Abstract
Background. The growth and proliferation of gingival fibroblasts are important in the process of oral wound healing, and photobiomodulation (PBM) might be able to modify this process.
Objectives. The aim of the current study was to evaluate the biomodulatory effect of a single session of laser PBM by means of 810 nm and 940 nm diode lasers alone and their combined application with different fluencies on human gingival fibroblasts (HGFs).
Material and methods. Cells were provided by the Pasteur Institute, the National Cell Bank of Iran (NCBI) (C-165). Laser irradiation was carried out using 810 nm, 940 nm and 810 nm + 940 nm in the continuous wave (CW) mode, 100 mW, and energy densities of 0.5, 1.5 and 2.5 J/cm2. Cell viability was evaluated at 24 h with the MTT assay. Trypan blue staining was used to evaluate proliferation 24, 48 and 72 h after laser therapy. Propidium iodine was used to stain DNA and the cell nucleus.
Results. Laser irradiation (810 nm, 0.5 J/cm2) increased the viability of gingival fibroblasts, while this dose had an inhibitory effect with 940 nm. No positive effect on cell viability was found with other settings at 24 h. The viability results were not statistically different from those of the control in the dual wavelength group. At all single-laser irradiation doses, the cell proliferation results were lower as compared to the control at 48 and 72 h. The dual wavelength group results were significantly better than those of the control for the 1.5 J/cm2 and 2.5 J/cm2 energy densities (p < 0.001). Propidium iodine staining showed no negative effect of laser irradiation on the cell nucleus in any of the groups.
Conclusions. Although a single irradiation dose of 810 nm, 0.5 J/cm2, resulted in a positive effect on cell viability at 24 h, no statistically significant stimulatory effect on viability and proliferation was observed for the other single wavelength group. When a combination of the 2 wavelengths was used, better results were observed as compared to the control, which needs to be further investigated in future studies.
Key words: wound healing, cell proliferation, lasers, cell survival
Introduction
Wound healing is a complex physiological process involving biological clot formation, angiogenesis, granulation tissue formation, and re-epithelialization. The growth and proliferation of various types of cells, such as fibroblasts, play important roles in this process,1 with fibroblasts having a key role and a critical anti-inflammatory effect. They are involved in the contraction of the wound and the production of collagen, elastin, fibronectin, and proteoglycans.2 Unlike skin fibroblasts, adult gingival fibroblasts are much more similar to embryonic fibroblasts in terms of morphology, growth, migration ability, and cytokine production, which is probably the reason for the rapid improvement of oral ulcers with the lowest amount of scars.3 Therefore, their proliferation and migration are essential for accelerating wound healing, and success in increasing their proliferation can directly influence the healing results.4, 5
Low-level (intensity) laser therapy (LLLT, LILT), or better called photobiomodulation (PBM), refers to the use of photons to modulate biological activity.6 This type of therapy uses non-thermal laser light, mostly from the red and near-infrared region of the spectrum. Its ability to stimulate the proliferation of different cell types has been shown to be its most important physiological effect.7 The influence of LLLT on the migration of gingival fibroblasts and the synthesis of collagen as well as its anti-inflammatory effects, which are directly related to the wound healing process, have also been shown in previous studies.2,
Although the mechanisms behind the biomodulatory effects of different wavelengths require further investigation, studies have shown that visible to near-infrared light is thought to be absorbed by mitochondrial and non-mitochondrial photoacceptors. The best known ones are mitochondrial respiratory chain components, which cause an increase in reactive oxygen species (ROS) and adenosine triphosphate (ATP) or cyclic adenosine monophosphate (cAMP), and initiate a signaling cascade, thereby promoting cellular proliferation.7, 11
Most studies on wound healing have been conducted on skin fibroblasts or epithelial cells. In recent years, in vitro and in vivo studies have also evaluated the effect of phototherapy on oral wounds and gingival fibroblasts, with different devices and settings.2, 3, 10, 12
The laser effect depends on various parameters, such as wavelength, power, energy density, and the duration and schedule of laser irradiation. Despite various studies in this field, there is still no optimal protocol for the application of PBM in dental treatment due to inconsistencies in the study design, a wide range of parameters involved, and phenotypic and genotypic differences that might exist in the cell lines used. Previous studies have mostly focused on certain wavelengths of the laser in the range of 600–810 nm, with few comparisons of different wavelengths.2, 4, 12, 13, 14, 15 Moreover, there are very few studies evaluating the simultaneous application of 2 wavelengths, which seems to have the potential of a novel synergistic effect due to different penetration depths and absorption by specific chromophores.16, 17, 18
Near-infrared laser dental devices with wavelengths of 810 nm and 940 nm have currently become popular adjunctive tools in different areas of dental practice (especially in periodontal treatment and soft-tissue oral surgery), and there is a limited number of studies comparing these wavelengths in order to find the ideal laser settings for adjunctive photobiostimulatory application in oral wound healing. This study was designed to evaluate the biomodulatory effect of a single irradiation session of these 2 lasers alone and their combined irradiation with different energy densities, chosen based on the previously reported suitable energy densities, on human gingival fibroblast (HGF) cells.
