Abstract
Background. Gingival recession (GR) represents a highly common mucogingival condition in the permanent dentition in the general population.
Objectives. The aim of this randomized, split-mouth clinical trial was to assess the 3- and 5-year results of the treatment of multiple GRs through modified coronally advanced tunnel technique (MCAT) and subepithelial connective tissue graft (SCTG) with or without 24% ethylenediaminetetraacetic acid (EDTA) root conditioning.
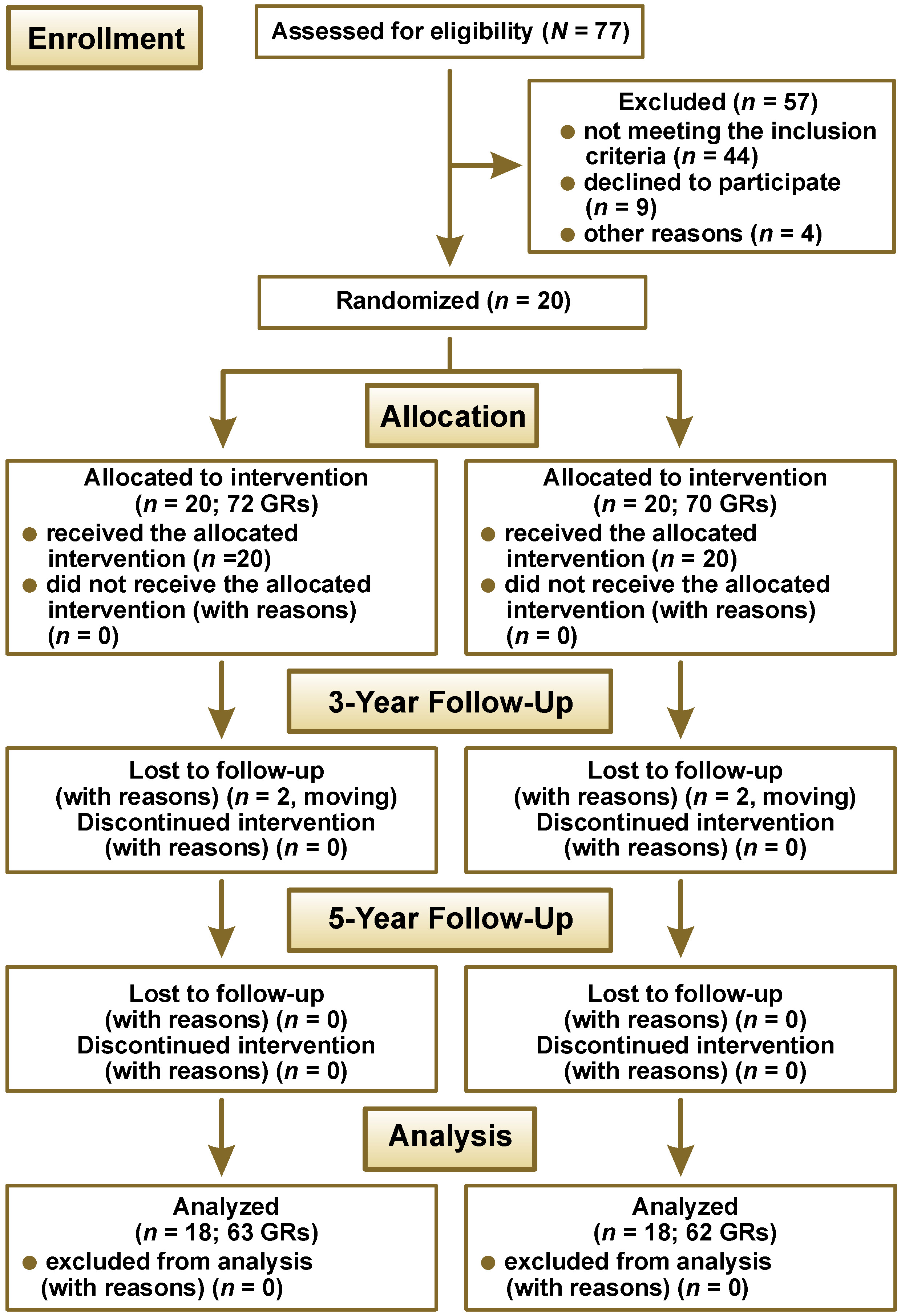
Material and methods. Twenty patients with 142 GRs received both test and control treatment (72 test sites: MCAT+SCTG+EDTA, and 70 control sites: MCAT+SCTG). Two patients were lost to follow-up before the 3-year mark; therefore, a total of 125 GRs were analyzed in 18 subjects. The mean and complete root coverage (MRC, CRC), gain in the clinical attachment level (CAL), gain in keratinized tissue width (KTW), gain in gingival thickness (GT), the root esthetic coverage score (RES), and patient-reported outcome measures (PROMs) were evaluated.
