Abstract
The objective of the study was to comprehensively synthesize and critically evaluate the effectiveness, safety and clinical applicability of various laser therapies in the management of oral lichen planus (OLP), including photodynamic therapy (PDT), photobiomodulation therapy (PBMT) and high-intensity laser therapy (HILT), and to provide guidelines for clinicians.
A structured literature review was conducted following the PRISMA (Preferred Reporting Items for Systematic reviews and Meta-Analyses) guidelines, and recommendations were formulated. A total of 23 relevant studies published between 2015 and 2025 were identified through searches of multiple biomedical databases. The risk of bias and quality of evidence were assessed, focusing on clinical outcomes such as reduction in lesion severity, pain relief, mucosal healing, and recurrence rates.
Laser therapies demonstrated effectiveness in reducing lesion size and pain, with safety profiles comparable to or superior to those of conventional corticosteroid treatments. Photobiomodulation therapy and HILT showed particularly favorable outcomes, including lower recurrence rates and fewer adverse effects. However, heterogeneity in treatment protocols and laser parameters limited direct comparisons, with corticosteroids remaining the first-line treatment.
Laser-based therapies are valuable adjuncts or alternatives for patients with OLP who are intolerant to corticosteroids. Standardized treatment protocols and further large-scale randomized controlled trials with longer follow-up periods are needed to optimize clinical guidelines and enable personalized therapy. Photobiomodulation therapy is recommended as the safest and most effective modality for erosive OLP.
Keywords: photodynamic therapy, oral lichen planus, low-level laser therapy, high-intensity laser therapy, photobiomodulation
Introduction
Rationale
Although lichen planus may present as a systemic mucocutaneous disorder involving both oral and extraoral sites, the present review focuses specifically on its oral manifestation (oral lichen planus (OLP)), as the majority of available clinical studies do not stratify outcomes according to systemic involvement.1, 2, 3 Oral lichen planus should be interpreted within the broader spectrum of lichen planus as a systemic, immune-mediated disorder that may involve multiple anatomical sites, including the oral mucosa, skin, genital mucosa, and nails. From a clinical and pathophysiological perspective, it is critical to distinguish isolated oral manifestations from multi-organ disease, as these phenotypes may differ in immune activity, chronicity and therapeutic response. These recommendations focus on the topical treatment of oral symptoms.3 The condition is characterized by periods of remission and exacerbation, which lead to fluctuating clinical manifestations and require long-term management and monitoring due to its chronic nature and potential for malignant transformation in some forms.3, 4, 5 The etiology of OLP is multifactorial and complex, involving a dysregulated immune response in genetically predisposed individuals. Oral lichen planus is considered a T-cell-mediated autoimmune disease where cytotoxic CD8+ T lymphocytes trigger apoptosis of basal keratinocytes in the oral epithelium. Lichen planus occurs in cutaneous (skin) and mucosal (organ-specific, e.g., oral) forms. In its etiology, mast cells play a key role by releasing mediators like histamine and cytokines, contributing to the inflammatory infiltrate and basal keratinocyte damage characteristic of the disease. In cutaneous and mucosal lichen planus, laser therapy is effective mainly in the superficial type, where laser radiation targets lesions limited to the epidermis, minimizing penetration into deeper layers. Factors implicated in the pathogenesis of OLP include genetic predispositions, infections such as the hepatitis C virus, psychological stress, certain medications, and dental materials that may act as triggers or exacerbating agents.6, 7 Epigenetic modifications and alterations in the oral microbial environment may also contribute to disease onset and progression. Systemic associations such as autoimmune thyroid disease, diabetes mellitus and hypertension (Grinspan’s syndrome) have also been described. Clinically, 6 types of OLP are distinguished: reticular; papular; plaque-like; atrophic; erosive (ulcerative); and bullous. The reticular form is the most common and is typically asymptomatic, characterized by bilateral white, lace-like Wickham’s striae on the buccal mucosa, tongue and gingiva. The erosive and atrophic forms present with pain, erythema, ulceration, and mucosal atrophy and are associated with a higher risk of malignant transformation and significant morbidity. The plaque-like form resembles leukoplakia, while the bullous type features vesicles or bullae that rupture easily. Each subtype may require different therapeutic approaches tailored to symptom severity and risk profile.7, 8, 9, 10, 11 Laser-based therapies have emerged as an important treatment modality in the management of OLP, especially its erosive and ulcerative types that are refractory to conventional corticosteroid therapy. Various types of lasers, including photobiomodulation therapy (PBMT),12, 13, 14, 15, 16, 17 diode lasers,18, 19 CO2 lasers,19, 20, 21, 22, 23 and neodymium-doped yttrium aluminum garnet (Nd:YAG) lasers,17, 19, 20, 21, 24, 25, 26 provide targeted anti-inflammatory12, 13, 14, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30 and analgesic effects,15, 16, 20, 21, 22, 27, 29 promote mucosal healing13, 14, 29 and reduce lesion size with minimal adverse effects.19, 22, 28, 29 The advantages of laser treatments include precision application, reduced need for systemic immunosuppressants, decreased recurrence rates, and improved patient comfort and quality of life.11, 31, 32 These features make laser therapy a valuable addition or alternative in the management of OLP.