Material and methods
This study was approved by the ethics research committee of Zahedan University of Medical Sciences, Iran (IR.ZUMS.94.5.11-7347). Human gingival fibroblasts were provided by the Pasteur Institute, National Cell Bank of Iran (NCBI) (C-165; NCBI, Tehran, Iran). This cell line was cultured in the minimum essential medium α modification (αMEM) containing 10% of fetal bovine serum (FBS) (Gibco®, Grand Island, USA) in sterile flasks, in 5% CO2 and at 37°C, in an incubator. After 2–3 days, the culture was replaced and after 1 week, the cells were passaged.
The third-passage cells were cultured at 5 × 103 cells per well in 96-well plates (Zhejiang Sofra Life Sciences Co., Ltd., Zhongguan, China) and the type of treatment was identified according to numbering. Six wells were allocated for each energy density at each study time, and the average result was reported.
Laser irradiation
Diode lasers with wavelengths of 810 nm (Picasso®; AMD Lasers LLC, Indianapolis, USA) and 940 nm (Epic®10; Biolase, Irvine, USA) were used. A 400-micron fiber and an output power of 0.1 W were used for both lasers. The fiber tip was placed perpendicular to the bottom of each well at a fixed distance of 15 mm to produce an irradiation spot size of 0.8 cm in diameter to cover only 1 well of a 96-well plate.
In order to have 3 energy densities of 0.5, 1.5 and 2.5 J/cm2, the cell culture wells were irradiated from underneath the plates for 2.5, 7.5 and 12.5 s, respectively. The output power was 100 mW and the power transmitted through the transparent, flat-bottom culture plates was checked with a power meter (Nova II®; Ophir Photonics, Jerusalem, Israel) before irradiation. An empty well was placed between the cultured wells in order to prevent overtreatment. Wells other than the one being irradiated were covered with a black cardboard. In order to evaluate the combined effect of the 2 wavelengths, a third group was also assessed. For this purpose, in order to produce the same energy densities as in the case of the single wavelength groups, each energy density was reduced by 2 for each wavelength to achieve the same density together. The 810 nm wavelength was applied first, followed by the 940 nm wavelength. The irradiation process was conducted in a semi-dark room, with no ceiling light turned on over the counter where laser irradiation was performed. This was to limit unintentional light from the environment reaching the cells. The control wells received no laser irradiation, but their plates were also removed from the incubator for the same duration as the laser-irradiated group wells to provide the same situation for both groups.
Evaluation of cell survival
Cell viability was assessed by evaluating mitochondrial activity by means of the MTT colorimetric assay 24 h after irradiation with different lasers. In the MTT method, the mitochondrial succinate dehydrogenase enzyme causes a breakdown in the MTT ring solution to change it to insoluble blue formazan; then, using photometry, the amount of formazan is measured and the intensity of the produced color is related to the number of live cells. For this, we placed the cells in the incubator for 24 h. Then, 10 μL of the MTT solution and 90 μL of αMEM containing 10% FBS were added to each well and placed in the incubator for 3–4 h at 37°C. Formazan crystals were dissolved by adding dimethyl sulfoxide. Subsequently, spectrophotometry was used to measure the cell metabolism. Light absorption was read using the enzyme-linked immunosorbent assay (ELISA) with the ELx808™ absorbance microplate reader (BioTek Instruments GmbH, Bad Friedrichshall, Germany) at a wavelength of 540–690 nm. Results were reported as percentage, and the control of this test was considered as 100%.
