Abstract
Background. The mechanical reliability of occlusal splints and their long-term behavior are significant factors determining the clinical outcome of temporomandibular disorder (TMD) therapy. However, improvements are still needed in this area.
Objectives. This in vitro study aimed to (1) compare the hardness and flexural properties of materials manufactured using 3 techniques for occlusal splint fabrication (conventional heat curing, thermoforming followed by light curing, and three-dimensional (3D) printing) and (2) analyze the effect of artificial aging on the properties of the materials.
Material and methods. A total of 120 disc-shaped specimens were manufactured for the Shore D hardness evaluation, and 120 bar-shaped specimens were fabricated for the flexural properties evaluation (n = 15 for each group). Each material was tested in 2 groups of specimens, non-aged and artificially aged (stored for 90 days in water at 37°C). Statistical differences were assessed using one-way or two-way parametric analysis of variance (ANOVA) with Tukey’s or Šídák’s post-hoc test, or the non-parametric Kruskal–Wallis test with Dunn’s post-hoc test. A p-value of less than 0.05 was considered statistically significant.
Results. The mechanical properties of the materials varied significantly. Among the non-aged materials, the 3D-printed resin exhibited the highest Shore D hardness (85.3D), but it decreased significantly after 90 days of water storage (80.4D, p < 0.0001). The unpolished heat curing acrylic showed the highest resistance to artificial aging (p = 0.0436). However, its hardness decreased significantly after polishing (81.0D vs. 83.4D, p = 0.0015). The conventional heat curing material also exhibited superior flexural properties (σ = 89.63 MPa, E = 2616 MPa). All tested materials were susceptible to deterioration due to aging.
Conclusions. The conventional method of occlusal splint fabrication remains the optimal choice, particularly for long-term use. However, it is still necessary to develop materials that are resistant to aging in order to ensure successful clinical performance.
Keywords: polymer, thermoforming, dental materials, 3D printing, oral splint
Introduction
In accordance with the Glossary of Prosthodontics Terms, an occlusal splint is “any removable artificial occlusal surface affecting the relationship of the mandible to the maxillae used for diagnosis or therapy; uses of this device may include, but are not limited to, occlusal stabilization for treatment of temporomandibular disorders, diagnostic overlay prior to extensive intervention, radiation therapy, occlusal positioning, and prevention of wear of the dentition or damage to brittle restorative materials such as dental porcelain.”1 In particular, occlusal splints are a critical component of the non-invasive management of bruxism and temporomandibular disorders (TMD). They can be used for occlusal positioning or stabilization and for preventing tooth wear, which is a multifactorial condition. Studies reported the beneficial effect of occlusal splints in reducing masticatory muscle activity.2, 3, 4, 5, 6 However, the effectiveness of occlusal splints in managing orofacial myalgia and myofascial pain still requires investigation.7 Similarly, various alternative treatment modalities are being validated for their ability to improve the quality of life of patients with TMD.8, 9, 10
Due to the high prevalence of TMD and the increasing popularity of occlusal splints, there is a need to search for new, reliable and convenient device manufacturing methods. Thermoplastics for vacuum forming or pressure forming offer an easier and faster fabrication process compared to heat-cured and self-cured (auto-polymerizing) acrylic resins, which were the first materials used to construct custom-made splints. Nekora et al. demonstrated that patients had no preference between vacuum-formed and heat-cured acrylic splints.11 In addition to conventional laboratory-made devices, digitally fabricated milled splints represent a viable alternative.12
Modern dentistry frequently employs computer-aided design/computer-aided manufacturing (CAD/CAM) techniques, which have led to the development of subtractive and additive techniques. As a result, three-dimensional (3D) printing is becoming increasingly popular among prosthodontists worldwide. The technology is used to produce prosthetic restorations, dental models, implants, surgical guides, custom trays, orthodontic appliances, and occlusal splints.13, 14, 15, 16, 17 Furthermore, it provides a simple means of manufacturing complex, custom-designed objects, reducing material waste and working time.18, 19 Despite its many advantages, there are several limitations to 3D printing that require comprehensive research into the materials and techniques used in the fabrication of oral appliances.20, 21, 22
The fabrication of occlusal splints may be simplified by the introduction of 3D printing or thermoforming technologies, provided that the materials used are mechanically reliable, biologically safe and long-lasting. This is necessary to ensure the clinical success of occlusal splint therapy.23, 24 Several in vitro studies have evaluated the mechanical behavior of occlusal splint materials processed using various methods,25, 26, 27, 28, 29, 30 while others have demonstrated that post-curing affects the results.31, 32 However, most of the research conducted to date has focused on evaluating the initial parameters of splint materials. Few studies have addressed the long-term behavior of such devices.22, 27, 33, 34, 35, 36, 37, 38, 39, 40 The problem of poor occlusal splint longevity, mainly due to fracturing or deforming over time, still needs to be solved.29, 34, 38
Flexural strength and hardness are important mechanical properties that determine a material’s capacity to resist bending and indentation, which are the main causes of splint damage. Hence, this in vitro study aimed to compare the hardness and flexural properties of specimens manufactured using 3 techniques for occlusal splint fabrication (conventional heat curing, thermoforming followed by light curing, and 3D printing). The study examined the impact of 90 days of water storage on material properties. The research hypotheses state that there are no significant differences in the selected material properties between (1) specimens manufactured using different techniques and (2) specimens subjected to artificial aging and non-aged ones.
