Abstract
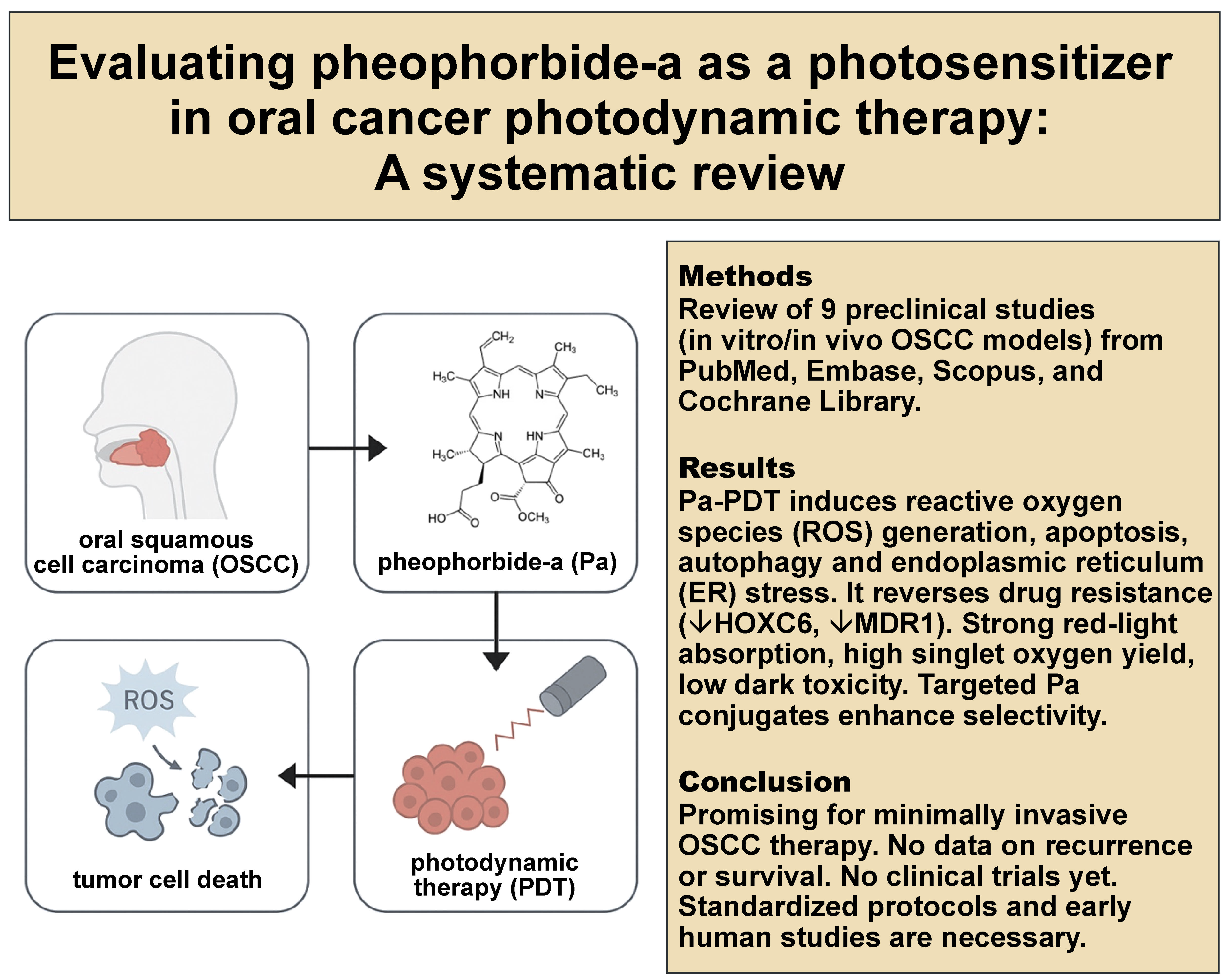
Oral squamous cell carcinoma (OSCC) is commonly treated with surgery, radiotherapy and chemotherapy, which often cause significant morbidity. Photodynamic therapy (PDT) has emerged as a minimally invasive alternative. Pheophorbide-a (Pa), a second-generation photosensitizer, shows promise, but has not been critically reviewed in oral malignancies. The aim of the present systematic review was to evaluate preclinical evidence on the mechanisms of action, efficacy and safety of Pa-mediated PDT (Pa-PDT) in oral oncology.