Evaluation of cell proliferation
The trypan blue assay was used to evaluate the number of cells in the culture after the application of LLLT. The trypan blue dye can penetrate only the porous, permeable membranes of the lethally damaged (dead) cells, which is clearly detectable under optical microscopy (OM) and the total number of viable cells is counted. Cells in the amount of 5 × 103 were cultured in the wells of 96-well plates. Then, 24, 48 and 72 h after irradiation, the cell number was determined by counting the viable cells with the trypan blue dye exclusion assay. For each time period, the cells from 6 wells of each group were counted and their increased ratio was reported numerically in comparison with the baseline number of cells.
Propidium iodide staining
In order to study the effects of laser irradiation on the nucleus of the cultured cells, after 72 h, propidium iodide was prepared with 1 mg/mL phosphate-buffered saline (PBS) and 100 times diluted, and 10 μL of it was added to each well of the cell cultures. After 1–2 min, the samples were washed with the PBS solution and examined under a fluorescence phase contrast microscope (BX51; Olympus Corp., Tokyo, Japan) for the morphological changes of the nucleus (pyknotic nuclei and nuclear fragmentation).
Statistical analysis
The data was analyzed using Student’s t test and subjected to the three-way analysis of variance (ANOVA). After that, Tukey’s post-hoc test was used for further comparisons between the subgroups. All statistical analyses were performed at a significance level of 0.05 and expressed as mean (M) ± standard deviation (SD) through graphics. The R statistical software, v. 3.3.3 (the R Project for Statistical Computing, Vienna, Austria; http://www.r-project.org), and SPSS for Windows, v. 16.0 (SPSS Inc., Chicago, USA) were used.
Results
Cell survival
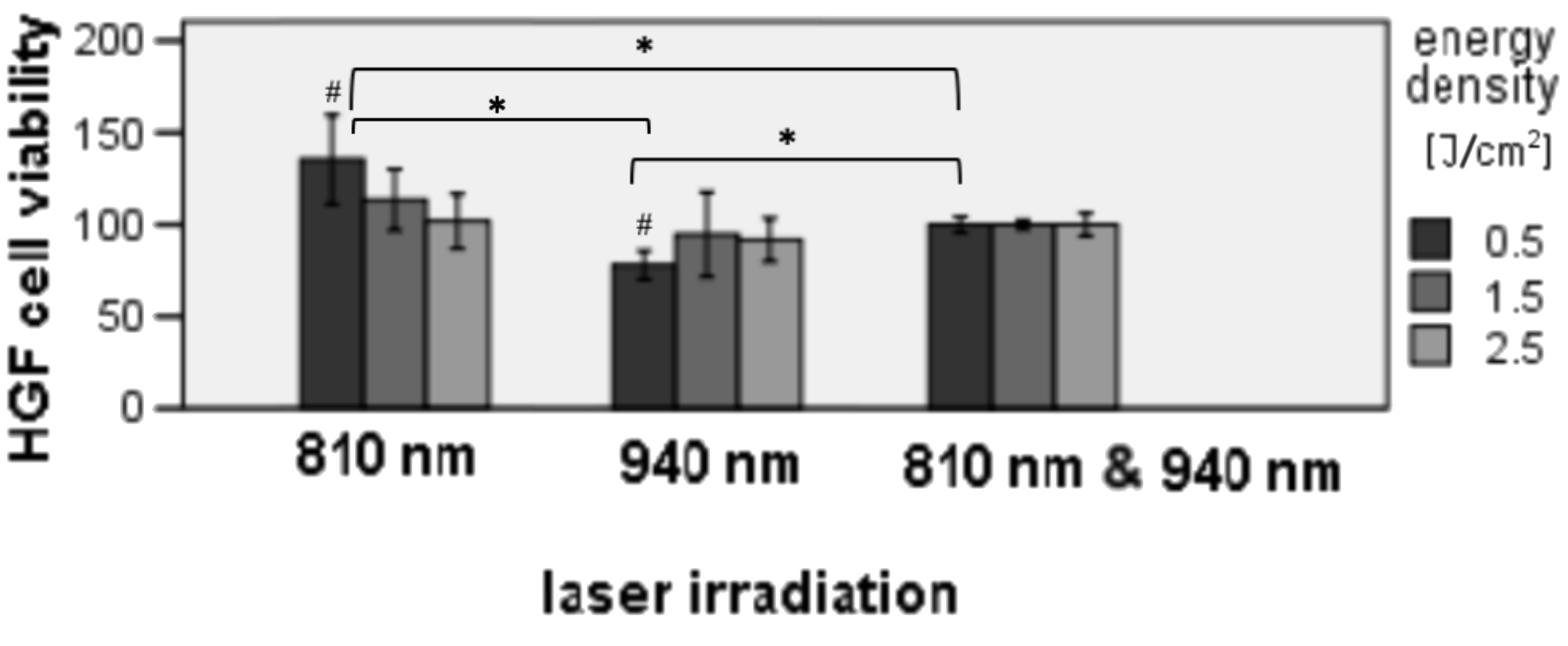
The MTT assay was used to determine the effect of laser irradiation on cell survival at 24 h. (Figure 1) Only the 810 nm 0.5 J/cm2 group had better viability results, which was about 35% higher than the control and was statistically significant (p = 0.013). There was also a reduced viability for the 940 nm 0.5 J/cm2 group as compared to the control (p = 0.001).