Results. All parameters showed significant improvement in both groups as compared with baseline. There were no statistically significant differences between 3 and 5 years within the groups except for GT, or between the groups at either time point. In the test group, MRC was 95.8% at 3 years and 94.2% at 5 years, with CRC obtained in 87.3% and 84.1% of GRs, respectively. In the control group, MRC was 92.6% at 3 years and 90.6% at 5 years, and CRC was achieved in 82.3% and 80.7% of GRs, respectively. Both groups achieved high RES: 9.0 and 8.9 for the MCAT+SCTG+EDTA and MCAT+SCTG sites after 3 years, and 9.1 and 9.0 after 5 years, respectively. After 5 years, the mean gingival recession height (GRH) decreased from 2.3 ±1.0 mm to 0.2 ±0.5 mm in the test group, and from 2.5 ±1.1 mm to 0.2 ±0.5 mm in the control group.
Conclusions. Root surface conditioning with 24% EDTA did not contribute to the outcomes of MCAT+SCTG – 3 and 5 years postoperatively. Thus, its application in clinical practice is not justified.
Keywords: modified coronally advanced tunnel technique, subepithelial connective tissue graft, ethylenediaminetetraacetic acid (EDTA), multiple gingival recessions, root surface biomodification
Introduction
Gingival recession (GR), defined as the apical displacement of the gingival margin relative to the cementoenamel junction (CEJ), is a common mucogingival condition that may lead to esthetic concerns, dentin hypersensitivity, increased susceptibility to root caries and non-carious cervical lesions (NCCLs), and psychological distress related to the perceived risk of tooth loss. Recent research indicates that approx. ⅔ of the global population experiences GR, and the prevalence of GR increases with age.1, 2 A variety of factors can contribute to the development of GRs, such as improper toothbrushing methods, tobacco use, inadequate oral hygiene, orthodontic therapy, and specific features of gingival morphology.3, 4
Various surgical techniques have been proposed for the management of GRs.5, 6, 7, 8 Among these, the tunnel technique (TUN), first described by Allen,9 has gained notable popularity in recent years.10 By avoiding vertical releasing incisions, preserving papillary integrity and limiting flap extension, the TUN approach enhances vascularization, promotes favorable healing, and minimizes postoperative discomfort.11 Several modifications to the original method have subsequently been introduced to further optimize clinical outcomes.12, 13, 14 Consequently, the modified coronally advanced tunnel technique (MCAT) combined with subepithelial connective tissue graft (SCTG) has been recognized as a highly predictable and effective treatment modality.15, 16, 17 According to a recent systematic review and meta-analysis by Tavelli et al., the MCAT approach achieved a mean root coverage (MRC) of 82.7 ±19.7% for localized recessions and 87.8 ±16.4% for multiple recessions.18
To further improve the outcomes of grafting procedures, root surface biomodification with the use of chemical agents, such as citric acid, phosphoric acid, ethylenediaminetetraacetic acid (EDTA), or tetracycline hydrochloride (TTC), has been proposed.15, 16, 17, 18 These agents enhance root surface decontamination, promote blood clot stabilization and facilitate the reattachment of the periodontal tissues; thereby, their use may potentially improve the clinical outcomes of GR treatment.19, 20 Low-pH agents, such as citric acid (pH ≈ 1), are capable of removing the smear layer, exposing collagen fibers and promoting cellular adhesion; however, highly acidic environment may exert cytotoxic effects on the surrounding tissues, resulting in collagen dissolution, root resorption or ankylosis.21
EDTA, on the other hand, has been shown to cause comparable smear layer removal while maintaining a neutral pH (7.0–7.2), which minimizes the risk of tissue damage.22 Several investigations have demonstrated that the application of 24% EDTA for 2 min results in complete removal of the smear layer, and generates a demineralized root surface with favorable characteristics for cell attachment, migration, differentiation, and proliferation.23, 24, 25 The exposure of intact collagen fibers on the conditioned root surface may also serve as a chemoattractant for periodontal fibroblasts.