Objectives
The aim of these recommendations and the structured literature review was to systematically synthesize and critically appraise high-quality evidence derived from systematic reviews and meta-analyses evaluating laser-based therapies in the management of OLP. Specifically, the review seeks to assess the clinical effectiveness, safety profile and practical applicability of different laser modalities, including PBMT, photodynamic therapy (PDT) and high-intensity laser therapy (HILT), with respect to pain reduction, improvement in lesion severity, mucosal healing, and recurrence control. In addition, the objective is to identify strengths and limitations within the existing evidence base, with particular emphasis on heterogeneity in laser parameters and treatment protocols. By consolidating current evidence, the review intends to support informed clinical decision-making, contribute to the optimization and standardization of laser treatment strategies, and provide evidence-based recommendations endorsed by the Polish Society for Laser Dentistry, especially for patients who are resistant or intolerant to conventional corticosteroid therapy.32, 33, 34, 35, 36
Material and methods
These recommendations and the structured literature review were conducted in accordance with the Joanna Briggs Institute (JBI) and PRISMA (Preferred Reporting Items for Systematic reviews and Meta-Analyses) 2020 guidelines for umbrella reviews. The study was registered in the PROSPERO database (registration No. CRD420251172714).37, 38, 39, 40, 41
PICO question
The following PICO question was formulated: In patients diagnosed with OLP (Population), do laser-based therapies, including PBMT, PDT and HILT (Intervention), compared with conventional pharmacologic management (such as topical corticosteroids), placebo or sham laser treatment, or other non-laser therapeutic approaches (Comparison), lead to improved clinical outcomes, including reduction in pain, lesion severity, enhancement of mucosal healing, and decreased recurrence rates (Outcome)?42
Search strategy
A detailed and systematic search was performed across major electronic databases, including PubMed®/MEDLINE, Embase, Scopus, and the Cochrane Library, to identify published systematic reviews and meta-analyses examining laser therapy for the prevention or treatment of OLP. Three reviewers conducted the search using a predefined set of Medical Subject Headings (MeSH) terms and free-text keywords related to OLP and laser therapy. The initial search, limited to English-language studies with no publication year restrictions, included all records available up to October 1, 2025. The screening process followed a two-step procedure: first, titles and abstracts were screened, and then full-text articles were assessed by 3 reviewers against predefined inclusion and exclusion criteria. To capture any additional relevant publications, reference lists of all eligible reviews were manually checked. Overall, the search strategy was designed to identify systematic reviews and other high-quality evidence on the efficacy of laser-based interventions in the management of OLP. The final syntax covered literature published between 2015 and 2025 across the selected biomedical databases (Table 1).
Study selection process
To ensure methodological rigor and minimize potential bias, study selection followed a structured and transparent screening process conducted independently by 3 reviewers. The procedure began with an initial assessment of titles and abstracts based on predefined inclusion and exclusion criteria specifically designed for the objectives of these recommendations and the comprehensive literature review. The study focused on systematic reviews and meta-analyses that examined the effectiveness of laser-based therapies and related photobiomodulation approaches in the management of OLP. Eligible studies were required to report clinically relevant outcomes, such as reductions in lesion size, pain intensity, burning sensation, erythema, or improvement in mucosal healing. To ensure data quality, only peer-reviewed articles with a transparent methodology, including control or comparison groups and measurable outcomes, were included. Exclusion criteria comprised non-peer-reviewed publications, opinion pieces (such as narrative reviews or editorials), conference abstracts, and unpublished theses. Studies not written in English, lacking sufficient methodological details, or not directly evaluating laser interventions for OLP were also excluded. Duplicated reports and secondary analyses derived from the same dataset were omitted unless they provided novel insights or additional data.
To ensure objectivity and minimize the potential for selection bias, the initial screening of titles and abstracts was performed independently by 3 reviewers. The level of inter-reviewer agreement was quantified using Cohen’s kappa coefficient, providing an objective measure of consistency across assessments. Any discrepancies in study selection were resolved through structured consensus discussions to maintain transparency and uniformity throughout the review process.40, 41, 42
Data extraction
Essential information collected from each included review comprised the first author’s name, year of publication, review design (systematic review or meta-analysis), clinical context, and demographic characteristics of the patient populations. Extracted data included the type of laser or light source, wavelength, power output, energy density, application technique, duration of exposure, and the number of treatment sessions. When available, additional parameters such as treatment frequency, target tissue site, and whether laser therapy was used as a primary or adjunctive treatment were also documented. Outcomes of interest encompassed both primary and secondary clinical measures, including reduction in lesion size or severity, pain or burning sensation relief, improvement in mucosal healing, and enhancement of patients’ quality of life, where applicable.
Assessment of the risk of bias
The methodological quality of each included study was evaluated independently by 3 reviewers using a structured assessment of the risk of bias. This tool encompassed 9 domains aimed at assessing both methodological transparency and internal validity. The evaluation criteria were as follows:
1. Comprehensive identification and description of the laser system and treatment parameters (e.g., wavelength, power output, energy density, and application technique);
2. Clear definition of treatment protocols, including frequency, duration, and whether the therapy was used as a primary or adjunctive intervention;
3. Specification of clinically relevant outcomes such as improvement in lesion severity, pain reduction or mucosal healing;
4. Inclusion of appropriate control or comparator groups;
5. Transparent inclusion criteria with detailed characterization of the patient population;
6. Consideration of bias control measures, including randomization and blinding where applicable;
7. Appropriateness, transparency and reproducibility of statistical analyses;
8. Clarity and completeness in outcome reporting, including adverse effects and follow-up data;
9. Disclosure of funding sources and potential conflicts of interest.
This risk-of-bias assessment was developed by the authors due to the lack of a standardized scoring system applicable to the synthesis of clinical recommendations.
