous study.8 Rheumatoid arthritis was diagnosed using the American College of Rheumatology/European League Against Rheumatism (ACR/EULAR) 2010 guidelines,21 while the periodontal condition was determined according to the criteria specified by Machtei et al.,17 with more than 4 real teeth (regardless of the largest third molars). All patients had established RA (>12 months), and were treated with disease-modifying anti-rheumatic drugs (DMARDs), non-steroidal anti-inflammatory drugs (NSAIDs) and corticosteroids. The exclusion criteria were as follows: patients under the age of 30 years; RA patients with other conditions, such as polymyalgia, gout, pseudogout, spinal stiffness, Sjögren’s syndrome, diabetes mellitus, and malignant diseases; patients who had recently received periodontal treatment (3 months or less prior to our study); and patients who were pregnant or breastfeeding.
Randomization
Independent staff members randomly assigned patients into the intervention or control group, using sealed envelopes. Each envelope was marked with a ‘0’ or a ‘1’. A patient was assigned to the control group if their envelope had a ‘0’; otherwise, the patient was assigned to the experimental group. A separate envelope was used for each kind of treatment, and the type of treatment for each patient was documented.
Blinding
Whether a patient belonged to the intervention or control group was fully disclosed to the patient and the clinician treating PD (the main investigator). Due to the nature of the intervention, blinding was not applied to the patients and the main investigator. The outcome was blinded to the assessing investigators and the statistician.
Interventions
Both the control and intervention group were treated with conventional regimens. The control group received only oral hygiene instructions, while the treatment group received an advanced periodontal treatment intervention consisting of supragingival scaling and root planing, the effectiveness of which was evaluated after 3 and 6 months, which was followed by retreating. The research team consisted of a periodontist, who recorded the periodontal indices, a rheumatologist, who assessed the swollen joints, and an assistant, who recorded the information. The researchers who treated the RA patients for periodontitis were not involved in the periodontal, joint or other clinical examinations. The team members were trained by experts before the study, achieving consistency from 80% to 90.4%.
Outcomes
Periodontal disease assessment
The assessment of PD was conducted in a similar manner as described in of our previous study.8 The periodontal indices included the plaque index (PlI), the gingival index (GI), the percentage of sites with bleeding on probing (%BOP), probing pocket depth (PPD), and clinical attachment loss (CAL), evaluated at 6 positions in all teeth.22, 23
Rheumatoid arthritis clinical assessment
Rheumatoid arthritis disease activity was measured as in our previous study.8 We assessed RA disease activity based on CRP (DAS28-CRP), with the following equation (Equation 1):
where:
TJC28 – tender joint count (0–28);
SJC28 – swollen joint count (0–28);
CRP – C-reactive protein level [mg/L];
GH – patient’s global health self-assessment by means of the visual analog scale (VAS).24
The determination of the DAS28-CRP score regards the pain and swelling of 28 joints, comprising shoulder joints, elbow joints, wrist joints, and knee joints. We applied the tool available at http://www.4s-dawn.com/DAS28 to compute the value of DAS28-CRP. Then, DAS28-CRP was classified as remission (DAS28-CRP < 2.3), low (2.3 ≤ DAS28-CRP < 2.7), moderate (2.7 ≤ DAS28-CRP < 4.1), or high disease activity (DAS28-CRP ≥ 4.1).
Measurement of ESR, and the RF, ACPAs
and CRP levels in serum
We determined the serum concentration of RF and CRP with the use of the latex particle-enhanced method.8 An RF serum value of <12 IU/mL was defined as normal (negative) and >12 IU/mL as positive. The CRP serum level was defined as normal if it was <6 mg/L. The serum level of ACPAs was determined by means of the sensitive enzyme-linked immunosorbent assay (ELISA). The ACPAs serum level was defined as normal or negative (<25 IU/mL) or positive (≥25 IU/mL). Erythrocyte sedimentation rate was measured using the flow through a capillary tube. The normal serum ESR was recorded at 5–10 mm/h.