A statistically significant difference existed between the 810 nm 0.5 J/cm2 group and the control; the former showed better viability results (p = 0.013). However, the 940 nm 0.5 J/cm2 irradiation provided lower viability results as compared to the control. The dual wavelength group showed no statistically significant difference as compared to its control.
When comparing the wavelengths with similar energy densities, the 810 nm 0.5 J/cm2 group had a higher viability percentage as compared to the 940 nm laser group and the combined irradiation group (810 nm + 940 nm) with statistically significant differences (p < 0.001). There was a statistically significant difference between the combined irradiation group with the 0.5 J/cm2 irradiation and the 940 nm laser group with the 0.5 J/cm2 irradiation (Figure 1)
Cell proliferation
The cell proliferation amounts were lower for the single wavelength irradiation groups as compared to the controls at 48 and 72 h, but the dual wavelength group results were significantly better than those of their control for the 1.5 J/cm2 and 2.5 J/cm2 energy densities (p < 0.001) (Table 1).
The three-way ANOVA was used to compare the effect of the laser wavelength, energy density and time on proliferation. The results of this test also showed that both laser and time factors affect proliferation (p < 0.05) (Table 2). In addition, the results of the study show that the laser effect depends on energy density and time (p < 0.05). Further two-fold comparisons between the groups were done using Tukey’s test and the results are shown in Figure 2.
When comparing different energy densities of the 810 nm laser, 0.5 J/cm2 showed higher proliferation amounts as compared to 2.5 J/cm2 at 24 h (p = 0.002) and 72 h (p < 0.001). The 810 nm 1.5 J/cm2 laser also provided better results as compared to 2.5 J/cm2 after 72 h (p = 0.038).
There was only 1 statistically significant difference in the 940 nm group, which was between irradiation with the 0.5 J/cm2 and 2.5 J/cm2 energy densities at 72 h, with better results for the 2.5 J/cm2 group (p = 0.038). Differences between the 3 energy densities of the combined irradiation group were statistically significant at all time points, with better results as the energy dose increased (p ≤ 0.001) (Table 3).
We also compared similar energy densities of different laser wavelengths. The 940 nm 2.5 J/cm2 group had statistically significantly higher results as compared to 810 nm 2.5 J/cm2 at 72 h (p < 0.001). The 810 nm 0.5 J/cm2 group showed better proliferation results as compared to 0.5 J/cm2 of the 940 nm laser at 24 h (p < 0.001), but this difference was not significant at the 0.05 level after 48 and 72 h. The 1.5 J/cm2 setting showed no statistical difference between the 2 lasers. The combined irradiation group with similar energy densities as compared to the 810 nm and 940 wavelengths applied individually presented significantly better results, except for comparisons with 810 nm 2.5 J/cm2 at 24 h and 810 nm 1.5 J/cm2 at 72 h. Differences between similar energy densities of the combined treatment and the 940 nm laser were also nonsignificant after 72 h (Table 4).
Propidium iodide staining
The cells were carefully evaluated after staining and no negative effect of laser irradiation on the cell nucleus was observed in any of the groups.
Discussion
Finding the appropriate PBM protocol for cells before clinical trials regarding wound healing is strongly recommended. Cell culture studies provide conditions which make it possible to better assess the effect of laser irradiation on cells by eliminating interventional factors in the clinic. However, differences should also be considered when translating the results of in vitro studies to clinical application.