Although in vitro studies have indicated the enhanced stability of collagen attachment following EDTA conditioning, its clinical benefit in root coverage procedures remains questionable. Most clinical studies have evaluated outcomes within relatively short observation periods (typically 6–12 months), and the additional effect of EDTA on root coverage predictability appears to be limited.26, 27, 28 Moreover, evidence regarding the adjunctive use of EDTA in combination with MCAT and SCTG for treating multiple GR defects is still scarce.29, 30, 31, 32
According to the findings of a recent systematic review, additional in vivo investigations and randomized clinical trials (RCTs) with adequate sample sizes and longer observation periods are required to more reliably assess the impact of root surface biomodification with 24% EDTA on the clinical outcomes of GR therapy.33 Therefore, the present study aimed to report the 3- and 5-year follow-up results of an RCT conducted in patients presenting with multiple type 1 and type 2 GRs treated with MCAT combined with SCTG, with or without 24% EDTA root conditioning. The objective was to evaluate the long-term stability of the obtained clinical outcomes. The null hypothesis assumed that the MRC achieved at the test sites (MCAT + SCTG + EDTA) would not differ significantly from that observed at the control sites (MCAT + SCTG).
Material and methods
Study design and patient population
The present study represents the 3- and 5-year follow-up of a single-center, double-blind, split-mouth RCT. The research was conducted in accordance with the principles outlined in the Declaration of Helsinki (1964) and its subsequent Tokyo revision (2004). Ethical approval for the study protocol was obtained from the Bioethics Committee of the Medical University of Warsaw, Poland (reference number: KB/208/2017), and the trial was registered at ClinicalTrials.gov (NCT03354104).
A total of 20 systemically healthy participants (11 females and 9 males), aged between 21 and 36 years (mean age: 28.9 ±4.5 years), were enrolled from the patients referred to the Department of Periodontology and Oral Mucosa Diseases, Medical University of Warsaw, Poland, between January 2019 and April 2020. Twenty patients with 142 GRs were treated (72 test sites: MCAT + SCTG + EDTA, and 70 control sites: MCAT + SCTG). Informed written consent was obtained from all subjects prior to participation. Prior to surgical intervention, all the participants received comprehensive oral hygiene instructions until a full-mouth plaque score (FMPS) of <20% was achieved.34 The study design is illustrated in Figure 1.
Inclusion and exclusion criteria
Subjects were considered eligible for inclusion if they met the following criteria:
– the presence of at least 2 adjacent buccal GRs classified as recession type 1 – RT1 (no interproximal attachment loss) or RT2 (interproximal attachment loss less than buccal attachment loss), each with a minimum depth of 1 mm, affecting homologous teeth in the maxillary or mandibular arches35;
– FMPS <20%;
– full-mouth bleeding on probing (FMBoP) <20%36;
– clearly identifiable CEJ; and
– age ≥18 years.
The exclusion criteria were as follows:
– the presence of uncontrolled periodontal disease;
– carious lesions or restorations involving the cervical area;
– systemic conditions compromising wound healing;
– the use of medications known to influence the periodontal tissues;
– smoking;
– pregnancy or lactation.
