Abstract
Background. Natural propolis is widely known to be an antibacterial agent with strong antioxidant and anti-inflammatory properties. In combination with a bovine xenograft, it is expected to promote bone formation in the alveolar ridge, following tooth extraction.
Objectives. The aim of the present study was to evaluate the effect of administering a propolis extract combined with a bovine xenograft on alveolar bone formation.
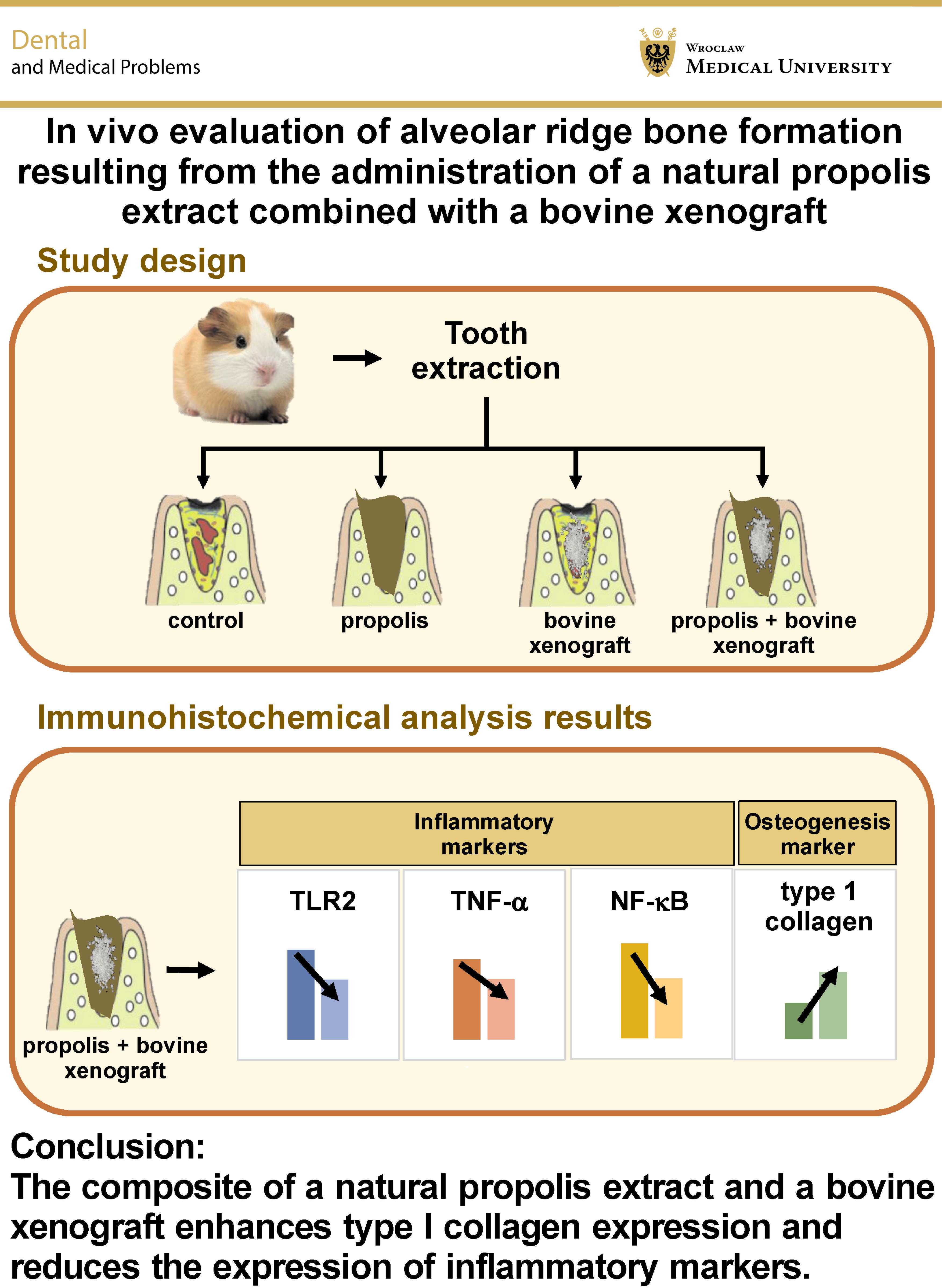
Material and methods. The lower incisor teeth of Cavia cobaya were extracted and the resulting alveolar sockets were filled with different materials based on the group assignment: polyethylene glycol (PEG); a propolis extract (PE); a bovine xenograft (BX); and the propolis extract–bovine xenograft composite (PE–BX). The alveolar sockets were examined after 7 and 30 days by means of immunohistochemical staining for the expression of nuclear factor kappa-light-chain-enhancer of activated B cells (NF-ĸB), toll-like receptor 2 (TLR2), tumor necrosis factor-alpha (TNF-α), and type 1 collagen. The one-way analysis of variance (ANOVA) and post-hoc Tukey’s test were used to compare the groups.