Sample size
Eighty-two patients with RA were randomly split into 2 groups: treatment group (n = 41); and control group (n = 41). The sample size was calculated as follows (Equation 2):
where:
n – sample size;
σ2 – population variance;
Z(1–α/2) – critical value of normal distribution at α/2;
Z(1–β) – critical value of normal distribution at β;
μ1 and μ0 – means of the 2 groups.
We first derived the value of (µ1 − µ0) from a study by Ortiz et al.25 According to them, the average Disease Activity Score 28 (DAS28) in RA patients after periodontal treatment was 3.51 ±1.11, with a change in DAS28 of 0.6, defined as treatment response.21, 25 Thus, we used a value of 0.6 for (µ1 − µ0). Using α = 5% and a power of 80% in Equation 2, we obtained a sample size of 27 (n = [1.11(1.96 + 0.84)2] / (0.6 − 0)2 = 27) patients for each group. According to the internal statistics provided by Cho Ray Hospital, 30–35% of patients cancel the re-examination. Therefore, in order to account for a sampling error and the potential sample size reduction, a sample size of 41 patients was used for our study.
Research ethics
The study was conducted in full accordance with the World Medical Association Declaration of Helsinki from 2008, and was approved by the Ethics Committee of the University of Medicine and Pharmacy at Ho Chi Minh City in Vietnam (No. 1781/DHYD-HD). Written informed consent was obtained from all participants prior to inclusion. At the end of the study, patients from the control group also received the same periodontal treatment as the treatment group.
Statistical analyses
We reported the mean (M) and standard deviation (SD) values for normally distributed data with the Shapiro–Wilk test, and the median (Me) and interquartile range (IQR) values otherwise. Differences between the 2 groups were assessed using the independent samples t test. The Mann–Whitney U test was applied to Me and IQR. For intergroup comparison, the quantitative parameter values between the groups were assessed with the Wilcoxon test. The χ2 test and Fisher’s test were used for rate comparison. The odds ratios (ORs) and 95% confidence intervals (CIs) were also reported. Statistical significance was defined at p < 0.05. We performed statistical analyses using the Stata statistical software, v. 13.0 (StataCorp, College Station, USA).
Results
Parameters of the 2 groups at baseline
The flow diagram of the study is shown in Figure 1. Between the control and treatment groups there were no differences in the distributions of age, gender, place of residence, occupation, smoking status, number of teeth examined, and duration of RA. Among the treatment group, the average age was 52.9 ±8.2 years and the proportion of women was 88%. Among the control group, the average age was 51.9 ±9.0 years and the proportion of women was 93% (Table 1). Besides, DAS28-CRP, ESR, and the serum levels of RF, CRP and ACPAs at baseline were also similar in the 2 groups. Around 2–3 patients per group were lost at 6 months due to not coming back (missing data).
Changes between the groups in the clinical parameters of periodontitis after 3 and
6 months in relation to baseline
On the 1st day of the examination, both the control and treatment groups were similar in terms of PlI, %BOP, periodontitis severity, and the CAL level (Table 2). On the other hand, GI, PPD and CAL were higher in the treatment group than in the control group (p < 0.05). Patients with RA in the treatment group tended to have more severe soft tissue inflammation than patients in the control group. After 3 months of treatment for periodontitis, most of the indices, including GI, %BOP, PPD, and CAL, significantly improved in the treatment group (p < 0.001). These indices in the treatment group continued to decline after 6 months (p < 0.001) as compared to baseline. The difference between the 2 groups became more evident as measured at 3 and then at 6 months (p < 0.001). After 6 months, the ORs for PlI, GI, %BOP, and PPD between the treatment and control groups were 0.80, 0.06, 0.32, and 0.02, respectively. The intensive periodontal treatment improved the periodontal status in patients with RA. Meanwhile, these indices in the control group stayed almost unchanged as compared to baseline.