Sample size calculation
Based on the assumption that the standard deviation (SD) of paired differences would not exceed 30%, a power analysis indicated that a minimum of 18 participants per group was required to achieve 80% power for detecting a true difference of 20% in MRC between the test and control sites.37 To compensate for the potential participant attrition over the follow-up period, a total of 20 subjects were ultimately recruited for the study.
Randomization and allocation concealment
Randomization was conducted using a computer-generated sequence prepared by an independent statistician who was not otherwise involved in the study. Allocation details were concealed in sequentially numbered, opaque, sealed envelopes. Immediately prior to surgery, one envelope was opened to determine the treatment assignment for the right side, which was then disclosed to the operator. The contralateral side was consequently assigned to the alternate treatment protocol. The participants remained blinded to their treatment allocation throughout the study period.
Surgical procedure and postoperative care
All GRs were treated by a single experienced operator (BG), following the protocol described by Sculean et al.17 The right side was always treated first.
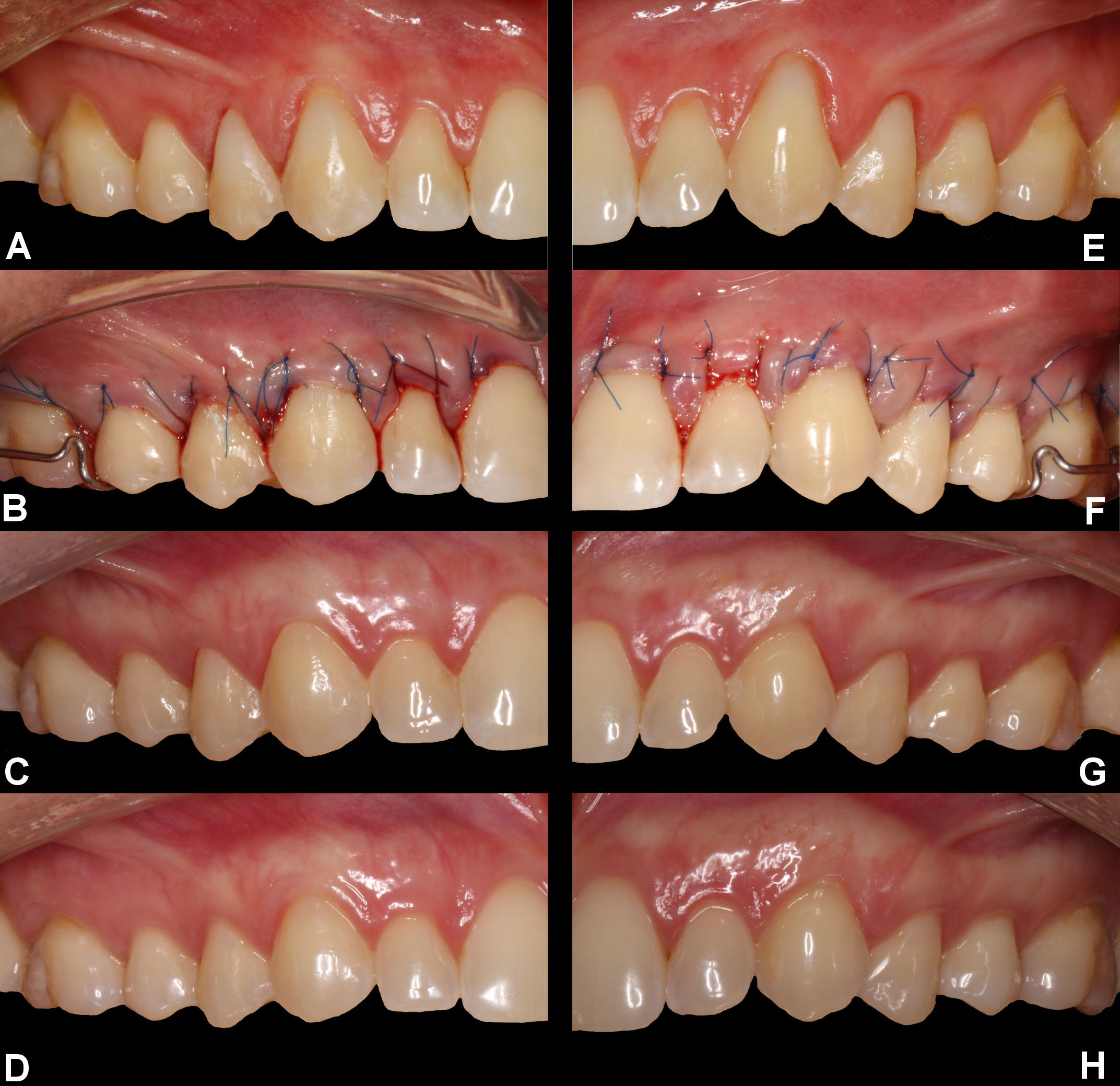
Local anesthesia was administered using 4% articaine hydrochloride with epinephrine (1:100,000) (Ubistesin™ forte 1.7 mL; 3M ESPE, St. Paul, USA). A full-thickness flap was elevated up to the mucogingival junction (MGJ) and continued as a split-thickness flap beyond MGJ. The buccal portions of the papillae were gently raised, preserving the periosteum. The exposed root surfaces were meticulously planed using Gracey curettes (Hu-Friedy, Chicago, USA). A palatal SCTG was harvested through the extraoral de-epithelialization of a free gingival graft, following the technique described by Zucchelli et al.38
At the test site, root surfaces were conditioned with 24% EDTA gel (PrefGel™; Straumann, Basel, Switzerland) for 2 min, followed by rinsing with sterile saline. The SCTG was then positioned beneath the flap and secured at the level of CEJ using sling sutures (PGA Resorba® 6/0, 11 mm, 3/8; Resorba Medical, Nuremberg, Germany). Subsequently, the buccal flap was coronally advanced to completely cover the graft and stabilized with 6/0 non-resorbable monofilament sling sutures (Seralon® 6/0, 12 mm, 3/8; Serag-Wiessner, Naila, Germany).