Results. The PE–BX group demonstrated promising results, with increased type I collagen levels and reduced expression of inflammatory markers (NF-κB, TLR2 and TNF-α) on days 7 and 30 (p < 0.001). Furthermore, in comparison with the BX group, this material demonstrated significantly higher type I collagen expression on days 7 and 30 (p < 0.01).
Conclusions. The composite of a natural propolis extract and a bovine xenograft enhances type I collagen expression and reduces the expression of inflammatory markers. Further research is warranted to explore its potential for alveolar bone preservation.
Keywords: propolis, medicine, bone formation, tooth extraction socket, alveolar socket healing
Introduction
Bone resorption following tooth extraction can result in significant complications for subsequent prosthetic rehabilitation, including removable and fixed dentures, as well as dental implants. Excessive force during extraction may trigger severe inflammation, leading to increased osteoclast activity and consequent alveolar bone resorption. Therefore, alveolar ridge preservation is essential to minimize post-extraction bone loss.1, 2 This principle should be applied in all cases of edentulous ridges to optimize the functional and esthetic outcomes of prosthetic treatment, particularly implant therapy.3, 4
Natural propolis is widely recognized as an alternative therapeutic agent for the treatment of various diseases. Numerous studies have demonstrated that propolis exhibits antibacterial activity (particularly against Gram-positive bacteria), as well as strong antioxidant and anti-inflammatory properties.5, 6, 7, 8 Propolis contains resins and various bioactive compounds, including bioflavonoids, artepillin, apigenin, and caffeic acid phenethyl ester (CAPE), which are known for their anti-inflammatory, antioxidant, antibacterial, antiviral, and immunomodulatory effects, and have been shown to promote tissue healing.9, 10, 11, 12
A study by Kresnoadi et al. investigated osteogenesis induced by a combination of propolis and a bovine bone graft, reporting higher expression of heat shock protein 70 (HSP 70) and osteocalcin, and increased osteoblast numbers as compared to the control group at days 3 and 7.13 Further research by the same group demonstrated that this combination enhanced transforming growth factor-beta (TGF-β) expression and osteoblast numbers, while reducing interleukin-1β (IL-1β) levels and osteoclast production during alveolar bone regeneration.14
Under physiological conditions, bone undergoes continuous remodeling through the coordinated activities of osteoclasts and osteoblasts. Nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) plays a pivotal role in osteoclastogenesis, and is activated via the canonical pathway by receptor activator of nuclear factor kappa-B ligand (RANKL) and tumor necrosis factor-alpha (TNF-α). In contrast, toll-like receptors (TLRs) contribute to the maintenance of bone homeostasis by modulating inflammatory responses.15, 16 Type I collagen constitutes a major component of the organic bone matrix and is synthesized primarily by osteoblasts.17 These markers are therefore important indicators in the evaluation of alveolar bone formation during socket healing.
This study aimed to develop an alternative material for alveolar ridge preservation based on a composite of a natural propolis extract and a bovine xenograft, with the objective of enhancing bone regeneration and reducing the risk of ridge resorption.
Materials and methods
This research was approved by the Ethical Clearance Commission at the Faculty of Dental Medicine of the Airlangga University, Surabaya, Indonesia (approval No. 014/HRCC FODM/III/2018).