Results
Study selection
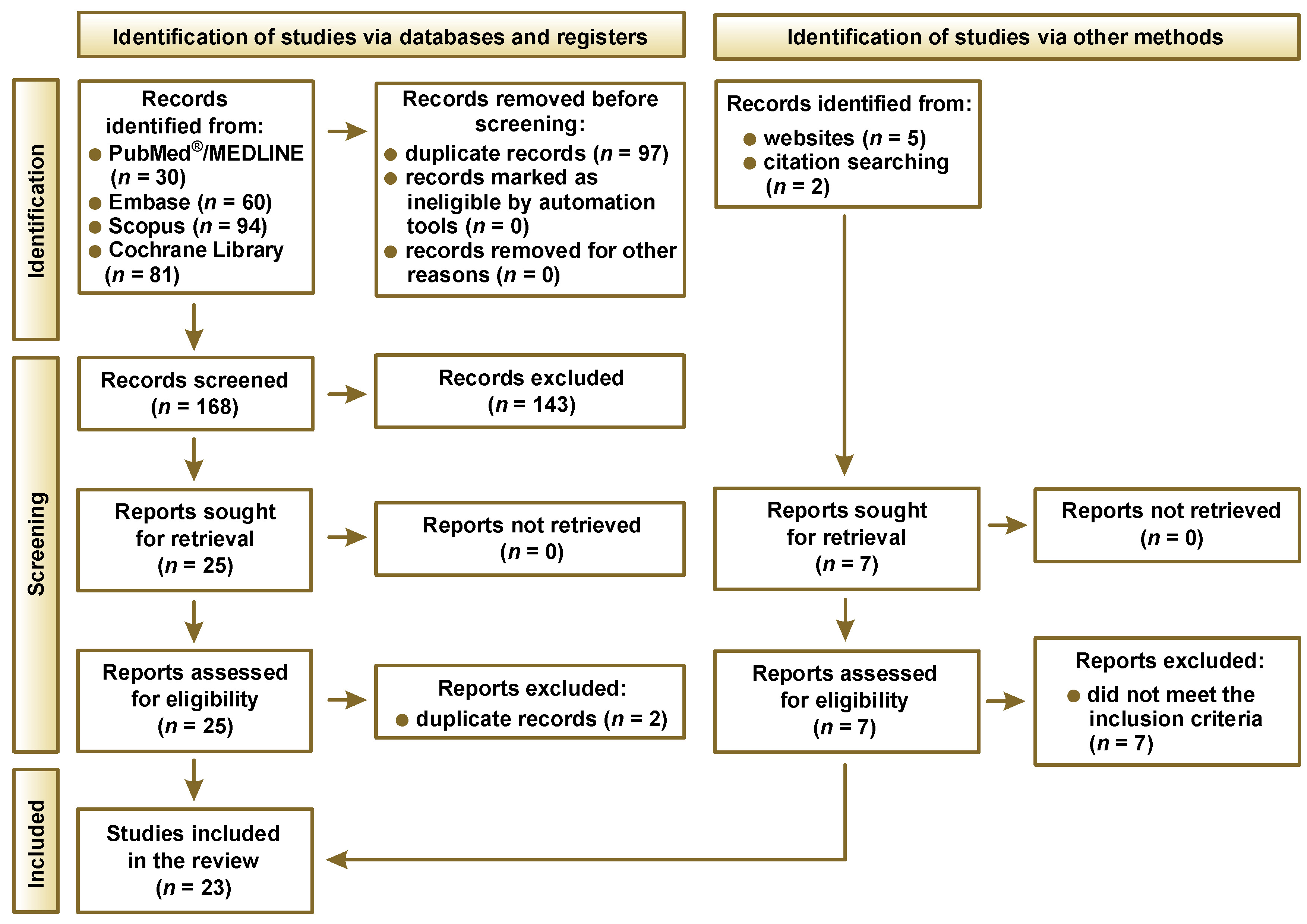
The PRISMA flow diagram outlines the process of study selection (Figure 1). A total of 265 records were initially identified through database searches, including PubMed®/MEDLINE (n = 30), Embase (n = 60), Scopus (n = 94), and the Cochrane Library (n = 81). After removing 97 duplicate records, 168 unique records were screened for eligibility. Of these, 143 were excluded based on title and abstract screening, leaving 25 reports for full-text assessment. Two of these were excluded as duplicates, resulting in 23 studies meeting the inclusion criteria and being included in the final review. Additionally, 7 records were identified through other sources, namely websites (n = 5) and citation search (n = 2). However, all 7 were excluded after full-text evaluation because they did not meet the inclusion criteria.
Risk of bias
As summarized in Table 2, all but one study were judged to be at low risk of bias. Importantly, no studies were excluded solely based on their risk-of-bias rating.