The control site was treated using an identical protocol, except that root conditioning was performed with sterile saline instead of EDTA. Immediately following surgery, the patients received analgesic medication and were instructed to rinse twice daily with 0.12% chlorhexidine digluconate solution for 3 weeks. Mechanical plaque control (toothbrushing and flossing) and mastication in the surgical areas were prohibited during this period. The sutures were removed 2 weeks postoperatively. Follow-up examinations were scheduled at 1, 3, 6, and 12 months after surgery, and the patients were subsequently recalled for clinical evaluation at 3 and 5 years (Figure 2).
Clinical assessment
Clinical measurements were recorded by a single calibrated examiner (DB) who was blinded to treatment allocation. Calibration was performed twice at a 24-hour interval on 6 non-study patients presenting at least 2 contralateral GRs. Calibration was deemed acceptable when ≥90% of the measurements were reproducible within ±1.0 mm and when exact agreement was achieved in at least 75% of cases.12
Clinical parameters were assessed at baseline, and at 3- and 5-year follow-up visits using a UNC-15 periodontal probe (Hu-Friedy). All measurements were rounded to the nearest 0.5 mm. The following variables were recorded:
– gingival recession height (GRH): the vertical distance from CEJ to the gingival margin;
– recession width (RW): the horizontal distance between the mesial and distal margins of the recession at the CEJ level;
– probing pocket depth (PPD): the distance from the gingival margin to the base of the sulcus;
– clinical attachment level (CAL): the distance from CEJ to the base of the sulcus;
– keratinized tissue width (KTW): the distance from the gingival margin to MGJ.
Gingival thickness (GT) was assessed at the mid-buccal aspect, 3 mm apical to the gingival margin, using an endodontic file with a silicone stopper inserted perpendicularly until contact with the alveolar bone was achieved. The distance between the file tip and the stopper was measured using an electronic caliper with a precision of 0.01 mm (YATO YT-7201; Toya, Wroclaw, Poland).