A systematic search of PubMed, Embase, Scopus, and Cochrane Library was conducted in line with the PRISMA 2020 guidelines (PROSPERO ID: CRD420251066127). Eligible studies included in vitro or in vivo models using Pa-PDT in oral malignancies. Data on therapeutic outcomes, the mechanisms of action, and study quality was extracted and analyzed. Nine studies were included. Pheophorbide-a-induced photodynamic therapy consistently induced apoptosis, autophagy and endoplasmic reticulum (ER) stress through reactive oxygen species (ROS) and mitochondrial dysfunction. It also overcame multidrug resistance by downregulating HOXC6 and MDR1. Conjugation with targeting peptides, nanocarriers and immune checkpoint inhibitors enhanced selectivity and cytotoxicity. Pheophorbide-a demonstrated favorable photophysical properties, including strong red-light absorption (664–675nm), high singlet oxygen yield and minimal dark toxicity. Evidence was limited by small sample sizes, heterogeneous protocols and the absence of human trials.
Pheophorbide-a-induced photodynamic therapy shows antitumor activity in preclinical OSCC models, supporting further preclinical development and carefully designed early-phase trials. Translation should proceed only within early-phase trials that directly benchmark Pa against the approved photosensitizers, using standardized protocols.
Keywords: oral squamous cell carcinoma, oral cancer, photodynamic therapy, photosensitizer, pheophorbide-a
Introduction
Oral squamous cell carcinoma (OSCC) is a major global health burden.1, 2, 3, 4, 5 According to the World Health Organization (WHO), oral cancer encompasses malignant neoplasms arising from the lips and intraoral sites, though some definitions also include oropharyngeal carcinomas.2 A significant proportion of cases develop from oral potentially malignant disorders (OPMDs).6 Despite advances in oncology, standard therapies such as surgery, radiotherapy and systemic chemotherapy continue to dominate OSCC management, yet these interventions are associated with substantial morbidity, including speech and swallowing impairment, cosmetic disfigurement, and a long-term reduction in quality of life (QoL). These challenges underscore the need for minimally invasive and tissue-preserving treatment modalities.6, 7 In parallel with advances in surgery, radiotherapy and systemic therapy, immunotherapy has emerged as an important component of oral cancer management, with immune checkpoint inhibitors improving outcomes in selected patients. The immunomodulatory effects of photodynamic therapy (PDT), including enhanced antigen release and dendritic cell activation, suggest potential synergy with immunotherapy. Pheophorbide-a-induced photodynamic therapy (Pa-PDT) may therefore complement future immuno-oncologic strategies by promoting tumor immunogenicity and augmenting host antitumor responses.5, 6, 7
Photodynamic therapy has emerged as an attractive adjunct in oral oncology, particularly for early-stage or superficial lesions.7, 8, 9 It combines a photosensitizer, targeted light irradiation and tissue oxygen to generate reactive oxygen species (ROS) that selectively trigger tumor cell death while sparing healthy tissues. Several first- and second-generation photosensitizers have been tested in oral cancers; however, each presents limitations in terms of tissue penetration, phototoxicity or systemic clearance.10, 11, 12, 13, 14, 15 Among second-generation candidates, pheophorbide-a (Pa), a chlorophyll-derived tetrapyrrolic macrocycle, has attracted increasing attention.16 Pheophorbide-a exhibits strong absorption in the red spectrum (664–675 nm), high singlet oxygen yield and negligible dark toxicity, making it well-suited for PDT application.16, 17 Distinct absorption peaks at approx. 670 nm (Q-band) and 410 nm (Soret band) have been documented, while its photodynamic activity persists even under hypoxic conditions through radical-mediated mechanisms.18
Preclinical studies have consistently shown that Pa-PDT induces apoptosis, autophagy and endoplasmic reticulum (ER) stress in OSCC models.18, 19, 20, 21 Moreover, Pa-PDT can overcome multidrug resistance by downregulating homeobox C6 (HOXC6) and multidrug resistance 1 gene (MDR1), and recent innovations such as conjugation with tumor-targeting ligands, nanocarrier-based delivery and combination with immune checkpoint inhibitors further enhance its therapeutic efficacy.16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27 Pheophorbide-a has relatively fewer studies in oral oncology than other photosensitizers. This systematic review aims to critically assess the available evidence and position Pa in relation to other agents. Despite promising preclinical findings, the clinical translation of Pa-PDT remains limited, with current evidence constrained by small sample sizes, methodological heterogeneity and the absence of randomized clinical trials (RCTs). Its clinical utility is further underexplored due to the lack of standardized treatment protocols for photosensitizer concentration, incubation time and light delivery parameters, which restricts reproducibility and hinder translation into practice. A systematic evaluation of the existing experimental literature is therefore essential to clarify the therapeutic potential and translational readiness of Pa-PDT. This is the first systematic review dedicated to Pa-PDT in oral malignancies. It synthesizes the available preclinical data on Pa-PDT in oral malignancies, emphasizing its mechanisms of action, efficacy and safety, as well as future directions, and aims to define its prospective role as a minimally invasive adjunct in oral cancer management. Several photosensitizers have already been used clinically in head and neck cancer, including temoporfin used in Europe and chlorin e6 derivatives applied in selected regions, with verteporfin supported by clinical studies.