Characteristics of the included reviews
A total of 23 systematic reviews and meta-analyses published between 2017 and 2025 were included in this overview (Table 3). These studies collectively examined the efficacy of various phototherapeutic approaches, such as PDT, PBMT and HILT, in comparison with conventional corticosteroid therapy for the management of OLP and related oral potentially malignant disorders. Early evidence indicated that both PDT and PBMT were effective in alleviating OLP symptoms but not consistently superior to topical corticosteroids.43, 44, 45, 46 Subsequent reviews confirmed that phototherapy improved pain and lesion severity with minimal adverse effects.47, 49, 53, 54, 62, 63 More recent meta-analyses provided stronger evidence that PBMT and HILT significantly reduced pain and produced clinical outcomes comparable or superior to corticosteroids.55, 56, 57, 58, 59 Reviews focused on aminolevulinic acid (ALA)-mediated PDT and symptomatic OLP further supported the effectiveness, safety and tolerability of these light-based treatments.43, 48, 51, 60 Overall, the included reviews consistently demonstrated that phototherapies are effective, safe and minimally invasive alternatives or adjuncts to corticosteroid therapy for managing symptomatic and erosive forms of OLP.
Photodynamic therapy consistently improves clinical scores and symptoms in OLP, though it is generally not superior to corticosteroid treatment in efficacy.43 Studies have shown that PDT is effective and safe. Some papers indicate similar or better outcomes after PDT compared to topical steroids, but heterogeneity in trials limits firm conclusions.46 Photodynamic therapy yields high response rates and is minimally invasive, though protocols vary widely.47 Aminolevulinic acid-mediated PDT specifically shows strong lesion reduction, symptom improvement and low recurrence without adverse effects.48 Multiple studies have confirmed that PDT using various photosensitizers significantly improves OLP symptoms, with ALA-mediated PDT often yielding the best outcomes.49 Meta-analyses reveal that the reductions in pain and lesion size with PDT are comparable to corticosteroid treatment, but limited RCTs and data heterogeneity prevent definitive conclusions.51 Phototherapy combining PDT and PBMT improves symptoms, although many studies have high risk of bias and use diverse laser parameters.52 Photodynamic therapy shows similar effectiveness to corticosteroids, and ALA-mediated PDT is a suitable alternative when steroids are contraindicated.53 According to a network meta-analysis, PDT improves clinical and pain scores to a greater extent than placebo treatment but is inferior to corticosteroids, which remain the most effective modality.62 Methylene blue (MB)-mediated PDT is a safe alternative comparable to corticosteroids, but its superiority is not established.65 Photobiomodulation therapy is generally considered effective and safe, though evidence remains weaker and protocols vary.44, 45, 61 This method has been shown to significantly reduce pain and lesion severity, comparable to the efficacy of corticosteroids, while maintaining a favorable safety profile.50, 58, 60 Photobiomodulation therapy also helps to relieve long-term pain and reduce the likelihood of recurrence; however, larger trials are required to confirm these outcomes.57 Some studies suggest that PBMT is a reasonable alternative to corticosteroids, while the effectiveness of PDT remains less conclusive.54 High-intensity laser therapy shows superior pain relief compared to steroids, with PBMT demonstrating similar efficacy.56 Photobiomodulation therapy is as effective as corticosteroids and is recommended for steroid-resistant cases, though standardization is needed.63 Overall, laser-based therapies are useful adjuncts to corticosteroids, especially in resistant or steroid-contraindicated OLP, but corticosteroids remain the first-line treatment.62
In general, the included evidence indicates that PBMT and PDT provide clinical improvement comparable to topical corticosteroids, while HILT may offer stronger analgesic effects. CO2 laser ablation shows promising long-term outcomes in refractory cases. However, heterogeneity in laser parameters and short follow-up periods limit the strength of the conclusions (Table 4).
Discussion
Rationale behind laser therapy
Topical corticosteroids remain the first-line therapy for symptomatic OLP due to their well-established effectiveness.66, 67, 68 However, recurrence rates after corticosteroid therapy often reach 30–60% within 3–6 months, and long-term remission is infrequent.66, 67, 68, 69 This has prompted interest in laser-based therapies as adjuncts or alternatives, particularly for patients with corticosteroid intolerance, mucosal atrophy, recurrent candidiasis, or refractory disease.70, 71, 72, 73, 74, 75, 76, 77, 78, 79, 80, 81 Across multiple systematic reviews, PBMT and HILT demonstrate analgesic and anti-inflammatory effects comparable to topical corticosteroids, while PBMT offers an excellent safety profile when energy density remains below 120 J/cm2.70, 71, 72, 73, 74, 79, 81 High-intensity laser therapy may provide stronger short-term pain relief but carries a higher risk of thermal injury, emphasizing the importance of operator experience and appropriate parameter selection.70, 79, 81 Photodynamic therapy also achieves clinically meaningful symptom reduction and, in some studies, lower recurrence rates than corticosteroids, although heterogeneity across protocols limits direct comparison.75, 76, 77, 78, 80 CO2 laser vaporization appears beneficial in selected refractory lesions, with some studies reporting markedly reduced recurrence rates and durable symptom resolution.82 Long-term data further supports the efficacy of the CO2 laser. A study reported that 85% of patients remained pain-free after a mean follow-up of 8 years.83 Recurrences were successfully managed by retreatment.83 Nevertheless, ablative approaches require advanced clinical training and careful case selection. Photobiomodulation therapy and HILT have shown durable symptom control and sustained efficacy, with meta-analyses reporting reduced recurrence rates compared to corticosteroids (relative risk for recurrence: 0.43 (PBMT), 0.33 (HILT)) and higher cure rates at 3–6 months.70, 71, 72, 73, 74, 79, 81 For refractory or steroid-resistant OLP, both PDT and PBMT have achieved lasting lesion healing and minimal side effects in case series