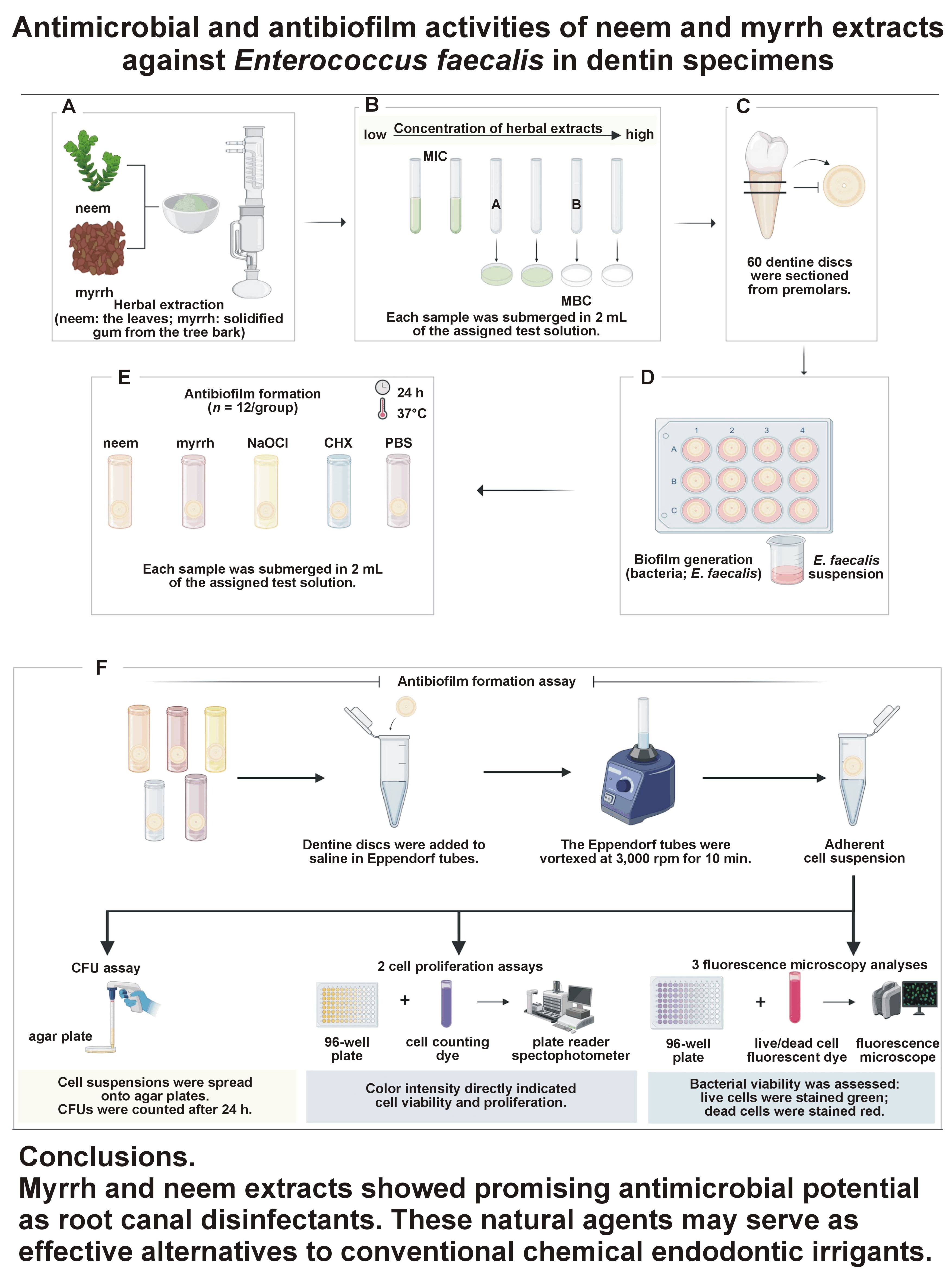
Abstract
Background. Enterococcus faecalis is a primary oral pathogen responsible for root canal treatment failures. The clinical use of conventional root canal disinfectants may be associated with cytotoxicity and other unfavorable effects.
Objectives. The aim of the study was to assess and compare the antimicrobial effect of Azadirachta indica (neem) and Commiphora molmol (myrrh) extracts against E. faecalis biofilm in dentin samples.
Material and methods. The minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) of neem and myrrh extracts against E. faecalis (ATCC 29212) were determined. Human dentin discs were prepared and inoculated with E. faecalis for 21 days. The antibiofilm activities of neem and myrrh extracts were compared using colony forming unit (CFU) assays, cell proliferation assays and live/dead fluorescence microscopy. A 5.25% sodium hypochlorite (NaOCl) solution and 2% chlorhexidine (CHX) were used as standard disinfectants.
Results. The MIC values for the neem and myrrh extracts were 0.0312 mg/mL and 0.0156 mg/mL, respectively, while the MBC values were 0.0625 mg/mL and 0.0312 mg/mL, respectively. The CFU assay revealed a significant decrease in bacterial counts in the neem and myrrh groups. Similarly, cell counting kit-8 (CCK-8) assays demonstrated decreased bacterial viability in the neem and myrrh groups compared with the phosphate-buffered saline (PBS) group. Furthermore, live/dead fluorescence microscopy confirmed the antibacterial effects of both extracts against E. faecalis.