The results obtained in the present study on gingival fibroblast cells showed that a single dose of 0.5 J/cm2 with the 810 nm laser could slightly increase the viability of gingival fibroblasts measured at 24 h. Other energy densities of this laser and also the 940 nm laser did not show a positive effect on cell viability at 24 h. Differences in the results of the same energy density between different wavelengths might be due to the fact that different wavelengths of the laser may operate through different mechanisms and each wavelength can be unique in terms of the chromophores it can stimulate, and also differences in their penetration depths can affect their results. This has been demonstrated in some studies.19, 20, 21 For example, in a study by Wang et al., different mechanisms of action were reported for 2 infrared wavelengths of 980 nm and 810 nm.20 They were able to demonstrate that the 980 nm wavelength affects temperature-gated calcium ion channels, while the mitochondrial cytochrome c oxidase is the most probable target of the 810 nm laser.20 Also, in a study on the effects of 2 wavelengths of red and near-infrared, Marques et al. observed different effects of laser wavelengths.19 According to their findings, an energy density of 5 J/cm2 applied with the red laser increased cell viability and proliferation, while the same dose of the near-infrared laser led to negative effects.19
Based on these differences, we also evaluated the effect of the combined irradiation of 2 wavelengths on gingival fibroblast viability and proliferation. A synergistic effect of applying 2 different wavelengths has been previously reported in sparse in vitro and in vivo studies.16, 17, 18, 22 We were also able to observe this effect. Based on our findings, the combined irradiation showed different results as compared to the separate application of each wavelength. This combined therapy seemed to have a better effect on proliferation than the lasers applied alone. It was also more effective in comparison with the control.
In a study by Fekrazad et al., the effects of the blue, green, red, and infrared lasers as well as their combination on the proliferation and differentiation of mesenchymal stem cells (MSCs) were assessed.18 They reported that some combinations, such as red + infrared, had negative effects, but the blue + green laser combination was better than each one applied alone. However, the authors stated that they were still not able to reach a clear conclusion as to the effect of the combined irradiation on these cells.18 On the other hand, Zare et al. reported that in the combination treatment with 630 nm + 810 nm LLLT, 3 sessions of irradiation at 2.4 J/cm2 resulted in an increase in the viability of human adipose stem cells (hASCs) as compared to the control cells and PBM-treated bone marrow-derived stem cells.16 However, this effect was not observed with 1 session of this combined treatment at 1.2 J/cm2.16 In the present study, we only evaluated a single session of irradiation and we probably could have obtained different results with multiple sessions, which needs to be investigated in future studies.
Interesting positive effects were reported in a recent in vivo wound healing study for the simultaneous application of the 660 nm and 808 nm lasers as PBM therapy in infected pressure ulcers in mice, where this combined treatment accelerated healing and had antibacterial effects.22 In another study, the combination of 2 wavelengths – 660 nm and 808 nm – at a higher dose of 300 J/cm2 applied twice a week was investigated clinically and was reported as effective treatment for oral mucosistis.17
In seems that the combined wavelength treatment may have many interesting synergistic effects, which need to be evaluated more precisely in further in vitro and in vivo studies with well controlled groups.
There are a number of studies on gingival fibroblasts, but mostly with many different wavelengths and laser parameters, showing a variation of results.2, 23, 24, 25 We have only tried to focus on discussing the results of similar wavelengths with regard to the ones used in the present study to better elucidate their effects on these cells.
Although the results of the current study show a slight positive effect of a single dose of the 810 nm laser in the 0.5 J/cm2 setting at 24 h, no irradiation was sufficient to result in increased proliferation rates during the 72 h of evaluation as compared to the control in the single wavelength groups; however, the dual wavelength results showed significantly better effects on proliferation as compared to the control. Similar to our results, low-level laser (LLL) irradiation with lower energy densities of <2.5 J/cm2 in a recent study by Marques et al. was not able to promote the metabolic activity and proliferation of stem cells with red and near-infrared wavelengths.19 The same authors reported lesser cell proliferation in the 2.5 J/cm2 660 nm red laser group at 48 h based on the sulforhodamine B (SRB) assay, but not in the near-infrared irradiated group.19 In the present study, we observed a reduction in the proliferation of cells, yet only in the 810 nm group at the 48 h time point, which needs to be considered and better elucidated in future studies.
Frozanfar et al. investigated the effect of the low-power diode laser with a wavelength of 810 nm, an output power of 50 mW and an energy density of 4 J/cm2, with 3 consecutive irradiation sessions, on the expression of type I collagen gene and the proliferation of HGF cells.26 Their study revealed that laser irradiation increased the expression of type I collagen gene on the 3rd day and the fibroblast proliferation rate after 48 and 72 h.26 It seems that the lower output power used in that study and the repeated delivery of laser radiation energy might have resulted in a cumulative positive effect, better in comparison with the one of the present study.