Material and methods
Material
The materials selected for this in vitro study could be used to fabricate occlusal splints using 3 different manufacturing techniques, as listed in Table 1: a conventional hand-processed heat-curing acrylic resin (polymethyl methacrylate (PMMA), Villacryl H Plus 0; Everall7, Warsaw, Poland); a plastic (polyethylenterephthalat+glycol (PET-G)) sheet processed via thermoforming (DURAN®; Scheu-Dental GmbH, Iserlohn, Germany) and adjusted with a build-up made of a light-cured (LC) mixture of acrylic resins, fillers and initiators (Durasplint LC; Scheu-Dental GmbH); a photopolymer resin for 3D printing via the stereolithography (SLA) method (Dental LT Clear; Vertex Dental, Soesterberg, Netherlands).
Specimen preparation
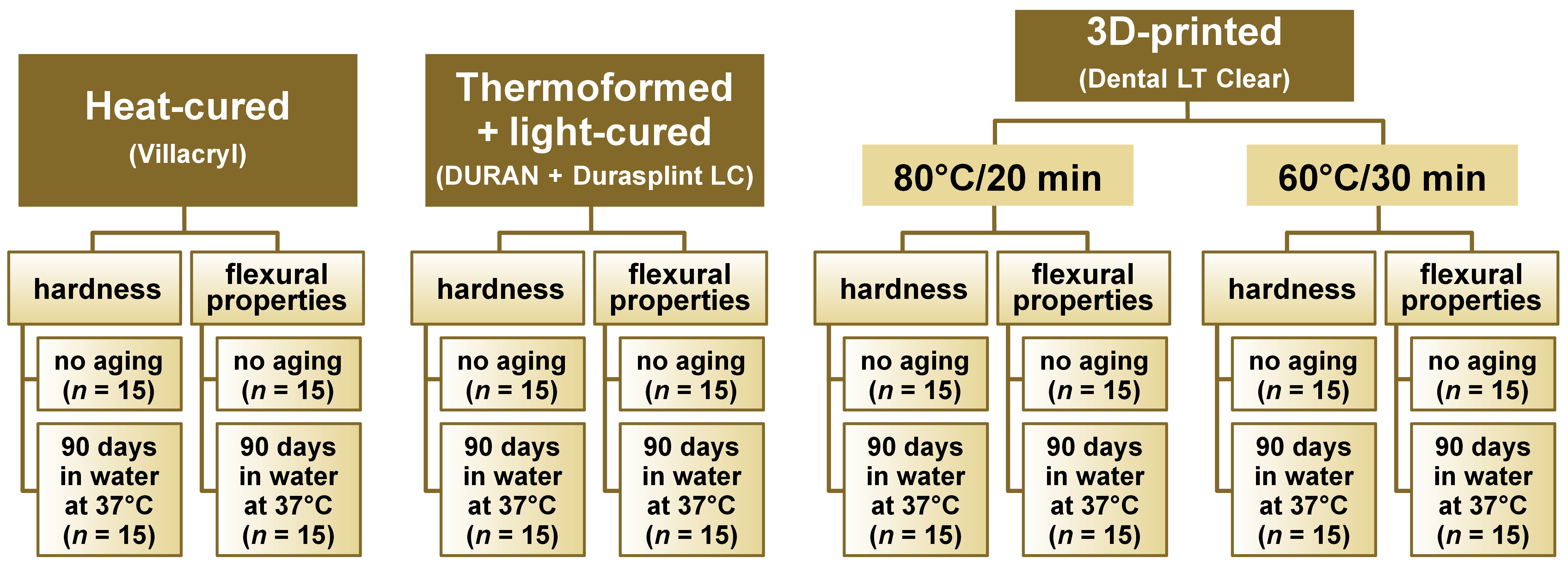
The materials were processed according to the manufacturer’s instructions to prepare disc-shaped and bar-shaped specimens that comply with the relevant International Organization for Standardization (ISO) standards. To limit intragroup variance, a single investigator was involved in specimen preparation. A total of 120 disc-shaped specimens (for the Shore D hardness evaluation) and 120 bar-shaped specimens (for the flexural properties evaluation) were fabricated. Each material was tested in 2 groups of specimens, non-aged and artificially aged (stored for 90 days in water at 37°C). The overall study design is illustrated in Figure 1.