Conclusions. Myrrh and neem extracts showed promising antimicrobial potential as root canal disinfectants. These natural agents may serve as effective alternatives to conventional chemical endodontic irrigants.
Keywords: neem, root canal, Enterococcus faecalis, myrrh, dentin
Introduction
Periradicular and pulpal diseases are mainly caused by microorganisms and their byproducts.1 Previous studies have documented the complex microbial ecosystem of the human dental pulp. A diverse range of microorganisms can inhabit dental pulp, particularly when it becomes necrotic, including gram-positive bacteria such as Enterococcus faecalis, facultative and obligate anaerobic bacteria such as Prevotella, Porphyromonas, Fusobacterium, and Peptostreptococcus, as well as Candida species. A study by Lana et al. reported that obligate and facultative anaerobes, microaerophilic bacteria and yeasts can be isolated from necrotic dental pulps.2 The isolated microbes included Prevotella, Fusobacterium, Lactobacillus, Streptococcus, Clostridium, and Peptostreptococcus among bacteria, as well as Candida and Saccharomyces among yeasts.2 Accordingly, for a successful root canal treatment, it is necessary to eradicate any microorganisms that may cause infection through disinfection, complete obturation of root canals, and prevention of reinfection.3
Enterococcus faecalis is the primary pathogen isolated from failed root canal-treated teeth.4 It has been identified in root-filled teeth with persistent apical periodontitis.5 Sadat Miri et al. reported prevalence rates of apical periodontitis of 51.9%, 40.98% and 38.28% based on the number of individuals, teeth and root-filled teeth, respectively.6 Wang et al. found that the prevalence of E. faecalis in root canals and saliva was 38% and 19%, respectively.7 Studies have suggested that extracellular vesicles of E. faecalis may contribute to the development of chronic apical periodontitis.8 Furthermore, this opportunistic pathogen can develop resistance to antibiotics and possesses multiple virulence factors that compromise immune cell function and their ability to eradicate infections.9 Dai et al. reported that E. faecalis may induce macrophage necroptosis, leading to refractory apical periodontitis.10 Gaeta et al. suggested that saliva may be a major source of E. faecalis infection.11 In addition, this microorganism can survive under harsh conditions because of its ability to form biofilms, making it more resistant to antimicrobial agents, phagocytosis and antibodies.12 Therefore, adequate mechanical root canal preparation by instrumentation and effective chemical irrigation in addition to intracanal medications between treatment sessions are fundamental for successful root canal treatment.13
Root canal disinfectants play a fundamental role in eliminating root canal infections. Sodium hypochlorite (NaOCl) and chlorhexidine (CHX) are the most commonly used irrigants in clinical practice. Sodium hypochlorite at a concentration of 5.25% exhibits broad-spectrum antimicrobial activity against diverse types of bacteria and effectively dissolves necrotic pulpal tissue. However, it is cytotoxic to vital tissues and has an unpleasant odor and taste.14 Chlorhexidine (2%) demonstrates antimicrobial potential against gram-negative and gram-positive bacteria.15 Unlike NaOCl, CHX lacks tissue-dissolving properties. Moreover, the interaction between CHX and NaOCl produces a carcinogenic precipitate known as parachloroanaline (PCA), which may coat the root canal surface, occlude dentinal tubules and compromise root canal sealing.16 Therefore, there is a need for effective, biocompatible and non-toxic root canal disinfectants.
Many in vitro studies have examined plant extracts as potential root canal disinfectants against E. faecalis. Lakshmanan et al. demonstrated strong antibacterial activity of white tea-mediated silver nanoparticles compared with standard irrigants.17 Arévalo-Híjar et al. reported the antibacterial effect of neem (Azadirachta indica) and Moringa oleifera extracts after 24 h and 48 h.18 The antimicrobial efficacy of neem was further confirmed in comparative studies with NaOCl and CHX.19 Similarly, myrrh (Commiphora molmol) extract has shown antibacterial activity as a root canal irrigant.12, 20 Anand et al. reported that the antimicrobial effect of myrrh was comparable to that of 2% CHX, followed by neem, liquorice and saline, using real-time polymerase chain reaction (PCR).21
Given that plant-derived products represent natural and non-toxic sources of antibacterial agents, neem and myrrh extracts are promising candidates for root canal disinfection. Owing to their therapeutic potential in endodontics, the present study aimed to evaluate and compare the antimicrobial effects of neem and myrrh extracts against E. faecalis, using CHX and NaOCl as controls.