Outcome measures
The primary outcome variable was MRC.39 Secondary outcome variables included GR reduction, CAL gain, KTW gain, GT gain, and the root coverage esthetic score (RES).40
The RES was assessed according to 5 parameters:
– gingival margin (GM) – scored as 0, 3 or 6 points;
– marginal tissue contour (MTC) – scored as 0 or 1 point;
– soft tissue texture (STT) – scored as 0 or 1 point;
– MGJ alignment – scored as 0 or 1 point; and
– gingival color (GC) – scored as 0 or 1 point.
The individual parameter scores were summed to obtain a total value, with a maximum possible esthetic score of 10 points indicating optimal esthetic integration.
Patient-reported outcome measures (PROMs)
At the 5-year follow-up visit, all the participants completed a questionnaire assessing their subjective perception of esthetics and overall satisfaction with the treatment outcome.41 Responses were recorded using a visual analog scale (VAS) ranging from 0 to 100, where “0” indicated complete dissatisfaction and “100” represented the highest possible satisfaction. In addition, the participants were asked to respond to several dichotomous (yes/no) questions designed to capture specific aspects of their subjective experience and perception of the treatment success.
Statistical analysis
Statistical analysis was carried out using Statistica, v. 13.3 (Statsoft, Krakow, Poland). Data is presented as mean and standard deviation (M ±SD). The normality of distribution for quantitative variables was assessed and confirmed by the Shapiro–Wilk test. Intergroup and intragroup comparisons were made using Student’s t test. The analyzed outcomes were calculated as follows:
(1) MRC3 = (GR0 − GR3) / GR0 × 100%; (2) MRC5 = (GR0 − GR5) / GR0 × 100%; (3) GR3 reduction = GR0 − GR3; (4) GR5 reduction = GR0 − GR5; (5) CAL3 gain = CAL0 − CAL3; (6) CAL5 gain = CAL0 − CAL5; (7) KTW3 gain = KTW3 − KTW0; (8) KTW5 gain = KTW5 − KTW0; (9) GT3 gain = GT3 − GT0; and (10) GT5 gain = GT5 − GT0. Significance was set at p < 0.05.
Results
The study included 20 participants (11 females and 9 males) aged 21–36 years, with a mean age of 28.9 ±4.5 years. In the test group, 72 GRs were treated: 15 incisors; 17 canines; 33 premolars; and 7 molars. Of these, 57 GRs were in the maxilla and 15 in the mandible. In the control group, 70 GRs were treated: 15 incisors; 16 canines; 32 premolars; and 7 molars; 58 GRs were located in the maxillary arch and 12 in the mandibular arch. There were no significant differences in baseline clinical characteristics between the 2 groups (Table 1).
All surgical sites healed uneventfully, and no postoperative complications were recorded. Two participants were lost to follow-up before the 3-year examination. Thus, a total of 125 GRs in 18 patients were evaluated at both the 3- and 5-year follow-up visits. In the test group, 63 GRs (13 incisors, 16 canines, 29 premolars, and 5 molars; 51 in the maxillary arch and 12 in the mandibular arch) were available for analysis. In the control group, 62 GRs (13 incisors, 15 canines, 28 premolars, and 6 molars; 50 in the maxillary arch and 12 in the mandibular arch) were assessed.
Clinical outcomes
All the evaluated clinical parameters – GRH, RW, CAL, KTW, and GT – showed statistically significant improvement from baseline to all follow-up assessments in both groups (Table 2). No significant differences were observed between the 3-year and 5-year outcomes within either group except for GT, where the difference was statistically significant, and no significant intergroup differences were detected at any time point.
In the MCAT + SCTG + EDTA group, MRC reached 95.8 ±11.8% at 3 years and 94.2 ±15.6% at 5 years. Complete root coverage was achieved in 87.3% (55 GRs) and 84.1% (53 GRs) of the treated sites after 3 and 5 years, respectively. In the MCAT + SCTG group, the MRC values were 92.6 ±16.5% at 3 years and 90.6 ±20.2% at 5 years, while CRC was achieved in 82.3% (51 GRs) and 80.7% (50 GRs) of the treated sites at 3 and 5 years, respectively. Consequently, the null hypothesis – that MRC in the test group would not differ from that in the control group – could not be rejected.