Comparison of the changes in the clinical and paraclinical indices of rheumatoid arthritis after 3 and 6 months between the groups
After 3 months of periodontitis treatment, the clinical indicator of RA (DAS28-CRP) decreased in the control group; however, the difference was not statistically significant (p > 0.05). After 6 months, all indicators decreased in the treatment group as compared to the control group, and the difference was statistically significant (Table 3). The treatment group showed reduced DAS28-CRP (p < 0.001) and disease activity level (p < 0.001) after 3 months, while the reduction in the control group was not statistically significant (p > 0.05). No significant difference was found between the 2 groups. However, after 6 months, both the treatment and control groups showed significantly decreased DAS28-CRP and disease activity levels (p < 0.001 for the treatment group and p < 0.05 for the control group). In particular, when comparing the 2 groups after 6 months, the treatment group significantly differed from the control group in terms of DAS28-CRP (p = 0.013; OR = 0.60) and disease activity level (p = 0.028). The ORs for the disease activity level with regard to the treatment and control groups were 0.35, 0.16 and 0.13, respectively. Thus, the intensive periodontal treatment led to a significant reduction in the disease activity indices in patients with RA as compared to those receiving only basic treatment.
Changes in the immunohistochemical features of rheumatoid arthritis after 3 and 6 months of periodontal treatment
The comparison of the RA immunohistochemical features between the treatment and control groups after 3 and 6 months is presented in Table 4. Some patients were not able to complete immunohistochemical tests (missing data). The results of periodontal treatment showed that the immunohistochemical indices decreased in both groups; these changes became more evident in the treatment group, but no statistically significant difference was found (p > 0.05). In particular, the RA patients who were treated for advanced periodontitis showed the ACPAs level (Me) decreasing from 156 to 42.7 (p < 0.001), while the ACPAs level (Me) in the control group decreased from 186 to 102.3 (p = 0.032). After 6 months, the treatment group showed significant reductions in serum ESR (p < 0.001), ACPAs (p < 0.001) and the ACPAs positive test (p = 0.014), while the control group showed a reduction only in the ACPAs concentration (p = 0.032). As the 2 groups were treated with the standard regimens performed at Cho Ray Hospital, the clinical and paraclinical parameters decreased, but the treatment group showed a more significant reduction than the control group. The ORs of RF, CRP and ACPAs between the treatment group and the control group were 0.96, 0.99 and 0.75, respectively. However, when comparing the 2 groups, this difference was not statistically significant. There was no reduction in the serum levels of RF and CRP during the treatment in either group.
Discussion
Clinical studies show different results regarding the role of non-surgical periodontal treatment in RA. Our study is the first to investigate comprehensively the effect of non-surgical periodontal treatment on the clinical characteristics and serum indices in Vietnamese patients with active RA and PD. The present RCT was performed on a large number of patients and with a long follow-up period (Table 5), with the full assessment of the RA condition, considering disease severity and immunohistochemical features. The study showed that after 6 months of treatment for RA and periodontitis, all RA clinical parameters decreased. Meanwhile, in the control group, only a few clinical parameters decreased, which is consistent with the results of Al-Katma et al.26 and Okada et al.27 In a study by Zhao et al., the authors divided patients into 4 groups: PD+/RA+; PD−/RA+; PD+/RA−; and PD−/RA−.14 In the PD+/RA+ group, the levels of all RA markers (DAS28, high-sensitivity CRP – hsCRP, ESR, and ACAPs) decreased significantly after non-surgical periodontitis treatment within 1 month.14
Most of the studies report no decrease in the serum CRP levels after reassessment. The CRP level increases in systemic inflammatory diseases, such as RA, or infections, such as PD. In our study, there was a statistically significant reduction in the CRP levels in the treatment group, while in the control group, the reduction was not statistically significant, which is consistent with 2 other studies.14, 28 Addressing the inflammation of the periodontal tissue after non-surgical treatment can be very important for reducing the CRP levels. In their study evaluating the serum ACPAs levels, Okada et al. did not find any difference after treatment, which could potentially be attributed to the small sample size and the short follow-up period.27 On the other hand, Lappin et al. found a slight difference.29 However, this study had no control group and the participants were not RA patients.29 In a study by Kaushal et al.,30 the value of the ACCP index, when evaluated for each group, showed no significant difference before and after the intervention, which is similar to the results of Okada et al.27 This may be due to the insufficient sample size or the mild severity of PD in the study group. However, the CRP levels in the treatment group at 2 months after treatment were significantly different from baseline.30 Our study showed that after the combined treatment of PD and RA, ACPAs and the rate of ACPA positive tests significantly decreased (p < 0.01) (Table 5).