Material and methods
Herbal extraction
Neem (A. indica) and myrrh (C. molmol) were obtained from local markets. Neem leaves22 and the solidified gum from myrrh tree bark23 were used for extract preparation. All samples were milled into powder using a Series 2000 standard starter (Brook Crompton, Huddersfield, UK). A total of 20 g of neem powder and 25 g of myrrh powder were weighed and defatted separately with 200 mL of ether using maceration. The defatted powders were extracted with ethanol using the Soxhlet method for 6 h. The resulting extracts were concentrated with the use of a rotary evaporator (Buchi R-215 Rotavapor System; Marshall Scientific, Hampton, USA) and stored in airtight glass containers at 5–10°C for further analysis. The extraction yield [%] was calculated by dividing the weight of dried extract by the weight of the drug sample and multiplying by 100. The extraction yields are provided in Table 1.24
Broth microdilution assay
The minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) of neem and myrrh extracts against a reference strain of E. faecalis (ATCC 29212) were determined using the standard broth microdilution method according to the Clinical and Laboratory Standards Institute (CLSI) guidelines, with minor modifications.25 Isolated colonies of E. faecalis (ATCC 29212) were inoculated into sterile brain heart infusion (BHI) broth at 37°C for 24 h. The bacterial suspension was adjusted to 0.5 McFarland standard, which is equivalent to 1.5 × 108 cells/mL (DensiCHEK™ Plus; bioMérieux, Marcy-l’Étoile, France). Two-fold dilutions of the extracts (0.25, 0.125, 0.0625, 0.0312, 0.0156, 0.0078, and 0.0039 mg/mL) were prepared in phosphate-buffered saline (PBS) and added to 96-well microplates containing the bacterial suspension. Plates were incubated at 37°C for 24 h. Positive control wells contained E. faecalis with PBS, while negative controls contained sterile PBS only. In the present study, the MIC90 was defined as the lowest concentration producing ≥90% growth inhibition based on turbidity. All tests were performed in triplicate. For MBC determination, 25 mL from each well was plated on blood agar and incubated at 37°C for 24 h. The minimum bactericidal concentration was defined as the lowest concentration showing no visible bacterial growth.26, 27
Ethical considerations
The present in vitro study was conducted in accordance with the Declaration of Helsinki. The research protocol was approved by the Institutional Review Board at Imam Abdulrahman Bin Faisal University, Dammam, Saudi Arabia, on May 29, 2022 (approval No. IRB-2022-02-210).
Specimen preparation
Sixty dentin discs were obtained from extracted human single-rooted premolars. Teeth were morphologically intact, and free from caries, restorations, fractures, curvature, or anatomical irregularities. The coronal and apical thirds were removed, and the middle third was sectioned using a precision saw (Isomet® 5000 Linear Precision Saw; Buehler, Lake Bluff, USA) at 4,500 rpm under cooling water, producing discs with a thickness of 1 mm. The discs were rinsed with sterile saline, disinfected with 70% isopropyl alcohol, and sterilized using an autoclave (3545EP; tuttnauer BRINKMANN, Breda, the Netherlands) to ensure complete sterility. The specimens were randomly assigned to 5 groups (n = 12/group): neem (0.0312 mg/mL); myrrh (0.0156 mg/mL); NaOCl (5.25%); CHX (2%); and sterile PBS. The sample size of the study was calculated using a power/sample size calculator (https://www.stat.ubc.ca/~rollin/stats/ssize), with 80% power and a level of significance of 0.05. The mean (M) and standard deviation (SD) values were determined based on a previously published study.28
Bacterial biofilm formation
The sterilized discs were inoculated with 3 mL of E. faecalis suspension (0.5 McFarland) and placed in sterilized 12-well tissue culture plates at 37°C for 21 days for biofilm formation.29, 30 During the inoculation period, the BHI broth was replaced every 72 h to remove dead cells and maintain bacterial viability.31 Positive and negative control wells were included to verify sterility. These wells contained randomly selected extra dentine discs immersed in 3 mL of BHI, and others contained 3 mL of BHI alone. After 21 days, the control samples were examined to verify the sterility using a colony forming unit (CFU) assay. After incubation, specimens were rinsed with sterile PBS for 1 min to remove non-adherent planktonic bacteria.32
Antibiofilm assay
Each sample was immersed in 2 mL of the assigned test solution and incubated at 37°C for 24 h.33, 34 Following this, the samples were rinsed with saline and transferred to 1.5-mL Eppendorf tubes containing 1 mL of sterile saline. The tubes were vortexed (MixMate®; eppendorf, Hamburg, Germany) at 3,000 rpm for 10 min to prepare for the assessment of antimicrobial activities. This evaluation was conducted using CFU assays, cell proliferation assays and fluorescence microscopy.35
Colony forming unit assay
A volume of 100 μL of each suspension was spread onto blood agar plates using sterile cotton applicators. The plates were incubated at 37°C for 24 h. The CFUs were counted in quadruplicate using the ImageJ 1.37b software (National Institutes of Health, Bethesda, USA).
Cell proliferation assay
Aliquots of 100 μL from each suspension were transferred into 96-well plates, followed by the addition of 4 μL of cell counting dye (CCK-8/WST-8, ab228554; Abcam, Waltham, USA). The plates were incubated at 37°C in the dark overnight. After incubation, a soluble orange formazan product was formed by cellular dehydrogenases. In the WST-8 kit, the reduction of salt by metabolically active cells resulted in the formation of an orange formazan product, which could be quantified by plate reader spectrophotometry. Color intensity indicated viability and proliferation of cells. The absorbance of the cell suspension was measured at 490 nm using a microplate reader. Live cells were directly correlated with formazan formation.36 The test was conducted in quadruplicate.