Given this landscape, the development of Pa requires a clear and differentiated value proposition. Our premise is not that Pa would replace the approved agents, but rather that it could complement them, provided it demonstrates one or more of the following advantages: enhanced tumor selectivity via peptide- or antibody-mediated targeting; improved compatibility with nanocarrier systems for site-specific delivery; or mechanistic synergy that helps overcome treatment resistance. This review therefore positions Pa as a candidate for niche indications, where selectivity, combination strategies or resistance modulation are critical. We do not assert superiority over the approved photosensitizers, but identify the absence of head-to-head comparisons as a key evidence gap.
Material and methods
Focused question
The present systematic review was designed based on the PICO framework, structured to address the following question: In preclinical models of oral malignancies (Population), does pheophorbide-a-mediated photodynamic therapy (Pa-PDT) (Intervention), compared with no treatment, conventional therapies or alternative photosensitizers (Comparison), demonstrate improved antitumor outcomes, such as apoptosis induction, tumor growth suppression and enhanced therapeutic selectivity (Outcome), and therefore, should it be studied further?28
Search strategy
This systematic review, registered with the International Prospective Register of Systematic Reviews – PROSPERO (ID: CRD420251066127), adhered to the PRISMA (Preferred Reporting Items for Systematic reviews and Meta-Analyses) 2020 guidelines to maintain methodological rigor and transparency.29, 30 An extensive search of electronic databases – PubMed/MEDLINE, Embase, Scopus, and Cochrane Library – was undertaken to identify studies investigating the application of Pa-PDT in oral malignancies. Three reviewers independently conducted the database search, using a structured set of keywords tailored to capture studies involving Pa in PDT (Table 1). Inclusion was restricted to English-language publications, irrespective of the publication date. The selection process comprised an initial screening of titles and abstracts, followed by independent full-text assessments by two reviewers based on the predefined eligibility criteria. To ensure comprehensive coverage, the reference lists of the included studies were also reviewed to identify any additional pertinent trials.
Study selection process
To maintain a high standard of methodological accuracy and limit the risk of bias, all retrieved studies were independently reviewed by several evaluators through a structured, multistage screening process. Initial assessments of titles and abstracts were conducted using clearly defined inclusion criteria. Any inconsistencies or disagreement in study selection were addressed through collaborative discussion to ensure reliable and consistent judgments. The inclusion criteria were specifically developed to identify robust studies that investigated the therapeutic potential of Pa-PDT in oral malignancies. This review included experimental studies investigating the antitumor effects of Pa-PDT conducted in vitro or in animal models, relevant to oral oncology. Eligible studies assessed the cytotoxic impact of Pa on oral cancer cells and employed Pa as the primary photosensitizer within the treatment protocol. Research exploring potential synergistic effects with conventional oncologic therapies, as well as studies using controlled designs with appropriate comparators, such as placebo, no treatment, or alternative therapeutic strategies, were considered. Only studies that clearly identified the targeted malignant cell lines and included follow-up assessments to evaluate the durability of the antitumor response and the potential for tumor recurrence were included. Non-peer-reviewed sources, such as conference abstracts, case reports, editorials, opinion articles, book chapters, and unpublished theses, as well as studies lacking scientific rigor or written in languages other than English, were excluded. Duplicate publications or studies derived from the same dataset without novel findings, research unrelated to oral cancer management, and studies without control or comparison groups were also excluded. Additionally, studies that did not involve Pa as the therapeutic photosensitizer, those that used other technologies without directly evaluating Pa, research on non-relevant pathogens, and in vitro experiments conducted under highly artificial conditions lacking clinical relevance were not considered.
Risk of bias in study selection
To promote objectivity and reduce the potential for selection bias, the screening of the titles and abstracts retrieved through the literature search was conducted independently by three reviewers. The level of agreement between the reviewers was quantitatively assessed using Cohen’s kappa (κ) statistic, offering an objective measure of inter-rater reliability.31 Any disagreement regarding study inclusion was addressed through structured deliberation until consensus was achieved. This rigorous and transparent methodology was employed to enhance the credibility of the review process and ensure the accurate identification of relevant papers for inclusion.
Quality assessment
The methodological quality of the included studies was independently evaluated by three reviewers. A structured risk-of-bias assessment tool was employed, using a binary scoring system that assigned 1 point for each criterion met (“yes”) and 0 points for the unmet criteria (“no”), across 9 predefined domains. Each study received a cumulative score out of 9, with the risk of bias classified as high (0–3 points), moderate (4–6 points) or low (7–9 points). Any discrepancies between the reviewers were resolved through discussion and, when necessary, by consultation with a fourth reviewer. This quality assessment followed the methodological guidance outlined in the Cochrane Handbook for Systematic Reviews of Interventions.32 The risk-of-bias assessment results are presented in Table 2. No studies were excluded due to a high risk of bias. Seven of the 9 studies were rated as low-risk. Studies with a moderate risk generally lacked details such as appropriate control groups, transparent statistical analyses, or clear reporting of data and conflicts of interest.