and clinical trials.80, 84, 85 Given these findings, laser-based therapies are most appropriate for patients who do not respond adequately to corticosteroids or for whom pharmacologic treatment is contraindicated.70, 71, 72, 73, 74, 75, 76, 77, 78, 79, 80, 81 Patient selection criteria for topical corticosteroids include symptomatic, erosive or ulcerative OLP, as these agents are considered first-line therapy for most patients due to their established efficacy and ease of use.66, 67, 68 Treatment choice should consider lesion severity, subtype, comorbidities, prior response to therapy, and practical factors such as equipment availability and clinician expertise.66, 67, 68, 69, 70, 71, 72, 73, 74, 75, 76, 77, 78, 79, 80, 81, 82, 83, 84, 85, 86, 87 For laser-based modalities, contraindications include photosensitivity disorders (especially for PDT), pregnancy (relative) and inability to comply with treatment protocols.70, 71, 75, 79, 86 Laser-based therapies, while non-invasive and generally safe, require specialized equipment (such as diode lasers or PDT kits), operator training and adherence to safety protocols, which can limit availability.70, 71, 73, 75, 79, 86, 87 Photobiomodulation therapy and PDT are particularly advantageous owing to their minimal adverse effects and favorable safety profiles.70, 71, 73, 75, 79, 86, 87 High-intensity laser therapy requires greater expertise due to the higher risk of tissue damage if misapplied.79, 87 Direct comparative and cost-effectiveness data remains limited, but accumulating evidence demonstrates that laser-based therapies (PDT, PBMT, HILT, CO2 laser ablation) offer at least equivalent, and in some studies superior, long-term efficacy and lower recurrence rates compared to topical corticosteroids, with fewer adverse effects and improved patient tolerance.70, 71, 72, 73, 74, 75, 76, 77, 78, 79, 80, 81, 82, 83 These modalities are particularly valuable for patients who cannot tolerate or do not respond to corticosteroids. In summary, topical corticosteroids remain the first-line treatment for symptomatic OLP due to established efficacy, while laser-based therapies represent effective, safe and durable alternatives for selected patients, particularly those with contraindications or refractory disease. Individualized therapy selection should be based on lesion characteristics, prior treatment response, comorbidities, and available resources.66, 67, 68, 69, 70, 71, 72, 73, 74, 75, 76, 77, 78, 79, 80, 81, 82, 83, 84, 85, 86, 87 While corticosteroids remain the standard of care, laser modalities provide safe, effective and adaptable options that complement or extend traditional management strategies (Figure 2).
Secondary infection of oral lichen planus
Secondary infection can exacerbate the inflammatory burden of OLP, intensifying pain and delaying healing.88, 89 Although topical or intralesional corticosteroids remain the standard management, recurrent or refractory cases may benefit from laser-based interventions.88, 89, 90 Photobiomodulation therapy and HILT have shown meaningful symptom reduction in infected or highly inflamed lesions, with PBMT providing predictable clinical improvement and an excellent safety profile at standard parameters. CO2 laser vaporization may also achieve effective lesion removal and reduced recurrence in persistent cases, though its use should be restricted to trained clinicians due to the ablative nature and risk of mucosal injury. Photodynamic therapy likewise offers an alternative for steroid-intolerant patients, improving lesion control with minimal adverse effects when delivered according to established protocols. Overall, laser modalities represent well-tolerated options that may help restore symptom stability and promote healing in OLP complicated by secondary infection, particularly when conventional therapy is insufficient or contraindicated.
Parameters for each laser type
Table 4 reflects diverse laser modalities with typical parameters and clinical outcomes reported in the current literature for OLP management. Photobiomodulation therapy (red diode ~630 nm) is widely supported for safe, effective pain reduction in erosive OLP. Diode lasers with a wavelength of approx. 980 nm show similar positive results. High-intensity laser therapy and CO2 laser ablation provide more aggressive treatment options, often reserved for refractory or severe lesions. Treatment frequency generally spans 3–4 weeks and encompasses multiple sessions tailored to lesion response and patient tolerance. These laser therapies offer promising adjunct or alternative options to conventional corticosteroid treatment with fewer side effects.88
From evidence limitations to clinical decision-making
Although the available evidence supports the clinical effectiveness and safety of laser-based therapies in OLP, several limitations must be considered when translating these findings into routine practice. Substantial heterogeneity in laser parameters, treatment protocols and outcome measures limits direct comparison between modalities and restricts the strength of long-term conclusions. Reporting of clinical outcomes was also inconsistent: while pain reduction was commonly assessed with the visual analogue scale (VAS), lesion size, clinical severity indices, healing dynamics, and recurrence rates were not uniformly measured or standardized. Follow-up periods were short in most studies (4–12 weeks), and the sample sizes were frequently small (often <50 participants), reducing generalizability. This limitation raises uncertainty as to whether reported outcomes reflect sustained disease control or only short-term symptomatic improvement. In this context, laser therapies should be interpreted predominantly as symptom-oriented interventions, unless long-term remission data is demonstrated. In addition, inconsistent reporting of adverse effects and malignant transformation further limits the interpretability and clinical applicability of the available evidence. Most included studies did not stratify patients according to systemic manifestations of lichen planus or associated autoimmune conditions, which may influence treatment response. Another major limitation is the substantial heterogeneity in laser parameters (wavelength, fluence, power density), treatment protocols (session frequency, duration) and outcome measures across studies. This precludes quantitative comparison between modalities and limits the validity of any hierarchy of effectiveness.