Fluorescence microscopy
Bacterial viability was assessed using the LIVE/DEAD™ BacLight™ Bacterial Viability Kit (L7007; Thermo Fisher Scientific, Waltham, USA). A volume of 100 μL from each suspension was mixed with 4 µL of live/dead cell fluorescent dye in 96-well plates. The test was conducted in quadruplicate. Subsequently, the 96-well plates were incubated at 37°C in the dark for 24 h and examined with the use of an inverted fluorescence microscope (ECLIPSE Ts2R; Nikon Instruments Inc., Melville, USA). The images were taken at excitation/emission wavelengths of 488/530 nm and 365/435 nm. Dead bacterial cells with compromised cell membranes were stained in red, whereas the live membrane-intact cells were stained in green.37 The images of the fluorescence microscopy were quantified (4 green filtered images and 4 red filtered images per group) and analyzed using the color histogram feature in the ImageJ software. The percentage area occupied by each color was calculated.
Statistical analysis
The data was analyzed using the GraphPad Prism v. 5 software (GraphPad Software Inc., La Jolla, USA) and presented as M ±SD. The normality of the data was evaluated using the Kolmogorov–Smirnov test. The statistical analysis was conducted using one-way analysis of variance (ANOVA) followed by Tukey’s multiple comparison test. A p-value <0.05 was considered statistically significant.
Results
The broth microdilution assay revealed that the MIC of neem and myrrh extracts was 0.0312 mg/mL and 0.0156 mg/mL, respectively, while the MBC was 0.0625 mg/mL and 0.0312 mg/mL, respectively (Table 2).
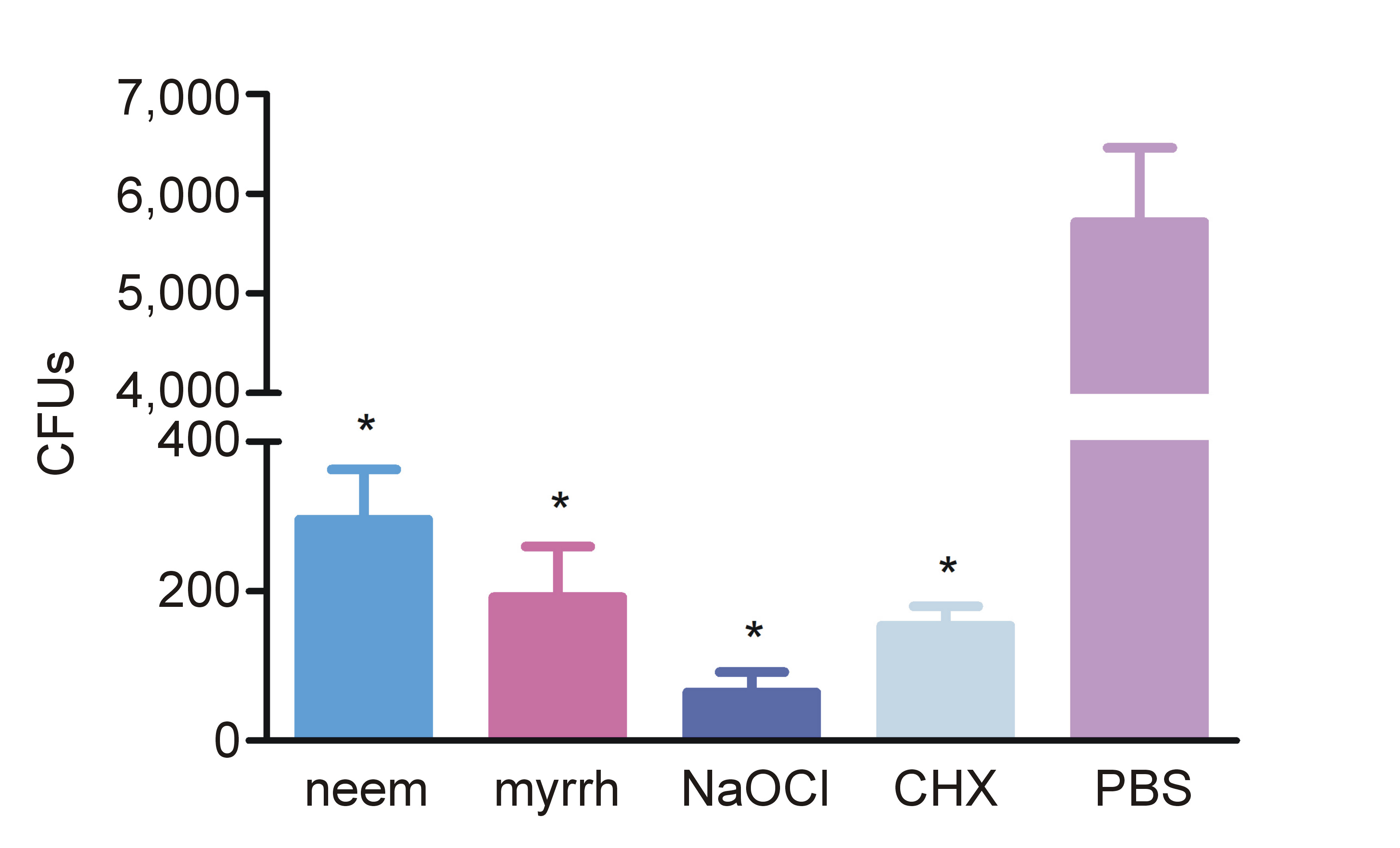
The analysis of the antibiofilm activity revealed that the mean CFU values in the neem and myrrh groups were significantly lower than those in the PBS (control) group (p < 0.001) (Figure 1). Similarly, the NaOCl and CHX groups exhibited significantly reduced CFU values in comparison to the PBS group (p < 0.001). Although the neem group demonstrated higher cfu values than the myrrh group, the difference was not statistically significant.