In their study, Cosgarea et al. assessed the impact of non-surgical treatment on the clinical periodontal indicators, the inflammation index and quantified periodontal bacteria in the gingival fluid of patients with PD and RA, in comparison with patients suffering from PD only.28 Their results showed that in both groups there was improvement in the clinical periodontal indicators. In the RA group, DAS28, CRP and ESR were decreased; however, only the reduction in CRP was statistically significant at 3 months. The DAS28 change was positively correlated with the amount of Porphyromonas gingivalis and was inversely correlated with PlI. The authors concluded that non-surgical treatment helped to improve the periodontal status of PD patients, with or without RA. Non-surgical treatment not only improved the periodontal status, but also reduced CRP in the group with both PD and RA.28 However, in a study by Mariette et al., the results showed that these interventions effectively reduced bacteria, but did not improve the status of RA.31 In patients with ACPAs(+), such treatment was not effective for RA.31 Our study showed that the treatment of RA in combination with non-surgical periodontal treatment improved the RA condition. After 6 months of advanced periodontal treatment, the Me value of DAS28-CRP decreased from 4.2 to 3.2. The effect on the treatment group was more significant as compared to the control group. After the intervention, the Me value of the serum ACPAs level decreased from 156 to 42.7. Furthermore, the rate of ACPAs positive test also decreased from 69.2% to 59%.
Limitations
The limitation of our study is that RA treatment methods may have affected the efficiency of PD treatment.18, 25, 30 However, all RA patients had established RA (>12 months), and were treated with DMARDs, NSAIDs and corticosteroids. It is important to keep in mind that RA is a chronic condition, which requires from patients changing their medications upon the recommendations of doctors. Hence, it is difficult to obtain exactly homogeneous study groups. Moreover, the combination of local treatment (periodontal sites) and a systemic intervention (RA treatment) may decrease the RA severity score and the PD condition synergistically. Previous studies also failed to obtain RA patient groups that were treated identically.27, 30, 32 Although we were not able to control RA treatment methods, our previous cross-sectional, descriptive study found that the incidence of periodontitis cases in the RA group was significantly higher than in the control group (67% and 28%, respectively), while severe and moderate periodontitis were common in the RA group.8 Therefore, we suggested that interdisciplinary and simultaneous treatment for both diseases be necessary to ensure overall efficiency. Finally, we mentioned that by the end of the study, all patients were to receive advanced periodontal treatment, including supragingival scaling and root planing. There was no risk of participating in the study, as after finishing the study, the patients from the control group received the same non-surgical periodontal treatment as the study group.
Conclusions
In conclusion, our study provided comprehensive evidence of the effect of non-surgical periodontal treatment on the clinical characteristics and serum indices in patients with active RA and PD. Non-surgical periodontal treatment can help decrease RA severity by significantly reducing DAS28-CRP, disease activity classification, the serum ACPAs levels, and ESR in patients. Dentists and rheumatologists should provide care as well as the treatment and prevention of dental diseases for RA patients in order to achieve better outcomes for both RA and PD.