The first preparation consisted of a natural propolis extract (PE) obtained from an apiary in East Java. The bovine xenograft (BX) used in this study was sourced from the tissue bank of Dr. Soetomo General Academic Hospital, Surabaya, Indonesia. The PE–BX composite was prepared using polyethylene glycol (PEG) as a carrier. PEG 400 and PEG 4000 (CARBOWAX™; Dow, Jakarta, Indonesia) were mixed at a 1:1 ratio prior to incorporation with the active materials.
The study comprised 4 groups: control; PE; BX; and PE–BX. In each treatment group, the material contained 2% active ingredient, as recommended in a previous study by Yuli Prabowo et al.18
Healthy male guinea pigs (Cavia cobaya) were selected as the experimental subjects based on the following criteria: 3.5 months of age; body weight of 300–350 g; the absence of visible lesions; and intact sensory function. The animals were acclimatized for 1 week prior to the experiment, and were provided standard pellet feed and water ad libitum.
Anesthesia was induced by the intramuscular injection of ketamine at a dose of 20 mg per 300 g b.w. (Kepro, Deventer, the Netherlands). Following anesthesia, the lower incisor was extracted. The extraction socket was filled with the designated material according to the treatment group, and subsequently closed using simple interrupted sutures.
On days 7 and 30, the animals were euthanized to allow the harvesting of the lower incisor tissue. The specimens were fixed in 10% neutral buffered formalin (Sigma-Aldrich, St. Louis, USA) and decalcified using ethylenediaminetetraacetic acid (EDTA) (Titriplex® III; Merck, Darmstadt, Germany). The tissues then underwent dehydration, clearing, paraffin embedding, and deparaffinization. Subsequently, the samples were processed for immunohistochemical analysis to evaluate the expression of NF-κB, TLR2, TNF-α, and type I collagen, using specific monoclonal antibodies (Abcam, Cambridge, UK). The prepared slides were examined under a light microscope at magnifications of ×100, ×400 and ×1,000.
Observations were focused on the apical third of the extraction socket. Type I collagen expression was assessed in osteoblasts, whereas the expression of NF-κB, TLR2 and TNF-α was evaluated in osteoclasts. Positive cells were counted manually to minimize potential counting errors.
Statistical analysis
The one-way analysis of variance (ANOVA) and post-hoc Tukey’s test were used to compare the groups (IBM SPSS Statistics for Windows, v. 22.0 (IBM Corp., Armonk, USA)). The p-value applied in this study was 0.05.
Results
NF-ĸB expression