Heat curing
The powder was mixed with the liquid (Villacryl H Plus 0), and when the material reached a dough-like consistency, it was introduced into the mold in a polymerization flask. Then, it was pressed under 8.6 bar using a P-400 hydraulic press (Sirio Dental, Meldola, Italy) and polymerized under short-term conditions (heating the water from 60°C to 100°C for 30 min and then boiling it for additional 30 min) in an ISP-1 polymerization unit (InterSonic, Olsztyn, Poland).
Thermoforming and light curing
The bases of the specimens were made of 1.5-mm DURAN (Scheu-Dental GmbH) thermoformed over a die of the appropriate size using the Ministar S® pressure molding unit (Scheu-Dental GmbH). Then, the surface of the material was sandblasted with aluminum oxide (Al2O3, 110 µm) (Ardent, Wrocław, Poland) in a sandblasting unit (Basic Eco; Renfert, Hilzingen, Germany) and dried. A thin layer of LC-Primer (Scheu-Dental GmbH) was applied to the upper surface of DURAN and polymerized for 5 min in an LC-6 Light Oven (Scheu-Dental GmbH). Then, Durasplint LC (Scheu-Dental GmbH) was adapted to the pressure-molded DURAN base. The final specimen was cured twice for 10 min using the LC-6 Light Oven.
Three-dimensional printing
The 3D printing objects were designed using Meshmixer v. 3.5.474 (Autodesk Inc., San Francisco, USA). The .stl files were created and uploaded to PreForm software, v. 3.28.1 (Formlabs Inc., Somerville, USA), for the addition of supports and the setting of printing parameters. The specimens made of the Dental LT Clear resin (Vertex Dental) were printed in a Form 2 printer (Formlabs Inc.) at a 90° angle to the building platform (edgewise orientation of the specimens) in 100-μm layers with supporting structures. The printed specimens were placed in the Cleaning and Finish Kit (Formlabs Inc.), washed twice for 10 min in 99% isopropanol (PPH Stanlab, Lublin, Poland), and left to air dry at room temperature for 30 min. They were then post-cured in a Form Cure (Formlabs Inc.) equipped with 405-nm multi-directional light emitting diodes (LEDs). Two sets of curing parameters were tested, namely 80°C for 20 min (as recommended by the manufacturer) and 60°C for 30 min (the alternative method).
Finishing and polishing
All specimens were finished using sandpaper (grit P500, P1000 and P1200; P.S. Trading, Ożarow Mazowiecki, Poland) and 0.6-mm pumice stone powder (Everall7). Finally, the upper side of each specimen was polished with polishing paste for resin and metals (Everall7) using a Poliret Mini unit (REITEL Feinwerktechnik GmbH, Bad Essen, Germany). For the specimens made of thermoformed and LC materials, the side made of DURAN was left unpolished, while the side made of Durasplint LC was polished, following standard clinical practice.
Artificial aging
The specimens were stored in distilled water (chemPUR, Karlsruhe, Germany) at 37°C for 90 days in the CLN 15 Smart incubator (Pol-Eko sp. k., Wodzisław Śląski, Poland). The water was changed weekly and a constant temperature was maintained.