Gingival recession reduction for the test and control groups, respectively, was 2.1 ±0.8 mm and 2.3 ±1.1 mm at the 3-year follow-up, and 2.0 ±0.9 mm and 2.2 ±1.1 mm at the 5-year evaluation. Gain in KTW in the test and control groups, respectively, amounted to 0.8 ±1.2 mm and 1.1 ±1.1 mm after 3 years, and 0.8 ±1.2 mm and 1.0 ±1.1 mm after 5 years. Gain in GT for the test and control sides, respectively, was 0.6 ±0.5 mm and 0.6 ±0.6 mm at 3 years, and 0.6 ±0.6 mm and 0.6 ±0.6 mm at 5 years.
Esthetic outcomes
Both groups demonstrated excellent esthetic results, as reflected by high RES values. The mean RES values for the test and control sites, respectively, were 9.0 ±1.1 and 8.9 ±1.3 at the 3-year follow-up, and 9.1 ±1.0 and 9.0 ±1.3 at the 5-year follow-up (Table 3). No statistically significant differences were detected between the 3- and 5-year evaluations within either group, nor between the 2 groups at any time point.
Patient-reported outcome measures (PROMs)
The patient-reported assessments of esthetics and overall satisfaction were comparable between the groups (Table 4). Both treatment modalities – MCAT +SCTG + EDTA and MCAT + SCTG alone – received high subjective ratings for gingival color and contour, and the coverage of GRs. All participants expressed a high degree of satisfaction with the treatment outcome and indicated that they would undergo the procedure again if required. Except for one individual, all patients stated that they would recommend the treatment to others.
Discussion
This split-mouth RCT aimed to evaluate whether root surface conditioning with 24% EDTA influences the long-term clinical and esthetic outcomes of the treatment of multiple Cairo type 1 and type 2 GRs with MCAT combined with SCTG. After 3 and 5 years of follow-up, both test and control sites showed highly predictable and stable results, and no statistically significant differences were detected for any of the investigated parameters. These findings indicate that, under the specific conditions of this protocol, the adjunctive use of 24% EDTA does not confer additional clinically relevant benefit in the treatment of multiple GRs.
Current clinical evidence on the use of EDTA as a root biomodification agent in recession coverage procedures remains limited. Most available trials have investigated coronally advanced flap (CAF)-based techniques with relatively short follow-up periods.42, 43, 44 Across these studies, both EDTA-treated and control sites consistently showed reductions in GR and improvement in MRC; when between-group differences were observed, they were typically modest. In semilunar flap techniques, less favorable outcomes have been reported when EDTA was used, potentially due to incomplete rinsing of the solution or higher technique sensitivity. Collectively, these findings suggest substantial variability and support the notion that flap design and surgical handling may be more critical determinants of the final outcome than the choice of the conditioning agent alone.45 Long-term data and evidence specifically addressing the tunnel or MCAT procedures are still scarce, making the present RCT a relevant contribution to this area.
Recent systematic reviews and meta-analyses have suggested that EDTA may offer modest statistical advantages in terms of GR reduction and CAL gain. However, the magnitude of these differences does not exceed 1 mm, which raises questions about their clinical significance in everyday practice. Thus, although pooled data can reveal statistically significant differences in favor of EDTA, the true clinical impact of such improvement remains uncertain and may be of doubtful relevance from a patient-centered perspective.33
Although long-term data for tunnel-based procedures is still limited, the available evidence supports the notion that MCAT combined with SCTG provides stable and predictable outcomes over extended periods.46 Increased GT and greater KTW have repeatedly been associated with improved long-term stability, and soft-tissue augmentation with SCTG appears to reduce the risk of apical relapse. The present findings align with these observations and suggest that SCTG is likely the primary driver of both root coverage and soft-tissue stability in this protocol.31 Since root coverage is largely responsible for determining RES, the sustained high MRC observed in both groups provides a straightforward explanation for the similarly favorable esthetic scores at 3 and 5 years.