Despite these limitations, the consistency of findings across systematic reviews allows for the formulation of cautious, evidence-informed clinical recommendations. Rather than defining a single universal protocol, current evidence supports a stepwise, indication-driven approach, in which laser modalities are selected based on the response to corticosteroids, disease severity, patient tolerance, and safety considerations. This approach enables clinicians to integrate laser therapies into OLP management while acknowledging existing evidence gaps and the need for individualized treatment planning.
These findings indicate that laser-based therapies should be integrated within a structured, stepwise treatment framework rather than used as universal replacements for pharmacologic management. In this context, corticosteroids remain the reference first-line therapy, while laser modalities may be integrated as adjunctive or alternative options in selected clinical scenarios.
Future research should prioritize well-designed randomized controlled trials that directly compare laser modalities with topical corticosteroids using standardized dosimetry and harmonized outcome measures, including pain scores, lesion severity, recurrence at 6–12 months, and patient-reported quality of life. Longer follow-up and cost-effectiveness analyses are essential to refine clinical pathways and support personalized treatment strategies.
Conclusions
This structured review and recommendations demonstrate that laser-based therapies, including PBMT, PDT and HILT, are effective and safe adjuncts or alternatives to conventional corticosteroid treatment for OLP. Across systematic reviews and meta-analyses, these modalities consistently achieved clinically meaningful reductions in pain and lesion severity, with safety profiles comparable to or better than topical corticosteroids. Photobiomodulation therapy showed the most favorable balance of efficacy and safety and was associated with lower recurrence rates, particularly in erosive or steroid-resistant disease. Despite these promising outcomes, substantial heterogeneity in laser parameters, treatment protocols and outcome measures, together with short follow-up periods, limits definitive conclusions regarding long-term efficacy and direct modality comparison. As a result, topical corticosteroids remain the first-line therapy for symptomatic OLP. However, laser-based approaches represent valuable complementary or alternative options for patients with contraindications, intolerance or inadequate response to pharmacologic treatment. Future research should focus on standardized laser protocols, longer follow-up and well-designed randomized controlled trials directly comparing laser modalities with corticosteroids. Such efforts are essential to refine clinical guidelines, support personalized treatment strategies and optimize long-term outcomes for patients with OLP.
Consensus-based clinical guidance from the Polish Society for Laser Dentistry
The Polish Society for Laser Dentistry presents the following evidence-based clinical guidance regarding laser therapy in the management of symptomatic OLP. These recommendations integrate findings from systematic reviews, meta-analyses and clinical trials published between 2015 and 2025 and reflect current best practices in oral medicine and laser dentistry.
General therapeutic principles
Subtype-guided selection of PBMT, PDT or ablative lasers is recommended, as different clinical subtypes of OLP respond differently to laser therapy:
• reticular and papular forms typically do not require laser intervention;
• atrophic and erosive lesions benefit most from PBMT due to its analgesic and anti-inflammatory effects, whereas ulcerative lesions may require extended PBMT or PDT protocols;
• bullous OLP is generally less suitable for laser treatment due to fragile, blistering epithelium and the higher risk of tissue disruption.
Topical corticosteroids remain the first-line treatment for symptomatic, erosive or ulcerative OLP due to their established effectiveness, accessibility and predictable short-term outcomes.
Laser-assisted therapies may be used as adjunctive or alternative treatments in the following clinical scenarios:
• inadequate response to topical corticosteroids (steroid-refractory OLP);
• contraindications or intolerance to corticosteroids (e.g., mucosal atrophy, recurrent candidiasis, hypersensitivity);
• patient preference for non-pharmacologic or minimally invasive interventions.
Additionally, treatment selection should be individualized based on:
• clinical presentation (atrophic, erosive, ulcerative lesions);
• lesion extent and localization;
• comorbidities and contraindications;
• previous therapeutic response;
• availability of equipment and operator experience.
Preferred laser modalities
Photobiomodulation therapy
Photobiomodulation therapy is recommended among laser-based approaches as a first-choice alternative or adjunct. The method is most effective when applied with energy densities below 120 J/cm2 and delivered in multiple sessions (2–3 treatments per week over 2–4 weeks), as supported by recent meta-analyses.
Treatment parameters
The following parameters of PBMT are recommended:
• wavelength: red (630–660 nm) for superficial mucosal lesions or near-infrared (808–980 nm) for deeper inflammatory components or symptomatic erosive lesions;
• power output: 50–120 mW (for contact or near-contact delivery);
• energy density (fluence): 4–8 J/cm2 for mild erythematous lesions or 8–12 J/cm2 for erosive or symptomatic lesions (delivered per point);
• application technique: contact or near-contact; perpendicular orientation to the mucosal surface; point-by-point technique, spacing points by ~1 cm;
• exposure time: typically 20–40 s per point, adjusted to reach the target dose;
• treatment frequency: 2–3 sessions per week for 4–6 weeks or until clinical improvement stabilizes;
• total number of sessions: usually 8–12 sessions for initial therapy, with additional sessions scheduled as needed for symptom recurrence.