Shore D hardness evaluation
The tests were conducted by a single investigator in accordance with the PN-EN ISO 868:2005 guidelines.41 Before testing, the disc-shaped specimens with a diameter of 30 mm and a height of 5 mm were conditioned for 88 h in a standard atmosphere (23/50). The Shore D hardness value was measured for each specimen at 5 points (at a distance of at least 9 mm away from the specimen’s edges and 6 mm away from each other) using an HBA 100-1 Shore durometer (Sauter AG, Basel, Switzerland). The polished and unpolished sides of the specimens were tested separately. The value was read 15 s after pressing the durometer foot against the specimen.
Flexural properties evaluation
The tests were conducted by a single investigator in accordance with the PN-EN ISO 20795-1:2013 guidelines.42 Before testing, the bar-shaped specimens (64 mm × 10.0 (±0.2) mm × 3.3 (±0.2) mm) were conditioned in distilled water at 37°C for 50 h. Then, the height and width of each specimen were measured at 5 points using a Magnusson digital caliper (150 mm) (Limit, Alingsås, Sweden), and the mean cross-sectional area was calculated immediately before testing. The three-point bending test was performed using the Universal Testing Machine (Z10-X700; AML Instruments Ltd, Lincoln, UK) at a constant displacement rate of 5 mm/min and a span length of 50 mm between the supports.
Flexural strength (σ [MPa]) was calculated using the following formula (Equation 1):
where:
F – maximum load [N];
l – distance between the supports [mm] (±0.01 mm);
b – width of the specimen [mm]; and
h – height of the specimen [mm].
Flexural modulus (E [MPa]) was determined using the following formula (Equation 2):
where:
load (F) divided by displacement (d) is the slope in the linear elastic region of the load/displacement curve; l, b and h are as defined above.
Statistical analysis
The results were analyzed using GraphPad Prism software, v. 9.1.2. (GraphPad Software, San Diego, USA). All measurements were carried out for n = 15 specimens in each group. The sample size was calculated using G*Power software, v. 3.1.9.7 (https://www.psychologie.hhu.de/arbeitsgruppen/allgemeine-psychologie-und-arbeitspsychologie/gpower), for an effect size of 0.4, α = 0.05 and power of 0.8. The results were presented as box plots, which provide a visual representation of the five-number summary of a dataset (minimum, first quartile, median, third quartile, and maximum). Data normality was tested with a Shapiro–Wilk test. The majority of data passed the normality test, with the exception of the results for the flexural modulus evaluation and the flexural strength after aging. These results were analyzed using the non-parametric Kruskal–Wallis test.
Differences between the hardness of the materials were tested using two-way parametric analysis of variance (ANOVA) for multiple comparisons with Tukey’s (for comparisons of materials within polished or unpolished groups) or Šídák’s (for comparisons between polished and unpolished groups within each material and for comparison between artificially-aged and non-aged groups) post-hoc multiple comparisons test. The study tested the differences in flexural properties of the materials using either one-way parametric ANOVA with Tukey’s post-hoc multiple comparisons test or the non-parametric Kruskal–Wallis ANOVA with post-hoc Dunn’s test. Differences in the flexural properties between artificially aged and non-aged groups were compared using two-way ANOVA with Šídák’s post-hoc multiple comparisons test. Differences between the groups were considered statistically significant at p < 0.05.
Results
Shore D hardness
The Shore D hardness of the materials varied between the groups (Figure 2). The two-way ANOVA showed significant differences in hardness between specimens manufactured using different techniques and between specimens subjected to artificial aging and non-aged ones (all p < 0.0001). On this basis, both null hypotheses were rejected, and detailed multiple comparisons were conducted. When unpolished non-aged specimens were analyzed, the lowest value was found for those made of DURAN and processed via thermoforming (p < 0.0001 for all comparisons), while the highest value was found for specimens made of a 3D-printable Dental LT resin cured for 20 min at 80°C (p = 0.0054 when compared to the heat-cured PMMA and p = 0.2171 when compared to the resin cured for 30 min at 60°C) (Figure 2). Additionally, polishing significantly reduced the Shore D hardness of the heat-cured PMMA (p = 0.0015). There was a significant difference between the 2 sides of thermoformed LC specimens. The side made of DURAN (unpolished) had a significantly lower Shore D hardness than the side made of Durasplint LC (polished) (p < 0.0001). No significant differences were observed between unpolished and polished specimens for both types of post-curing. The resin used for 3D printing exhibited the highest Shore D hardness value among all polished materials, regardless of the final post-curing parameters applied.