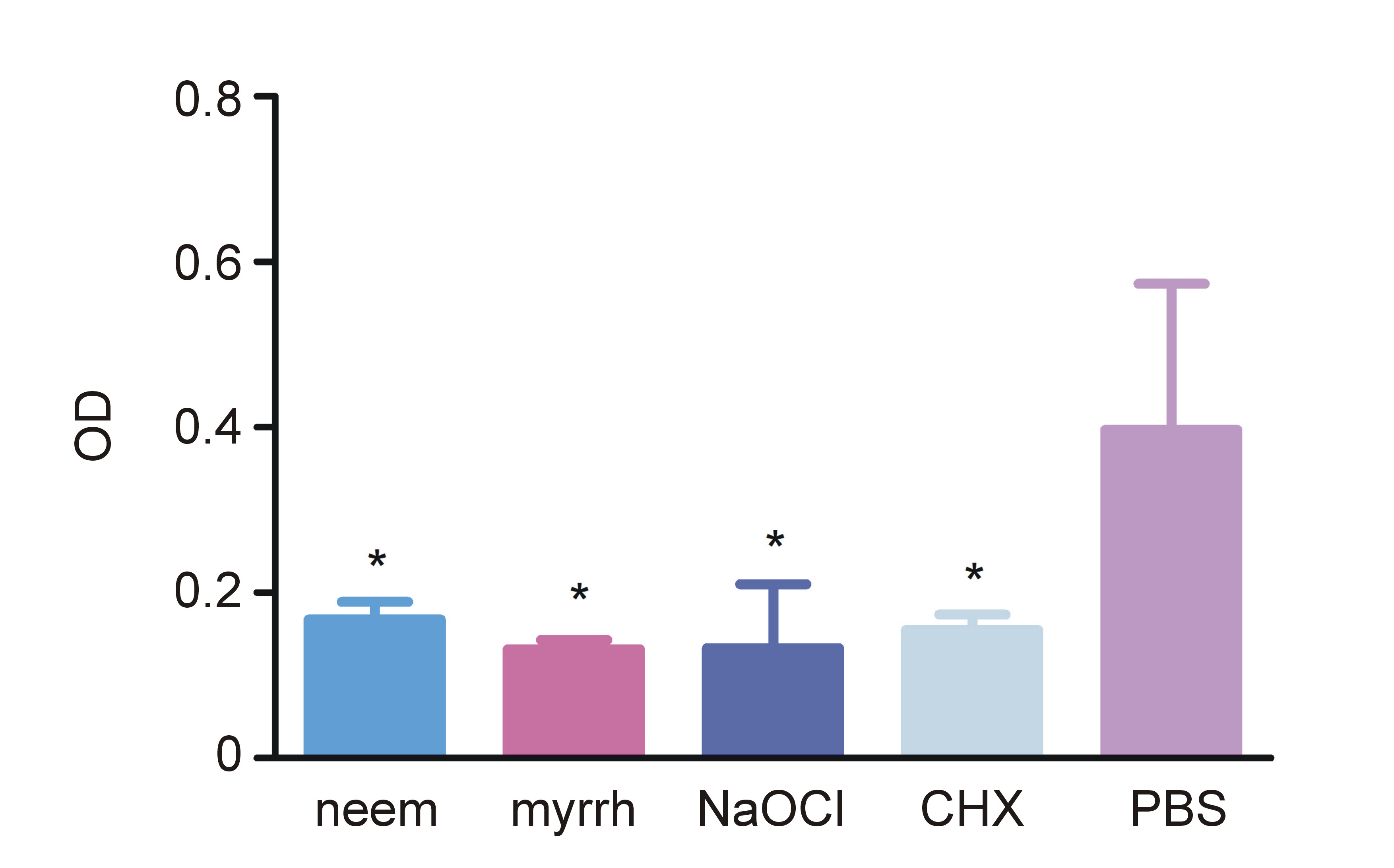
Cell counting kit-8 (CCK-8) assay results, measured at 460 nm, indicated the viability of bacterial cells in the tested and controlled groups (Figure 2). The neem, myrrh, NaOCl, and CHX groups demonstrated significantly reduced cell viability compared with the PBS group (p < 0.001). No statistically significant difference in optical density (OD) values was observed between the neem and myrrh groups.