Photobiomodulation therapy has demonstrated consistent pain reduction and improvement in lesion severity, particularly in erosive or ulcerative forms of OLP, while maintaining an excellent safety profile with no documented adverse events when energy densities remained below 120 J/cm2. These characteristics support its preferential use as the first-choice laser-based modality in appropriate clinical scenarios.
Photobiomodulation parameters in OLP must be carefully individualized. The selection of dose, wavelength and application technique depends on several factors, including tissue characteristics (such as thickness, sensitivity and vascularity), anatomical location (e.g., lateral tongue vs. palate), severity and morphology of lesions (erythematous, erosive or ulcerative), and the patient’s response during the course of therapy. The anatomical site plays a significant role in protocol adjustment. Lesions of the buccal mucosa and inner lips are typically flat or mildly erythematous, located in well-tolerated tissue where standard PBMT parameters can be applied. In contrast, lesions on the lateral borders or dorsal surface of the tongue require reduced energy densities due to the thin, highly innervated, and more reactive nature of the lingual mucosa. Similarly, lesions on the floor of the mouth and sublingual area – regions characterized by delicate, highly vascular tissues – necessitate conservative dosing and shorter exposure times. Conversely, the palatal mucosa, being thicker and less sensitive, can tolerate higher energy densities. Gingival and marginal periodontal lesions often require moderate parameters because of their sensitivity and variable inflammatory burden.
The severity of the disease further influences PBMT adjustment. Erythematous lesions, representing mild inflammatory activity, generally respond well to lower energy densities aimed at modulating surface inflammation. Erosive lesions of moderate severity are typically symptomatic and demand more robust anti-inflammatory and analgesic dosing. Chronic, ulcerative or extensive lesions, which are often fragile, long-standing and highly reactive, require the most cautious approach, beginning with conservative energy densities and progressively increasing them based on tissue tolerance and clinical response.
Malignant transformation considerations
Oral lichen planus is recognized as an oral potentially malignant disorder, with an estimated malignant transformation rate of 0.5–1.4%. Current evidence does not demonstrate an increased risk of dysplasia or malignant transformation associated with PBMT, PDT, HILT, or CO2 laser ablation. Photobiomodulation therapy operates at non-thermal energy levels that do not induce DNA damage, while PDT selectively targets dysplastic epithelium and may theoretically reduce malignant risk by eliminating atypical keratinocytes.
However, long-term surveillance remains essential, and any OLP lesion, whether treated with corticosteroids or lasers, should undergo periodic clinical follow-up and biopsy when changes in appearance occur.
Current evidence does not indicate an increased risk of malignant transformation related to laser therapy. Photobiomodulation therapy operates at low energy levels that modulate cellular activity without causing DNA-damaging effects, and clinical studies using PBMT for OLP have reported symptomatic improvement without signs of increased oncogenic potential. However, due to limited long-term data, the possibility cannot be completely excluded.
Photodynamic therapy
Photodynamic therapy is recommended as a strong alternative for symptomatic, steroid-intolerant, or steroid-resistant OLP. The method offers favorable safety, low recurrence and minimal systemic effects. ALA- and MB-mediated PDT demonstrate efficacy comparable to topical corticosteroids in many studies. However, patient photosensitivity and adherence to light-avoidance protocols must be considered.
Treatment parameters
Pre-treatment procedures include clinical assessment (documentation of lesion morphology (reticular, erythematous, erosive), pain scoring and photographic evaluation), mucosal preparation, removal of surface debris, and gentle drying of the lesion.
A typical research-grade protocol of the photosensitizer phase using ALA-mediated PDT includes the following elements:
• photosensitizer form: 5-ALA gel or cream (commonly 5% formulations);
• application: a thin, uniform layer over the affected mucosa;
• incubation period: tens of minutes to a few hours, depending on the formulation, allowing conversion of ALA to protoporphyrin IX (PpIX) within epithelial cells;
• occlusion: in some studies, lesions are covered with a light-shielding dressing to prevent premature activation.
A protocol for MB-mediated PDT is as follows:
• photosensitizer form: MB aqueous solution or gargle (commonly 1–5%, often 5%);
• application: direct application via a microbrush;
• pre-irradiation time: a short-defined interval to allow tissue binding (3–10 min);
• excess removal: gentle blotting to prevent pooling.
Additional parameters encompass the following factors:
• light source: approx. 630–660 nm for MB and for activation of PpIX (ALA → PpIX);
• illumination technique: direct, perpendicular exposure to the lesion, fixed distance from the mucosal surface based on device specifications, using uniform scanning or a point-by-point technique;
• energy density (fluence): typical clinical PDT studies report the use of high light doses (many MB studies report ~100–120 J/cm2 per session). Aminolevulinic acid protocols in recent series have used session-based dosing tailored to deliver the desired effect;
• treatment schedule: multiple sessions (weekly or several sessions over 1–3 weeks for MB, or longer series such as 8–10 weekly sessions for ALA).
High-intensity laser therapy
High-intensity laser therapy is recommended for selective use in steroid-resistant cases requiring rapid pain relief. Due to the thermal nature of HILT, inappropriate parameter settings may cause mucosal overheating or tissue damage, making operator experience essential for safe use. This method provides significant analgesic effects and may reduce recurrence risk. However, its higher thermal potential requires experienced operators, careful parameter selection and strict safety monitoring.