The analysis of the specimens subjected to artificial aging revealed interesting results (Figure 2). When specimens without polishing were compared, the heat-cured PMMA and thermoformed DURAN specimens showed the highest Shore D hardness. However, among the polished specimens, the 3D-printed resins had the highest Shore D hardness, while the Durasplint LT specimens had the lowest value (all p < 0.0001).
A comparison of the non-aged and aged specimens revealed that the Shore D hardness of the 3D-printable Dental LT resin was significantly reduced among the unpolished materials after 90 days of water storage (p < 0.0001). Conversely, the Shore D hardness of DURAN significantly increased after artificial aging (p < 0.0001). Only the conventional heat-cured PMMA demonstrated greater resistance to changes in hardness; however, the difference between aged and non-aged specimens was still significant (p = 0.0436). When comparing the polished specimens, prolonged water storage resulted in a significant reduction of the Shore D hardness for all analyzed materials (all p < 0.0001) (Table 2).
Flexural properties
The materials differed in terms of ultimate flexural strength and flexural modulus (Figure 3). Statistical tests, including the one-way ANOVA, Kruskal–Wallis test and two-way ANOVA revealed significant differences in flexural properties between specimens manufactured using different techniques and between specimens subjected to artificial aging and non-aged ones (all p < 0.0001). On this basis, both null hypotheses were rejected, and detailed multiple comparisons were performed. When non-aged specimens were compared, the highest values were observed for the conventional heat-cured PMMA, while specimens made of the 3D-printable resin had significantly lower flexural strength values. The material post-cured for 20 min at 80°C had the lowest value, but the difference between the 2 types of post-curing was not statistically significant (p = 0.2629). The comparison of flexural modulus revealed that the lowest modulus was obtained for the 3D-printable resin post-cured for 30 min at 60°C (Figure 3). Upon analysis of artificially aged specimens, the materials were ranked in terms of flexural strength or modulus in the same manner as non-aged specimens. However, the differences between the materials were more pronounced (Figure 3). The comparison of flexural strength differences between non-aged and artificially aged materials revealed that long-term exposure to increased temperatures and humidity significantly influenced their mechanical properties (Table 3). For materials processed by thermoforming and light-curing and by 3D printing, water storage significantly reduced their flexural strength and modulus (all p < 0.0001). In the case of heat-cured acrylic resin, artificial aging did not affect its ability to withstand bending forces, as there was no significant decrease in ultimate flexural strength observed (p = 0.9992). However, the flexural modulus was significantly reduced (p = 0.0347).
Discussion
The 3D printing technology has the potential to revolutionize the fabrication of occlusal splints. This technology enables the precise and cost-effective production of complex parts, while reducing material waste and environmental impact. However, the benefits of this technology must be accompanied by clinical success, which is strongly dependent on the mechanical reliability, biological safety and longevity of the materials used.