Data extraction
Following agreement on the final selection of studies, three reviewers independently extracted data, using a predefined, standardized protocol designed to ensure accuracy and consistency. For each included study, key information was collected, including the first author, the publication year, study design, the characteristics of the study population, the descriptions of experimental and control groups, follow-up duration (if applicable), and the primary and secondary outcomes relevant to therapeutic endpoints. Focus was placed on the technical parameters of Pa-PDT, such as the photosensitizer concentration, the specifications of the light source (e.g., wavelength, power output, energy density), the method of administration, and any adjunctive therapies. Procedural details, including irradiation duration, the technique and the frequency of application, were also systematically recorded to facilitate robust cross-study comparisons.
Results
Study selection
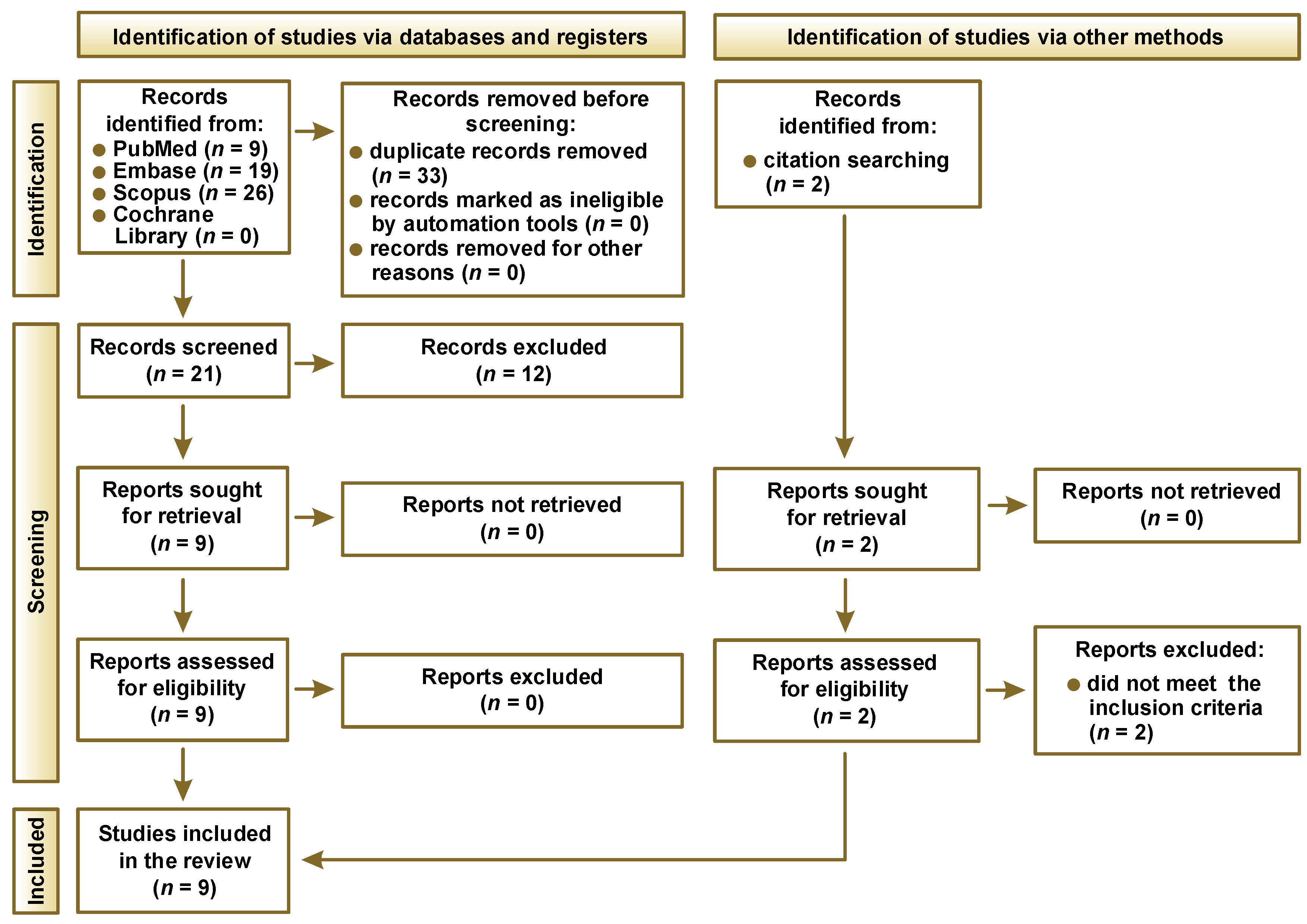
Following the PRISMA 2020 guidelines, the study selection process is summarized in Figure 1. An initial search across multiple databases yielded 54 records. After removing duplicates, 21 unique articles remained for title and abstract screening. Of these, 9 studies were deemed potentially eligible and subjected to full-text review. All 9 met the established inclusion criteria and were incorporated into the final analysis. Of the 9 studies, 5 were in vitro only, 3 combined in vitro and in vivo, and 1 was conducted in vivo. A general overview on the included research is presented in Table 3.