From a biological standpoint, EDTA effectively removes the smear layer, increases root surface wettability and enhances fibrin interaction. In vitro studies consistently show improved fibroblast migration, spreading and attachment on EDTA-conditioned root surfaces, and animal models have demonstrated more favorable connective tissue attachment with reduced epithelial downgrowth.47 While this data provides a strong biological rationale for the use of EDTA, clinical studies in humans have not consistently translated this potential into superior clinical outcomes. This discrepancy suggests that biological plausibility does not necessarily translate into measurable clinical benefit, particularly when the underlying surgical technique already ensures a high degree of predictability.
In the present study, the adjunctive use of EDTA did not result in additional clinical benefit as compared to SCTG alone, despite previous reports suggesting that root biomodification has positive effects. A plausible explanation lies in the minimally invasive nature and high vascularity of MCAT, which already create highly favorable healing conditions and may therefore constrain the potential impact of EDTA. Moreover, although EDTA can improve root surface characteristics in vitro, these changes do not necessarily translate into clinically meaningful or long-term advantages in the context of a stable and well-documented procedure such as SCTG. Consequently, the intrinsic long-term effectiveness of SCTG may overshadow any subtle, short-term effects of EDTA on root coverage outcomes.
The slightly more favorable numerical values recorded in the EDTA group, although not statistically significant, may reflect a modest effect that did not reach the threshold for clinical relevance or statistical detection in the present sample. Therefore, the results should not be interpreted as evidence that EDTA exerts no biological effect, but rather that any additional benefit in this setting appears small, clinically limited, and insufficient to justify its routine use in MCAT + SCTG for multiple GRs. It is possible that EDTA could play a more significant role in other clinical scenarios, such as graftless protocols, thinner periodontal phenotypes, or in combination with bioactive agents, which warrants further investigation.48, 49, 50
Limitations
This study has several limitations. Only Cairo type 1 and type 2 GRs were included, and most sites were located in the maxilla, which restricts the generalizability of the findings. Although the split-mouth design helped minimize inter-individual variability, all procedures were performed by a single experienced operator, which may limit external validity. In addition, no histological evaluation was performed, so conclusions regarding the nature of the attachment must rely solely on clinical parameters. Two patients were lost to follow-up, and, more broadly, long-term data beyond 5 years remains limited in the literature.
Another limitation of this study is the relatively small difference in baseline GRH values between the groups (2.25 mm vs. 2.47 mm) may have reduced the statistical power to detect significant between-group effects, and therefore the results should be interpreted with caution.
In the present study, the suspension of the sutures with orthodontic or composite buttons was not employed at the palatal donor site. Instead, hemostasis and wound protection were achieved by placing a gelatin sponge over the palatal wound, securing it with sutures and using a removable palatal stent. This approach provided gentle compression of the surgical area, limited postoperative bleeding, and protected the wound from mechanical irritation during mastication and speech. However, some studies recommend using the suspension of sutures with orthodontic or composite buttons during the initial healing phase (1–2 weeks).51
Within these limitations, root surface conditioning with 24% EDTA did not yield additional clinical or esthetic benefit in the long-term management of multiple GRs treated with MCAT and SCTG. Future controlled studies should further investigate the role of EDTA in various anatomical conditions, including single recessions, and in the treatment protocols that do not involve SCTG. Ideally, these studies should incorporate histological or surrogate markers to better elucidate the underlying biological mechanisms.
Conclusions
Within the limitations of the present study, it can be concluded that MCAT combined with SCTG yields highly predictable and stable outcomes in treating multiple type 1 and type 2 GRs over both 3- and 5-year follow-up periods. Aadjunctive root surface biomodification with 24% EDTA does not confer any additional benefit in terms of clinical or esthetic outcomes.
Trial registration
The trial was registered at ClinicalTrials.gov (NCT03354104).
Ethics approval and consent to participate
Ethical approval for the study protocol was obtained from the Bioethics Committee of the Medical University of Warsaw, Poland (reference number: KB/208/2017). Informed written consent was obtained from all individual study participants.
Data availability
The datasets supporting the findings of the current study are available from the corresponding author on reasonable request.
Consent for publication
Not applicable.
Use of AI and AI-assisted technologies
Not applicable.