Occlusal splints must be mechanically resistant due to the excessive occlusal forces they are often subjected to.6 Numerous research studies have compared the mechanical properties of 3D-printed splints and conventionally fabricated appliances, although an indisputable conclusion has not been reached. Similarly, studies on thermoplastic foils, in particular those with a build-up made of an LC resin, are scarce.14 A study by Patzelt et al. revealed that a digital workflow was more time-efficient, and the fit of the digitally-fabricated splints was better than that of the conventionally-fabricated ones. The wear of the 2 different materials showed comparable results.19 In contrast, Lutz et al. demonstrated that 3D-printed splint materials had lower wear and fracture resistance compared to those that were milled or conventionally fabricated.29 Furthermore, a comparison between thermoformed DURAN clear dental aligners and the 3D-printed Dental LT clear resin revealed that 3D-printed material was more accurate and able to withstand higher compressive loads.28
In the current study, we evaluated 2 mechanical properties of materials that determine the clinical performance of occlusal splints and are closely related to the resistance to localized deformation. Splints are exposed to flexural stress during teeth clenching and grinding, which can cause deformation, crack formation and, ultimately, fracture. The basic qualities that can be used to describe the resistance of a material to such processes are Shore D hardness, which measures its resistance to indentation, and flexural strength and modulus, which measure its resistance to bending. To the best of the authors’ knowledge, this is the first comparative study of material properties used in 3 types of splint fabrication techniques, including thermoforming and LC. The study accounts for differences between polished and non-polished specimens and complies with the relevant standards. The results indicated that 3D-printed materials had the highest Shore D hardness, while the conventional heat-cured PMMA demonstrated superior flexural properties. The hardness and elastic modulus of occlusal splint materials manufactured using 4 different methods (heat curing, CAD-milling, 3D printing, and vacuum forming) were also evaluated by Grymak et al., revealing that both the processing method and the printing angle had an influence on the hardness and elastic modulus of the materials.26 The same group revealed that vacuum-formed occlusal splints showed the highest wear,43 while 3D-printed materials exhibited significantly lower wear resistance compared to CAD-milled and heat-cured materials.27
A significant strength of the present study is the use of artificial aging, which is crucial for simulating the impact of long-term usage in the oral environment. Several methods of artificial aging are used for the in vitro evaluation of dental materials, including thermal degradation (simulated via thermocycling), physical and chemical degradation in various environments (storage in water, artificial saliva, ethanol, and sodium hydroxide solutions), as well as mechanical loading under varying conditions (static and dynamic tests and simulated chewing or tooth brushing). As occlusal splints are specific oral devices that are removed before eating or drinking, they are not exposed to food, beverages or temperature changes resulting from contact with hot or cold meals. For this reason, instead of thermocycling or immersion in more aggressive media, artificial aging in water at 37°C was performed. An interesting continuation of the preliminary research would be the simulation of mechanical degradation (e.g., dynamic loading).
The effects of 90-day aging on dental splints were analyzed, as their longevity can vary from 6 to 8 weeks to several years, depending on the severity of the disorders. To simulate degradation upon exposure to factors present in the oral cavity, the specimens were immersed in water at elevated temperatures. Several studies on this issue have provided inconsistent conclusions. Antonopoulou et al. showed that 3D-printed occlusal devices were dimensionally stable after applying various storage methods and concluded that splints could be stored with or without water and light, without any clinically detectable effect on fitting accuracy.40 On the other hand, Berli et al. revealed that 3D-printable resins had significantly lower flexural strength and hardness, as well as higher water sorption and solubility, compared to pressed or milled materials. Moreover, 3D-printed materials were found to be more susceptible to thermal aging.34 Similarly, Reymus and Stawarczyk found that 3D-printed materials were more susceptible to artificial aging than the conventionally milled materials.38 In the research presented in this paper, the authors confirmed that all materials tested were prone to a decrease in hardness and flexural properties due to artificial aging. The significant decrease in the mechanical properties of both 3D-printable and conventional materials may result from the plasticization of the polymer structure due to water sorption.33
Limitations
The main limitation of the current study is the limited number of materials and brands compared, the narrow range of properties investigated, and the laboratory nature of the research. For example, analyzing wear resistance through two-body wear tests using a chewing simulator would be a very valuable continuation of the study. Moreover, the clinical success of splint therapy is contingent upon several properties, including the degree of conversion, biocompatibility and microbial adhesion, which are closely related to surface roughness.23, 24, 44, 45Additionally, it would be beneficial to investigate the influence of various cleaning or disinfecting techniques on the properties of splints. Finally, the results of comprehensive in vitro tests could suggest methods to improve the materials and techniques used, paving the way for the fabrication of splints with optimal clinical performance.
Conclusions
Both research hypotheses should be rejected in the light of the results obtained in this paper. The 3D printing resin exhibited the highest Shore D hardness among all polished and artificially aged materials, while the conventional heat-cured PMMA revealed superior flexural properties. All materials were susceptible to the deterioration of mechanical properties due to artificial aging, but the conventional PMMA demonstrated the highest resistance to changes after storage in water at 37°C.
There is still a need to develop new materials and techniques used for occlusal splint production in order to improve resistance to deformation or fracture. Currently, the conventional method still remains the optimal choice for successful clinical performance, especially for long-term splint use.
Ethics approval and consent to participate
Not applicable.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Consent for publication
Not applicable.