Data presentation
Table 4 provides the key outcomes of the 9 included studies and methodological details, such as study design and treatment groups. Table 5 presents the characteristics of the PDT protocols, including the specifics of the light source (wavelength and energy fluence), as well as the description of the properties of the photosensitizer used – Pa (form of administration, dose/concentration, and incubation time).
Main study outcomes
Ahn et al. demonstrated that Pa-PDT inhibited cell growth and induced apoptosis in AT-84 murine OSCC cells, both in vitro and in vivo.33 The treatment triggered ROS and singlet oxygen generation, mitochondrial membrane depolarization, cytochrome c release, caspase activation, and poly(ADP-ribose) polymerase (PARP) cleavage, with intravenous (i.v.) administration proving more effective than intraperitoneal (i.p.) delivery in suppressing tumor growth.33 In a subsequent study, Ahn et al. showed that Pa-PDT exerted potent antitumor effects in YD10B human OSCC cells by inducing apoptosis through extracellular signal-regulated kinase (ERK) pathway inactivation and promoting autophagy via microtubule-associated protein 1 light chain 3 beta (LC3B-II) upregulation; moreover, the inhibition of autophagy with chloroquine (CQ) enhanced cytotoxicity by increasing necrosis.34 Ahn et al. further evaluated intra-tumoral Pa-PDT in C3H mice bearing AT-84 tumors, confirming significant tumor growth inhibition, increased apoptotic cell death on TUNEL staining, the upregulation of cleaved caspase-3 and PARP cleavage, as well as the feasibility of fluorescence-based Pa detection for simultaneous diagnosis and treatment.35 Kim et al. investigated Pa-PDT in paclitaxel-resistant FaDu-PTX cells and found that the treatment downregulated HOXC6 and MDR1 expression, thereby sensitizing resistant cells to apoptosis; moreover, combined Pa-PDT and HOXC6 siRNA markedly suppressed xenograft tumor growth in vivo.36 Lim et al. employed Pa as a control in head and neck cancer cell lines and confirmed its high singlet oxygen generation capacity (quantum yield 0.52 in alcohol), efficient intracellular uptake, and ability to induce apoptosis and G2/M cell cycle arrest, albeit with slightly lower efficacy than the comparator photosensitizer.37 Moon et al. reported that Pa-PDT induced ROS-dependent cytotoxicity and apoptosis in both immortalized human oral keratinocytes (IHOK) and OSCC cell lines, while identifying runt-related transcription factor 3 (RUNX3) expression as a biomarker correlating with treatment sensitivity.38 Moon et al. further demonstrated that Pa-PDT efficacy could be enhanced through the modulation of miR-145-5p, as its mimic increased ROS generation, apoptosis, and RUNX3 and Bcl-2-like protein 11 (Bim) expression, suggesting microRNA targeting as a potential adjuvant strategy.39 Yoon et al. established that Pa-PDT induced apoptosis and autophagy via ER stress pathways in both in vitro and in vivo OSCC models, with the upregulation of markers such as C/EBP homologous protein (CHOP), phosphorylated protein kinase RNA-like ER kinase (p-PERK) and activating transcription factor 6 (ATF6); the inhibition of ER stress reduced apoptosis, but increased autophagy, indicating pathway-specific therapeutic modulation.40 Finally, You et al. designed Pa conjugates with targeting moieties, such as CRGDLASLC and cyclic RGD peptide (cRGDfK), demonstrating that receptor-mediated uptake enhanced selectivity and photodynamic efficacy, with the CRGDLASLC-Pa conjugate showing the strongest activity against αvβ6-positive FaDu cells.41 Table 4 provides a summary of the extracted information.
Discussion
Comparison of Pa with other photosensitizers
Five of the 9 included studies were conducted in vitro. Such cell culture data does not account for the key factors influencing PDT efficacy in vivo, including stromal barriers, tissue perfusion, immune responses, and intra-tumoral hypoxia. Accordingly, the present conclusions are limited to biological plausibility, and clinical extrapolation should be avoided, with in vivo findings considered preliminary. Overall, these results are hypothesis-generating and warrant validation in prospective human studies.