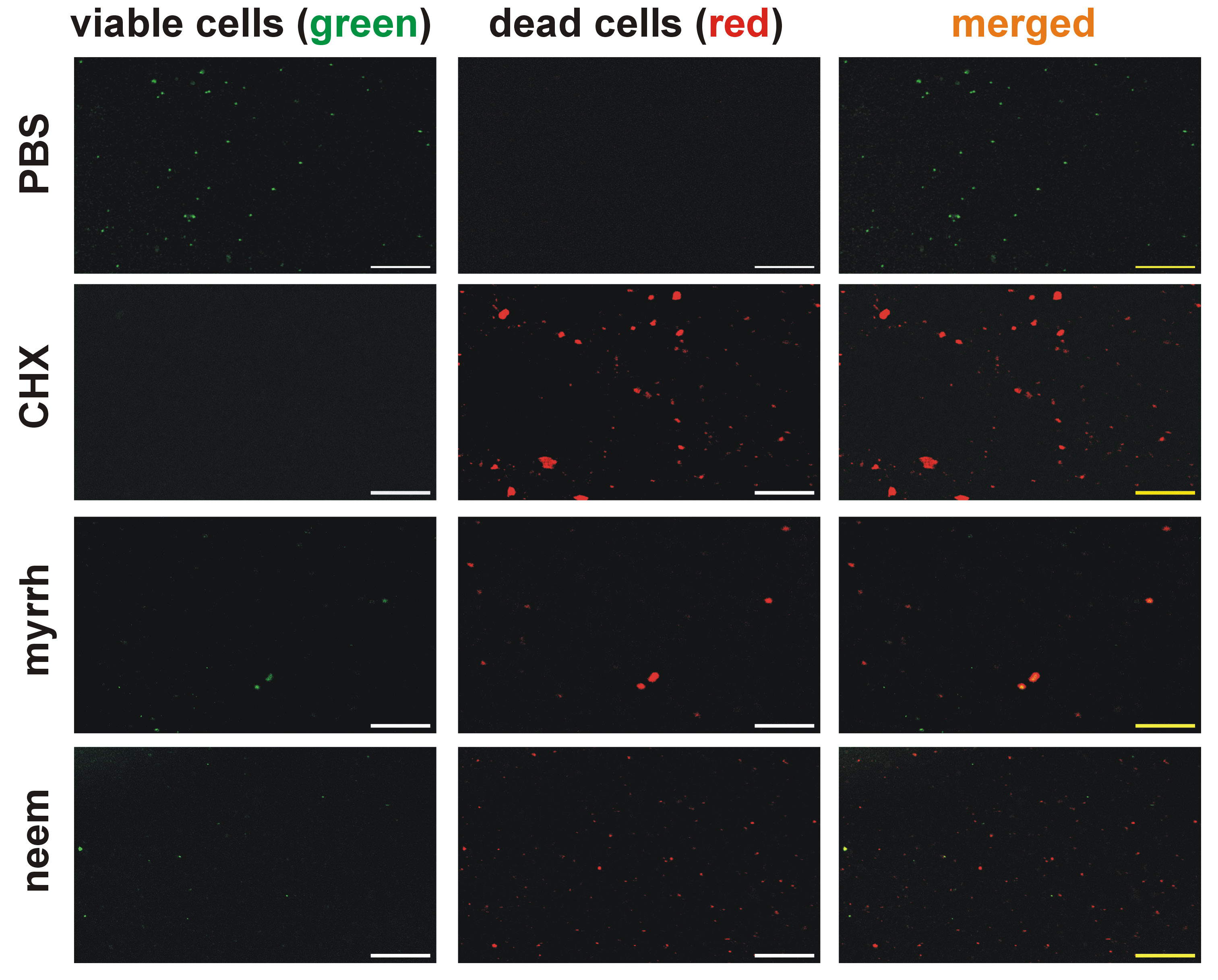
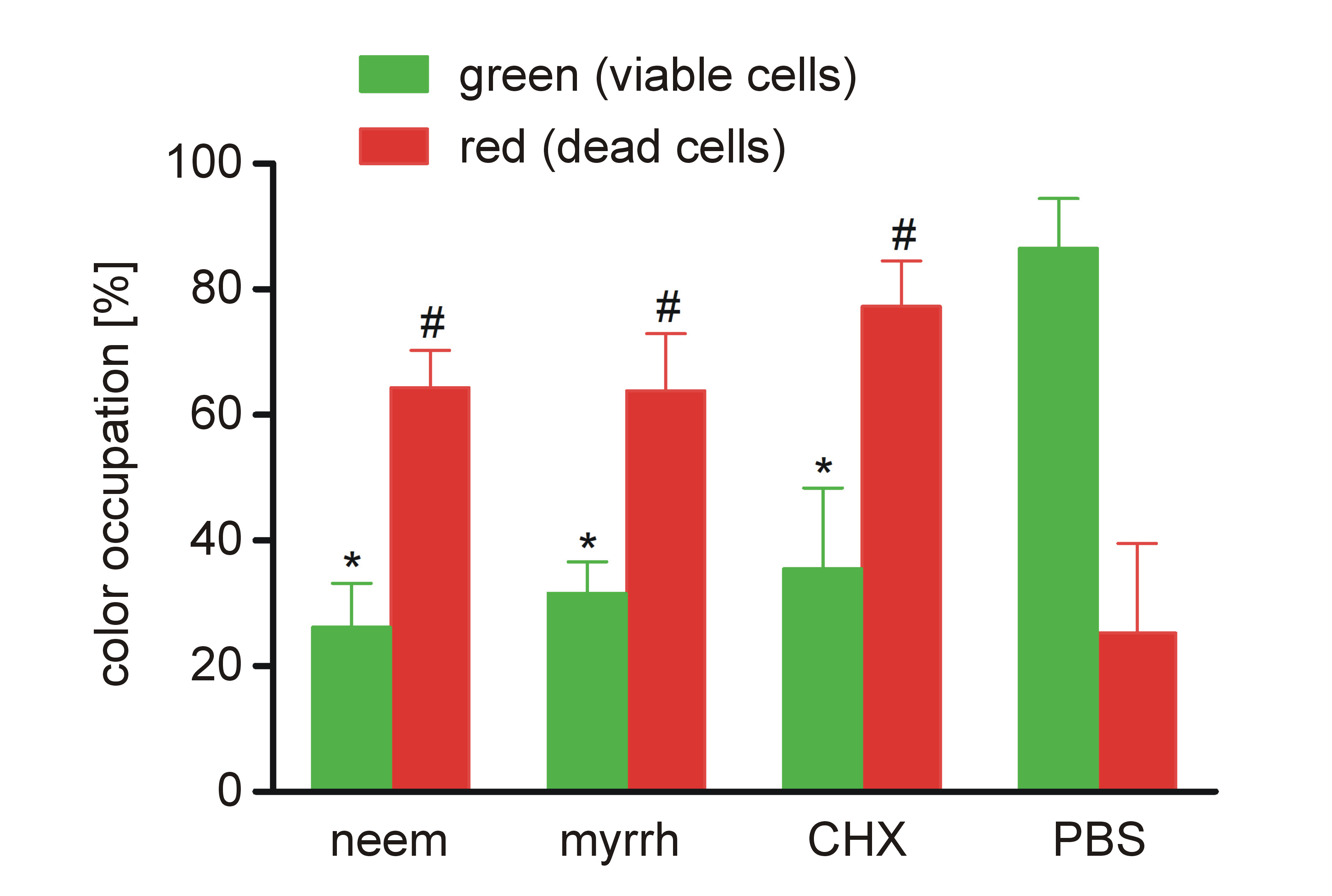
The antibiofilm effects of neem and myrrh extracts were confirmed by the live/dead fluorescence bacterial viability assay. Fluorescence images showed a high proportion of viable green-stained cells in the control group, while the CHX group exhibited predominantly red-stained dead cells with almost no green cells. In the myrrh and neem groups, the number of red-stained dead cells was higher than the number of green-stained live cells (Figure 3). There was a significant increase in the red fluorescence area in the neem, myrrh and CHX groups compared to the PBS group (p < 0.001), while the green fluorescence area was significantly higher in the PBS group (p < 0.001) (Figure 4).
Discussion
The use of natural compounds to promote oral health has been documented in several studies. Plant extracts, including those derived from kiwifruit and cranberry, have demonstrated protective effects on human gingival fibroblasts38 and non-cavitated carious lesions in children.39 Chamomile extract has also shown antibacterial activity against cariogenic pathogens, such as Streptococcus mutans.40 In line with these findings, A. indica (neem, 0.0312/mL) and C. molmol (myrrh, 0.0156/mL), as well as 2% CHX and 5.25% NaOCl, were evaluated in the present study for their antimicrobial efficacy against E. faecalis. The null hypothesis was rejected, as cell proliferation assays revealed significant differences among the tested groups.
Myrrh has long been used as a natural remedy for pain and inflammation of the mouth and throat.41 In addition to its anti-inflammatory activity, it possesses antibacterial, antifungal and anticancer properties.21, 41 Several studies have demonstrated the antimicrobial efficacy of ethanolic myrrh extracts against E. faecalis and Fusobacterium nucleatum.12, 20, 42 Therefore, ethanol was selected as the extraction solvent in the present study. The results of the cell proliferation assay showed that myrrh at a concentration of 0.0156 mg/mL significantly reduced the viability of E. faecalis. The antimicrobial effect of myrrh was comparable to that of 5.25% NaOCl, which is consistent with the findings of Al-Madi et al., who reported that the 0.03 mg/mL ethanolic myrrh extract significantly reduced CFU counts of E. faecalis and F. nucleatum.20 In contrast, Sisodiya et al. reported that 0.2% CHX and 2.5% NaOCl were more effective than ethanolic myrrh extract against E. faecalis, F. nucleatum and Staphylococcus epidermidis.12 This discrepancy can be attributed to differences in extraction techniques. Their study employed maceration, which is generally less efficient than the Soxhlet extraction. The Soxhlet extraction requires less time and less solvent consumption than the maceration method.43 Furthermore, the decrease in E. faecalis count observed in the current study may be due to the antibacterial compounds present in myrrh, including phenols, alkaloids and sesquiterpenes.44 Phenolic compounds exhibit antibacterial activity by binding to nucleophilic amino acids in bacterial proteins, leading to protein inactivation and loss of function, thus preventing the life cycle of bacteria.45 Alkaloids inhibit protein biosynthesis and alter the permeability of bacterial biomembranes.46 Moreover, sesquiterpenes interact with cell envelopes, causing the disruption of cell membranes and subsequent bacteriolysis.21