In a study by Kreisler et al., the 809 nm diode laser with a very low output power of 10 mW and energy densities of 1.96, 3.92 and 7.84 J/cm2 was used for irradiation 2 and 3 times in 24-hour intervals.5 They reported an increase in the proliferation of gingival fibroblasts after 24 h. However, this increase of the proliferation gradient declined 48 and 72 h later, with a density-dependent pattern.5 The reason for this decrease can be explained by the gradual vanishing effect of the laser or the cells reaching the saturation point after 3 days of incubation. If the latter is the case, we might have achieved a different result by seeding a lower number of cells in each well. Interestingly, in a report by Moore et al., similar results to ours were reported.27 They observed that although the proliferation of fibroblasts increased after a single irradiation session with red wavelengths, with a maximum effect in the case of the 665 nm and 675 nm wavelengths, a single dose of 810 nm low-power diode laser radiation (5 mW/cm, 10 J/cm2) resulted in an inhibitory effect. However, the energy density they utilized was much higher as compared to the ones in the present study and had a much lower output power.27
Wavelengths of around 940 nm have also been studied in a few reports. Similar to the results of the present study, Hakki et al. reported that a single application of high-power 2 W and 1.5 W in the pulsed wave (PW) and low-power 0.3 W in the continuous wave (CW) of the 940 nm laser did not have any significant effects on the proliferation of fibroblasts.28 However, they reported a significant increase in the insulin-like growth factor (IGF), vascular endothelial growth factor (VEGF) and transforming growth factor-β (TGF-β) mRNA expressions in all 3 laser setting groups as compared to the control group. An increase in the type I collagen mRNA expression was also observed, but only in the 0.3 W setting.28
In another study, Pereira et al. evaluated the effect of using the 904 nm laser in 2 sessions of irradiation with a 6-hour interval on cell growth and the synthesis of procollagen in the cultured NIH-3T3 fibroblasts.29 Cell stress was also simulated in this study. The cells were submitted to low-power laser irradiation with an output power of 120 mW and energy densities varying from 3 to 5 J/cm2 over a period of 1–6 days. The results indicated that the 5 J/cm2 irradiation had no effect on cell growth, but an energy density of 3 or 4 J/cm2 caused a remarkable increase in the cell number; however, laser irradiation did not affect the synthesis of procollagen.29 Following this study, Marques et al. performed another study on HGF cells, using the same wavelength, and obtained similar results to ours.30 They irradiated the cells at an output power of 120 mW and an energy density of 3 J/cm2. A decrease in type I collagen was observed in the irradiated group, and also a change in the cytoplasmic matrix of the cells was noted. The irradiated cells became more electron-dense, which is similar to apoptosis. They suggested that laser irradiation has the potential to both stimulate cell division and sometimes result in the apoptosis of cells.30
As it can been seen, PBM studies on gingival fibroblasts still vary regarding the irradiation protocols despite similarities in the cells and wavelengths studied.2 On the other hand, one possible application of LLLT, connected to its potential inhibitory effects, is in the management of conditions with abnormal collagen or extracellular matrix deposition, in cases with the risk of keloid formation or conditions such as hereditary gingival fibromatosis.30 The biological mechanisms of low-power laser radiation are not fully understood yet and more advanced molecular experiments are needed to help better understand laser–cell interactions and the mechanisms of different wavelengths in order to be able to design novel, more effective clinical applications for wound healing.
Conclusions
We have only studied the effect of a single session of laser irradiation with 2 laser devices which are normally used at a high output power in clinical practice. The comparison of the effects of single and multiple irradiation with these wavelengths is recommended in future studies. Also, it would be interesting to compare the effects of laser devices designed for PBM therapy (which can produce very low output powers) and of the devices used in the present study, which are usually used as adjunctive in dental practice. Moreover, we studied the effect of the combined laser irradiation provided on a different day than the lasers applied alone and suggest further investigations on this interesting dual wavelength therapy with the same control samples. According to the results of the present study, although a single irradiation dose of 810 nm 0.5 J/cm2 resulted in a slight positive effect on cell viability at 24 h, no significant stimulatory effect on viability and proliferation was observed for other settings evaluated in this study. When the combination of the 2 wavelengths was used, better results were obtained as compared to the controls, which needs to be further investigated in future studies.