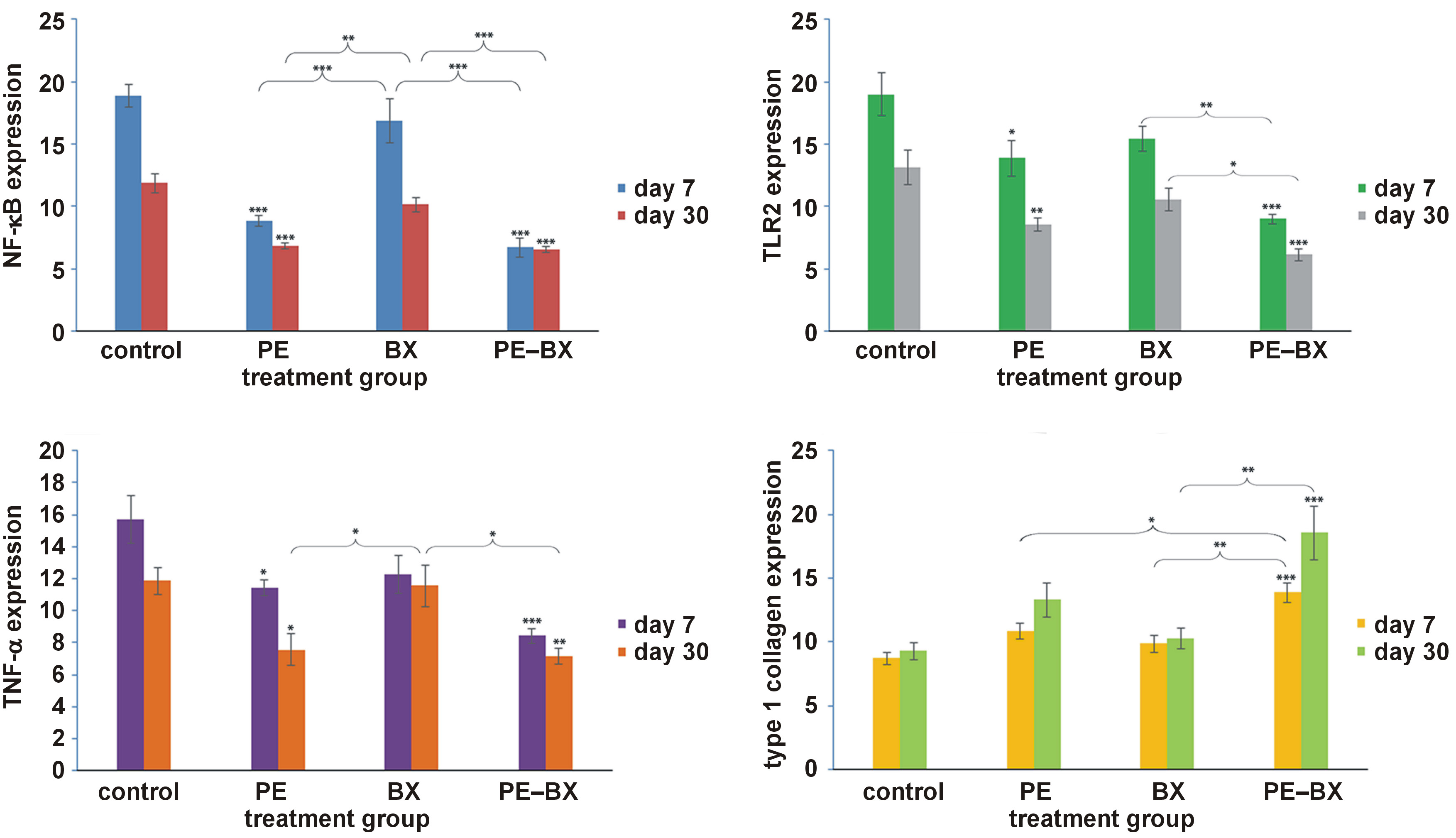
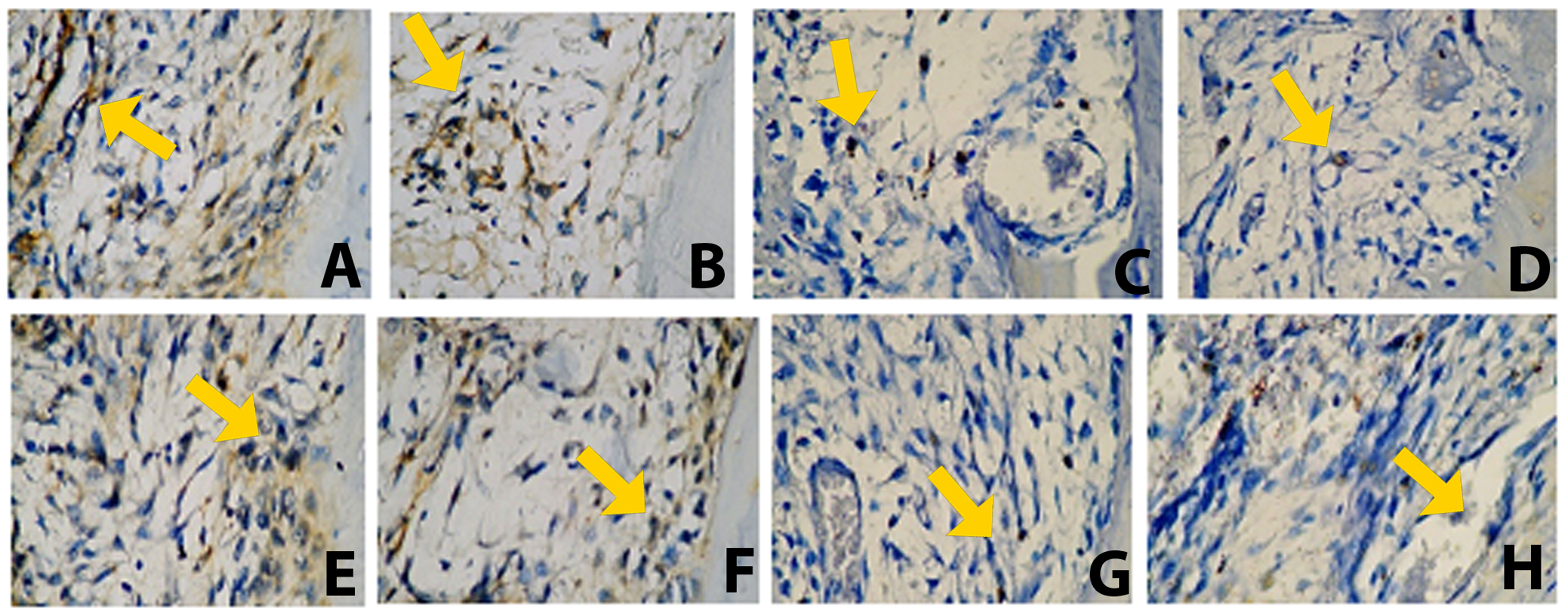
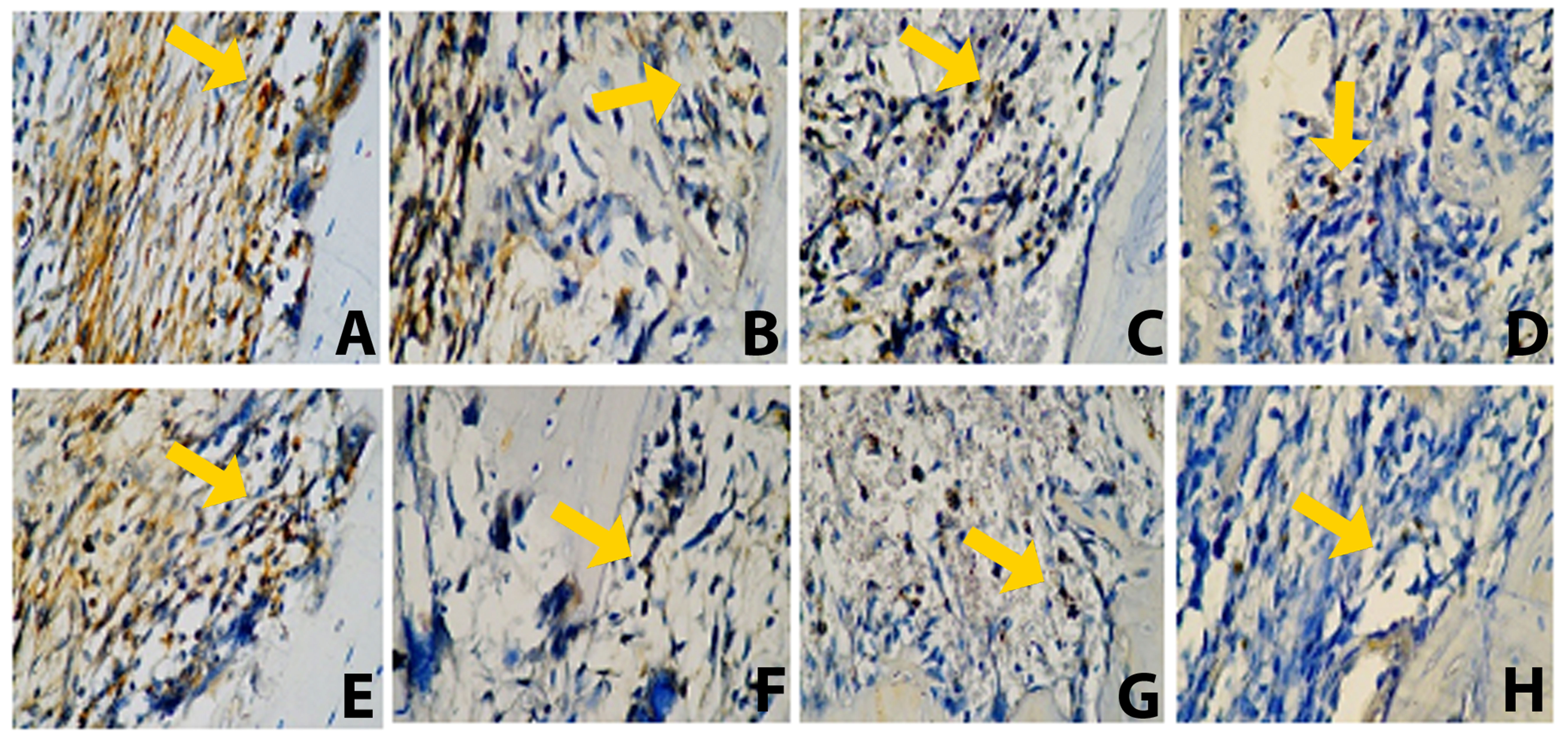
NF-κB expression in osteoclasts demonstrated a pattern similar to that of TLR2 and TNF-α on both day 7 and day 30. The control group exhibited the highest NF-κB expression, followed by the BX group, whereas the PE–BX composite group showed the lowest expression levels. All groups demonstrated a reduction in expression from day 7 to day 30. Statistical analysis revealed that the PE and PE–BX groups differed significantly from the control group (p < 0.001). The PE group also differed significantly from the BX group (p < 0.001), as did the BX group from the PE–BX group (p < 0.001). On day 30, a significant difference was observed between the PE and BX groups (p < 0.01). However, no significant differences were found between the PE and PE–BX groups (Figure 1). Representative histological images of the apical third of the extraction socket are shown in Figure 2. The yellow arrow indicates osteoclasts along the bone margin exhibiting positive NF-κB expression.
TLR2 expression
On day 7, the PE and PE–BX groups differed significantly from the control group (p < 0.05 and p < 0.001, respectively). Similarly, on day 30, both the PE and PE–BX groups showed significant differences as compared to the control group (p < 0.01 and p < 0.001, respectively). The control group exhibited the highest TLR2 expression, whereas the PE–BX group demonstrated the lowest expression levels. On both day 7 and day 30, the PE–BX group differed significantly from the BX group (p < 0.01 and p < 0.05, respectively). However, no significant differences were observed between the control and BX groups (Figure 1). Representative histological images of the apical third of the extraction socket are presented in Figure 3. The yellow arrow indicates osteoclasts along the bone margin showing positive TLR2 expression.
TNF-α expression
On day 7, the PE and PE–BX groups differed significantly from the control group, with p-values of <0.05 and <0.001, respectively. A slight reduction in TNF-α expression was observed in the PE group on day 30. The PE–BX group also showed a significant difference as compared to the control group on day 30 (p < 0.01). Furthermore, the propolis-containing groups demonstrated significant differences in comparison with the BX group on day 30 (Figure 1). Representative histological images of the apical third of the extraction socket are shown in Figure 4. The yellow arrow indicates osteoclasts along the bone margin exhibiting positive TNF-α expression.