Neem has traditionally been used to treat inflammation, infections, fever, skin disorders, and dental diseases. It also exhibits antibacterial, antiviral, antifungal, antioxidant, anti-inflammatory, and antimalarial properties.21, 47 Ethanolic neem leaf extracts have demonstrated greater antimicrobial activity than aqueous extracts.48 Rajasekaran et al. concluded that both ethanolic and dichloromethane neem extracts were active against several bacterial species.49 Therefore, the ethanolic extract of neem was used in the current study. Previous investigations by Ravikumar et al. and Khan et al. have reported strong antimicrobial activity of neem against E. faecalis.50, 51 Bohora et al. also demonstrated that the neem leaf extract exhibited superior antimicrobial effects against E. faecalis, Candida albicans and mixed cultures.52 Khan et al. further reported that the neem leaf extract produced the maximum zone of inhibition against Enterococcus at a 6-mL concentration and remained effective at lower concentrations against Staphylococcus aureus and Enterococcus.51 These results are consistent with the current study, in which neem showed the antimicrobial activity against E. faecalis at 0.0312 mg/mL. The antimicrobial activity of neem may be attributed to its bioactive constitutents, including nimbidin, nimbolide, gedunin, azadirachtin, mahmoodin, margolone, and cyclic trisulfide.21, 53 In the present study, the neem leaf extract demonstrated antimicrobial efficacy comparable to that of 2% CHX, 5.25% NaOCl and 0.0156 mg/mL myrrh extract. These results differ from previous studies in which neem was reported to be more effective than NaOCl.52, 54 This discrepancy may be explained by the lower NaOCl concentrations (2% and 3%) used in those studies compared with the present study.
In the current study, the differences between neem and myrrh were not statistically significant. However, both extracts showed variable performance across different assays. In the CFU assay, myrrh demonstrated slightly superior antibiofilm activity compared with neem. Similarly, in the CCK-8 assay, myrrh exhibited marginally better results. In contrast, the live/dead fluorescence assay revealed no marked visual differences between the two extracts. These findings are consistent with those by Anand et al.,21 who reported greater inhibition of E. faecalis by myrrh, followed by neem, using real-time PCR analysis. Yet, Arora et al. found that neem exhibited greater antimicrobial activity than myrrh against E. faecalis using the disc diffusion method.55 Additionally, the Victors model56 described multiple virulence factors that could be targeted in the Enterococcus species (Supplement 1 (available on request from the corresponding author)). The impact of myrrh and neem on virulence genes of E. faecalis and their encoded proteins, such as enterococcal surface protein, lipoteichoic acid, gelatinase, hemolysin, extracellular superoxide production, and toxin cytolysin, should be determined to validate their effects.