In comparison with other photosensitizers used in OSCC therapy, Pa demonstrates several advantages that support its candidacy as a second-generation agent.25, 36 First-generation compounds, such as Photofrin®, exhibit suboptimal photophysical properties, including weak absorption above 630 nm, relatively low singlet oxygen yield and prolonged skin photosensitivity, which limit both the therapeutic range and patient acceptability.10 Chlorin e6, one of the most extensively studied second-generation agents, offers improved absorption in the red spectrum and greater ROS generation than Photofrin, but its rapid systemic clearance and relatively low tumor selectivity may reduce sustained therapeutic efficacy.15 In contrast, Pa appears to offer a more balanced profile, although this has not been validated against the approved comparators in oral cancer. It exhibits strong absorption in the red-light spectrum (664–675 nm) and high singlet oxygen yield.36, 40 Pheophorbide-a also demonstrates efficient intracellular uptake in tumor cells, contributing to selective phototoxicity. Although several included studies used fluorescence imaging or receptor-mediated uptake assays to suggest increased intracellular accumulation of Pa in cancer cells, none performed quantitative biodistribution analyses to confirm preferential tumor accumulation in vivo.33, 34, 35, 36, 41
Importantly, Pa-PDT has been shown to overcome multidrug resistance through the downregulation of HOXC6 and MDR1 expression, an effect not consistently reported with other photosensitizers.36 Moreover, its therapeutic efficacy may be enhanced through molecular targeting, nanocarrier-based delivery and combination with immune checkpoint blockade, thereby broadening its translational potential.19, 26, 42, 43 Other photosensitizers, such as hypericin, hypocrellin and toluidine blue, have demonstrated utility in oral oncology; however, Pa distinguishes itself by combining strong photodynamic efficacy with notable mechanistic versatility. It induces apoptosis, autophagy and ER stress, thereby engaging multiple tumor cell death pathways.33, 34, 35, 36, 37, 38, 39, 40, 41
Collectively, these properties suggest that Pa compares favorably with the currently available photosensitizers, although direct, head-to-head clinical trials remain necessary to establish its superiority in OSCC management.10, 11, 12, 13, 14, 15
No included study directly compared Pa with temoporfin, verteporfin or chlorin e6 under matched experimental conditions. Consequently, the strength of evidence supporting comparative claims remains very low, and further controlled head-to-head studies using identical light parameters are required. Additionally, none of the included studies evaluated recurrence, the durability of tumor control or long-term survival following Pa-PDT in animal models. Future preclinical investigations should incorporate extended follow-up and recurrence assessments to determine whether Pa-PDT confers sustained tumor control beyond the acute treatment period.
Results in the context of other evidence
Preclinical evidence indicates that Pa-PDT exerts its antitumor activity in OSCC primarily through ROS generation, leading to mitochondrial dysfunction, membrane depolarization and the activation of caspase-dependent apoptotic pathways.33, 34, 35 In addition, Pa-PDT downregulates HOXC6 and MDR1 expression, thereby resensitizing multidrug-resistant oral cancer cells to therapy.36 Pheophorbide-a also demonstrates favorable photophysical properties, including high phototoxicity and efficient intracellular uptake.37 By comparison, Photofrin, a first-generation photosensitizer, exhibits lower singlet oxygen yield (~0.30) and limited absorption above 630 nm, which restricts tissue penetration.10 Chlorin e6, a widely studied second-generation agent, has comparable singlet oxygen yield (~0.65), but is limited by reduced tumor selectivity and more rapid systemic clearance.15 Thus, although Pa is not uniformly superior across all parameters, its balanced profile offers distinct advantages for OSCC therapy.33, 36, 40
Beyond photophysical properties, several molecular markers modulate Pa-PDT efficacy. RUNX3 has been identified as a biomarker associated with enhanced treatment sensitivity, suggesting potential utility in patient stratification.38 Similarly, the overexpression of microRNA miR-145-5p augments Pa-PDT cytotoxicity by upregulating RUNX3 and Bim, thereby promoting apoptosis in OSCC.39 Pheophorbide-a-induced photodynamic therapy also induces ER stress, contributing to both apoptosis and autophagy; targeted modulation of this pathway may further optimize therapeutic outcomes.40
Therapeutic performance can be further enhanced through molecular targeting strategies. The conjugation of Pa with cancer-targeting peptides, such as CRGDLASLC and cRGDfK, increases tumor selectivity and cellular uptake, resulting in greater cytotoxicity in receptor-positive tumors.41, 42, 43, 44, 45, 46, 47, 48, 49, 50 Likewise, combining Pa-PDT with immune checkpoint inhibitors (e.g., αPD-L1) has been shown to elevate PD-L1 expression, promote dendritic cell activation and enhance cytotoxic T lymphocyte infiltration, effectively converting immunologically “cold” tumors into “hot” ones and prolonging survival in vivo.42