CO2 laser ablation
CO2 laser ablation is recommended for localized, severe or refractory lesions. It should only be performed by clinicians with advanced training in ablative laser procedures. The method is effective for long-term symptom control and for reducing recurrence. However, CO2 laser ablation is not intended as a first-line therapy due to the risk of mucosal damage and the need for specialized equipment.
Key conceptual aspects (principles) for HILT or CO2 laser ablation include:
• superficial removal of affected epithelium only (minimal thermal penetration);
• controlled ablation while avoiding deep stromal injury;
• use of a non-contact or near-contact technique, depending on system design;
• use of local anesthesia due to the ablative nature of the procedures;
• protection of adjacent mucosa with shields or barriers;
• the aim to produce a clean, uniform superficial wound bed.
Safety, documentation and procedural standards
The following procedural standards were formulated:
• all laser procedures require appropriate ocular and tissue protection according to device manufacturer guidelines;
• clinicians must document all treatment parameters, including wavelength, power, energy density (fluence), exposure time, spot size, and the number of sessions89;
• PDT requires additional precautions for photosensitive individuals;
• HILT necessitates heightened attention to avoid overheating and collateral tissue injury.
Research and standardization needs
The Polish Society for Laser Dentistry emphasizes the need for:
1. Large, multicenter, long-term randomized controlled trials directly comparing PBMT, PDT, HILT, CO2 laser ablation, and corticosteroids using standardized settings;
2. Uniform outcome measures, including:
• VAS/numeric rating scale (NRS) pain score,
• validated clinical severity indices (e.g., Thongprasom score),
• lesion dimensions,
• recurrence rates at 6 and 12 months,
• patient-reported quality of life (Oral Health Impact Profile-14 (OHIP-14) or equivalent);
3. Cost-effectiveness analyses of laser modalities compared to corticosteroid regimens;
4. Assessment of biological response markers (cytokine modulation, oxidative stress markers, microbiome composition) to aid personalized treatment selection.
Summary of key recommendations
• Corticosteroids remain the first-line therapy for symptomatic OLP.
• PBMT is recommended as the safest and most broadly applicable laser modality.
• PDT is an effective alternative for patients unable or unwilling to use steroids.
• HILT is reserved for experienced operators managing steroid-resistant disease.
• CO2 laser ablation is appropriate for localized, recalcitrant lesions in specialist settings.
• Laser therapies should be used with clear documentation, appropriate safety measures and clinical justification.
Practical clinical protocol for laser use in OLP
Laser therapy may be initiated in symptomatic erosive or ulcerative OLP when (1) symptoms persist despite optimized topical corticosteroid therapy, (2) corticosteroids are contraindicated or (3) the patient prefers a non-pharmacologic alternative. The following approach is recommended:
• PBMT should be the first-choice modality, applied with red (630–660 nm) or near-infrared (808–980, 1,064 nm) wavelengths, using energy densities of 4–12 J/cm2 delivered 2–3 times per week for 2–4 weeks;
• ALA- or MB-mediated PDT may be applied weekly in steroid-intolerant or steroid-refractory cases, with standard illumination at 630–660 nm;
• HILT and CO2 laser ablation should be restricted to refractory lesions and performed only by experienced operators due to the high thermal risk.
All procedures require documentation of wavelength, power, energy density (fluence), exposure time, spot size, and the number of sessions, according to the standards of the Polish Society for Laser Dentistry.
Stepwise practice-oriented clinical recommendations
Based on the synthesized evidence from this structured literature review, laser-based therapies for OLP should be implemented using a structured, stepwise clinical strategy rather than as standalone or empirical interventions.
Step 1 – First-line management
In patients with symptomatic erosive or ulcerative OLP, topical corticosteroids remain the first-line therapy due to their established efficacy and accessibility.
Step 2 – Second-line therapy
The effectiveness and optimal selection of laser modalities may vary depending on disease phenotype. In localized oral disease, PBMT and PDT may provide meaningful symptomatic control. In contrast, in patients with multi-organ involvement or strong autoimmune background, these modalities should be considered adjunctive, as they do not address the underlying systemic immune dysregulation.
Step 3 – Alternative or adjunctive therapy
In patients with steroid intolerance, recurrent disease or partial response to PBMT, PDT, particularly ALA- or MB-mediated protocols, may be used as an alternative or adjunctive modality, provided that appropriate photosensitivity precautions are observed.
Step 4 – Refractory disease management
High-intensity laser therapy may be considered in selected steroid-resistant cases requiring rapid analgesia but should be restricted to experienced operators due to its higher thermal risk profile.
Step 5 – Localized refractory lesions
CO2 laser ablation should be reserved for localized, severe or treatment-refractory lesions and should not be used as a first-line approach.
Step 6 – Surveillance
Regardless of the therapeutic modality used, all patients with OLP require regular clinical follow-up, with vigilance for lesion progression and potential malignant transformation.
Trial registration
The study was registered in PROSPERO (registration No. CRD420251172714).
Ethics approval and consent to participate
Not applicable.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Consent for publication
Not applicable.
Use of AI and AI-assisted technologies
AI-assisted tools were used exclusively for language editing and improvement of readability. No AI tools were used for data analysis, interpretation or generation of scientific conclusions. All content was reviewed and approved by the authors.