Using appropriate microbiological methods is essential to accurately evaluate antimicrobial and antibiofilm effects. One of the main strengths of the present study is the use of various complementary assays to validate the findings, including CFU assays and cell proliferation analysis using CCK-8/WST-8, a reliable and highly sensitive method for evaluating cell viability. Moreover, the effects of neem and myrrh extracts on E. faecalis viability were evaluated using the LIVE/DEAD™ BacLight™ Bacterial Viability Kit. Fluorescence imagining provided valuable qualitative information regarding biofilm integrity and bacterial viability.57 Future studies may include scanning electron microscopy and molecular techniques to further assess morphological changes of the bacterial cells, mechanisms of bacterial resistance and virulence factors.
Limitations
Several limitations may have influenced the present findings and should be taken in consideration when interpreting the results. In this study, vortexing was used to detach bacterial biofilms from treated samples. Although ultrasound techniques are generally considered more efficient for biofilm removal, both methods are valid and yield a considerable number of biofilms.58, 59 While no comprehensive phytochemical analyses were performed to identify specific bioactive compounds in the extracts, the neem and myrrh extracts are well-documented and a wide range of bioactive constituents was identified in previous studies.21, 44, 53 Even though the traditional use of myrrh and neem may warrant their safety,60 further investigations are required, particularly regarding their effects on tooth enamel and oral keratinocytes, before clinical application in endodontic therapy.
Conclusions
The current study highlights the strong potential of myrrh and neem extracts to reduce bacterial adhesion on dentinal surfaces and act as effective antibiofilm agents. These findings suggest that both extracts may serve as promising natural disinfectants in root canal treatment. Future research should focus on identifying the bioactive compounds in myrrh and neem extracts, which are responsible for their antimicrobial activity. Evaluating the effects of myrrh and neem extracts against other oral pathogens associated with endodontic failure may enhance their therapeutic value. Further studies are also warranted to investigate their mechanisms of action and optimize their potential clinical applications in endodontic practice.
Ethics approval and consent to participate
The present in vitro study was conducted in accordance with the Declaration of Helsinki. The research protocol was approved by the Institutional Review Board at Imam Abdulrahman Bin Faisal University, Dammam, Saudi Arabia, on May 29, 2022 (approval No. IRB-2022-02-210).
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Consent for publication
Not applicable.
Use of AI and AI-assisted technologies
Not applicable.