Pheophorbide-a also demonstrates a favorable safety profile. In vitro studies report less than a 10% reduction in cell viability at Pa concentrations up to 2 μM in the absence of light exposure.36, 41 In contrast, Photofrin and chlorin e6 exhibit higher dark toxicity, with viability reductions of approx. 20–25% at comparable concentrations.10 These findings support a broad therapeutic window for Pa, consistent with reports of good biocompatibility, selective cytotoxicity, and minimal adverse effects in dental and oncology contexts.12, 13, 14, 42
Recent advances in nanotechnology further strengthen the potential of Pa-PDT. Fakudze et al. reviewed multiple studies, demonstrating that the incorporation of Pa into nanocarriers improves solubility, stability and tumor selectivity.43 For example, folate-modified poly(lactic-co-glycolic acid) (PLGA) nanoparticles have been shown to increase Pa tumor accumulation in vivo and enhance PDT efficacy.44 The bioresponsive nanoparticles co-delivering Pa and paclitaxel enabled meaningful dose reductions while maintaining therapeutic effectiveness.45 Amphiphilic N-vinylpyrrolidone copolymer nanoparticles achieved strong in vitro phototoxicity with Pa.46 Additionally, protein-based carriers, such as Pa-loaded casein micelles, improved stability, tumor uptake, and in vivo tumor suppression.47, 48, 49, 50, 51, 52, 53 Together, these findings support the development of Pa-based nanocarrier systems to overcome the limitations associated with free Pa formulations and to expand its applicability in oncology.
Recurrent oral cancer often presents with limited treatment options due to prior surgery or radiotherapy. Although current evidence for Pa-PDT remains preclinical, its tissue-preserving nature and capacity to induce apoptosis, autophagy and immunogenic cell death suggest potential value in heavily pretreated settings. At present, Pa-PDT should be regarded as investigational rather than palliative; however, its minimally invasive profile supports its consideration as a plausible candidate for managing superficial or localized recurrences in future clinical trials.42, 43, 44, 45, 46, 47
Limitations of the evidence
The current body of evidence on Pa-PDT in oral malignancies is constrained by several important limitations.
First, most available studies are preclinical, relying on in vitro assays and small animal models. While these provide valuable mechanistic insights, they do not fully replicate the complexity of the human tumor microenvironment, immune interactions or clinical treatment responses. To date, no RCTs in humans have evaluated Pa-PDT, leaving its clinical safety, optimal dosing and long-term efficacy unverified.
Second, methodological heterogeneity across studies limits comparability. Variations in photosensitizer concentration, incubation times, light sources, wavelengths, and energy fluences hinder the establishment of standardized treatment protocols and preclude meaningful meta-analytic synthesis. These inconsistencies raise concerns regarding reproducibility and translational reliability.
Third, most experiments were conducted with small sample sizes and short follow-up periods, restricting conclusions about the durability of response, tumor recurrence and potential late adverse effects. Reporting of toxicity and adverse events was also limited, raising the possibility that safety profiles may be incompletely characterized.
Fourth, few studies included direct head-to-head comparisons with the established photosensitizers, such as Photofrin or chlorin e6, or with conventional treatment modalities. This gap prevents clear positioning of Pa-PDT relative to current therapeutic standards. In addition, a notable proportion of the included studies originate from overlapping research groups, which may limit generalizability and increase the risk of publication bias.
Taken together, these limitations underscore the need for well-designed, independently conducted preclinical studies and, crucially, early-phase clinical trials. At present, optimal Pa-PDT parameters, such as photosensitizer concentration, light dose and incubation time, remain under refinement, and standardization is not yet achievable. Singlet oxygen quantum yield should be benchmarked against other photosensitizers. Although it is a key photophysical metric, its translational impact depends on intracellular localization, light dosimetry and tumor oxygenation, which were variably characterized across the included studies, and should be reported more consistently in future work. Importantly, no included study assessed recurrence, the durability of tumor control or long-term survival after Pa-PDT in animal models. Future investigations should incorporate extended follow-up periods to determine whether Pa-PDT provides sustained tumor control and to better define its potential role in managing recurrent disease.
Fluorescence is central to PDT in head and neck oncology, as it enables margin assessment, treatment monitoring and real-time dosimetry through photobleaching analysis. However, the included Pa studies provided limited and non-standardized fluorescence readouts. Future work should therefore report excitation and emission spectra in relevant biological media, photobleaching kinetics during light delivery, tissue autofluorescence (AF) subtraction methods, and fluorescence-to-drug concentration calibration. Without these parameters, reliable intraoperative guidance and dose optimization cannot be translated into clinical practice.
Limitations of the review process
Although this review adhered to the PRISMA 2020 guidelines and was prospectively registered in PROSPERO to ensure methodological rigor, several limitations should be acknowledged. First, restricting inclusion to English-language publications may have introduced language bias, potentially excluding relevant studies published in other languages. Second, gray literature, such as conference abstracts, dissertations and non-peer-reviewed reports, was excluded, which may have contributed to publication bias by favoring studies reporting positive or significant outcomes. Third, the limited number of eligible studies constrained the breadth of evidence available for synthesis. Although independent screening and consensus discussions were undertaken to reduce selection bias, the inherently subjective nature of quality assessment tools may still have influenced risk-of-bias evaluations. Finally, variability in reporting standards across the included studies limited the consistency of the extracted outcome data and precluded more advanced statistical synthesis, such as meta-analysis.
Implications for practice, policy,
and future research
The findings of this review suggest that Pa-PDT holds considerable promise as a minimally invasive adjunctive therapy for oral malignancies, particularly OSCC. In preclinical models, Pa consistently induced apoptosis, autophagy and ER stress, while also overcoming multidrug resistance through the downregulation of HOXC6 and MDR1.33, 36, 40 Biomarker studies have identified RUNX3 and miR-145-5p as the potential predictors of treatment sensitivity,38, 39 highlighting opportunities for more precise, individualized therapeutic approaches.19, 26, 43 Combination strategies with immune checkpoint inhibitors further broaden the potential role of Pa-PDT within multidisciplinary cancer care.42
From a clinical perspective, these findings indicate that Pa-PDT may serve as an organ-preserving option for early-stage or recurrent oral cancers, particularly in patients who are poor candidates for surgery or radiotherapy. However, the absence of standardized treatment protocols, particularly regarding photosensitizer concentration, light dose and the route of administration, currently limits its immediate clinical applicability.
From a health policy standpoint, prioritizing funding for translational research and early-phase clinical trials will be essential to validate preclinical findings and facilitate clinical adoption. The development of regulatory guidance and treatment frameworks will also be necessary as the evidence base expands.
Future research should focus on several key areas: (1) conducting well-designed phase I/II trials to establish safety, dosing and feasibility in humans; (2) performing direct comparisons between Pa-PDT and the established photosensitizers or conventional therapies; (3) evaluating long-term outcomes, including recurrence, quality of life, and functional preservation; and (4) incorporating biomarker-driven patient selection to optimize therapeutic benefit. Progress in these areas will be critical to determine whether Pa-PDT can transition from an experimental modality to an evidence-based component of oral cancer management.
Clinical relevance remains unproven, as no human safety or efficacy data is currently available for Pa in oral cancer. Accordingly, the existing evidence does not support its use outside the context of clinical trials. For researchers, the most immediate priority is a phase I dose-escalation study incorporating pharmacokinetic profiling, the assessment of tissue Pa concentrations, the evaluation of mucosal photosensitivity, and a predefined light-dose escalation schema. This should be followed by a randomized phase II trial comparing Pa-PDT with an approved photosensitizer in a clearly defined indication, such as superficial recurrent OSCC.
Conclusions
Pheophorbide-a has been investigated less extensively in oral oncology than other photosensitizers; nevertheless, this systematic review aimed to critically appraise the available evidence and position Pa in relation to the established agents. Despite encouraging preclinical findings, the clinical translation of Pa-PDT remains limited, with the current evidence base constrained by small sample sizes, methodological heterogeneity and the lack of RCTs. Its clinical utility is further undermined by the absence of standardized protocols for photosensitizer concentration, incubation time and light delivery parameters, which limits reproducibility and hinders translation into practice. A systematic evaluation of the existing experimental literature is therefore necessary to clarify the therapeutic potential and translational readiness of Pa-PDT.
To our knowledge, this is the first systematic review dedicated to Pa-PDT in oral malignancies, synthesizing the available preclinical data with an emphasis on the mechanisms of action, efficacy, safety, and future research directions, with the aim of defining its prospective role as a minimally invasive adjunct in oral cancer management.
Several photosensitizers are already in clinical use for head and neck cancers, supported by clinical evidence. Within this context, the development of Pa requires a clearly defined value proposition. The rationale is not that Pa should replace the approved agents, but rather that it may complement them by enabling one or more of the following: enhanced tumor selectivity through peptide or antibody targeting; improved compatibility with nanocarriers for site-specific delivery; or mechanistic synergy that may help overcome treatment resistance. Accordingly, this review positions Pa as a candidate for niche indications in which selectivity, combination strategies or resistance modulation are particularly relevant, while identifying the lack of head-to-head comparisons as a key evidence gap rather than asserting superiority over the established photosensitizers.
Ethics approval and consent to participate
Not applicable.
Data availability
All data for this study is included within the article.
Consent for publication
Not applicable.
Use of AI and AI-assisted technologies
AI-assisted tools were used exclusively for language editing and improvement of readability. No AI tools were used for data analysis, interpretation, or generation of scientific conclusions. All content was reviewed and approved by the authors.