Type 1 collagen expression
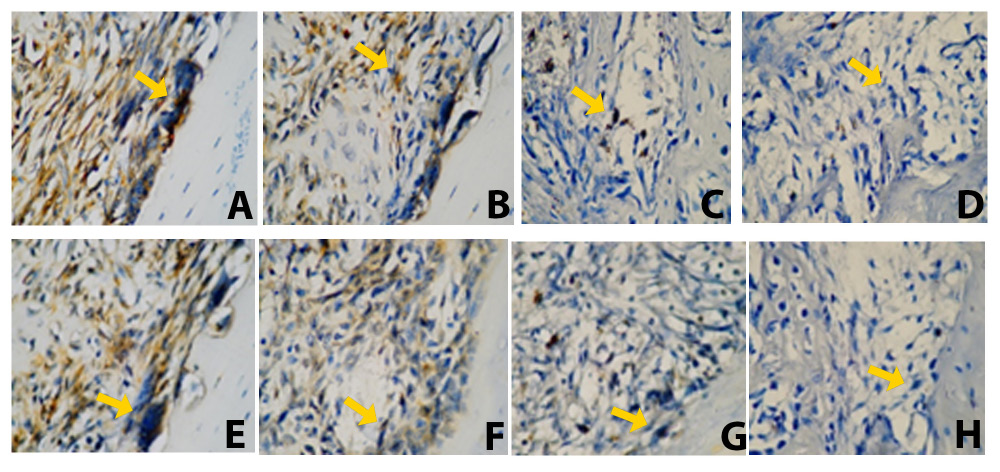
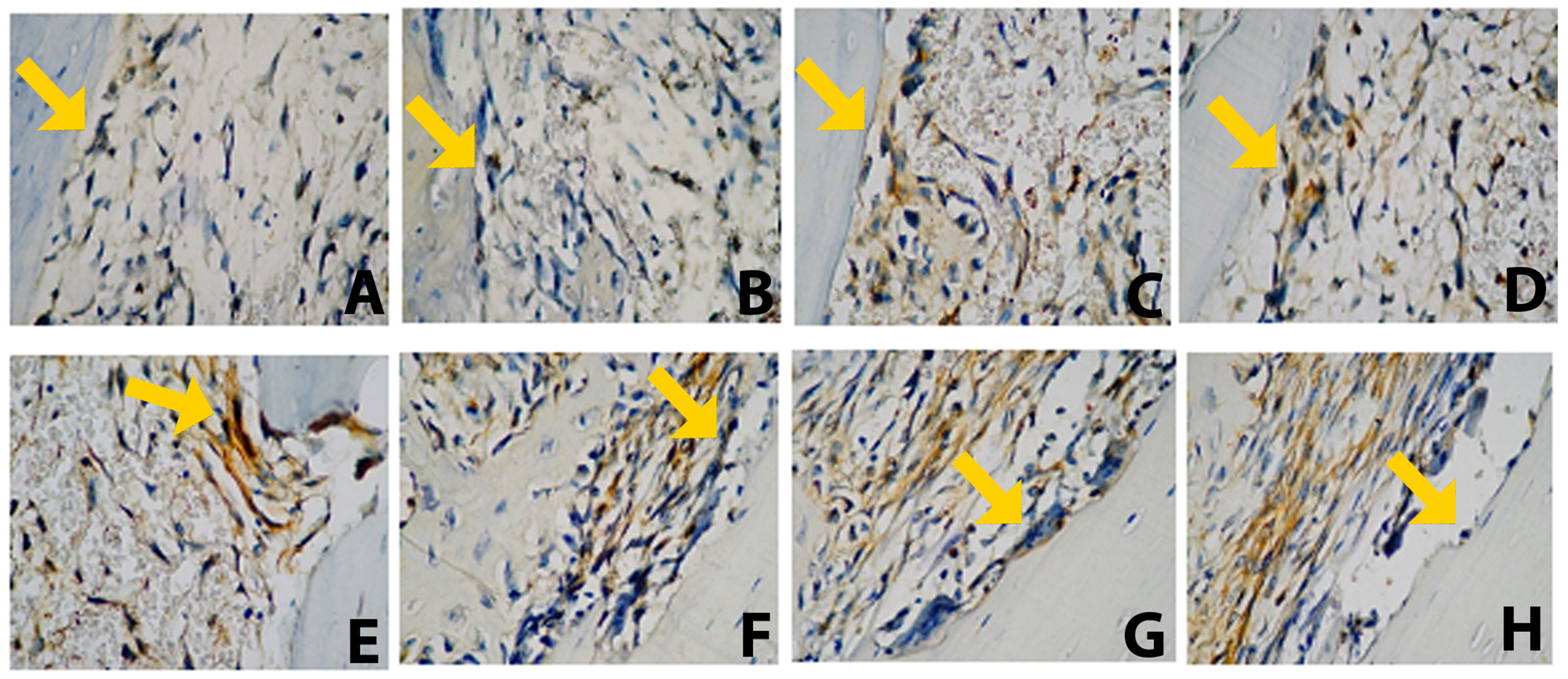
An increasing trend in type I collagen expression was observed in all groups from day 7 to day 30. However, only the PE–BX group showed a significant difference in comparison with the control group (p < 0.001). A significant difference was also identified between the PE and PE–BX groups at the early observation time point (day 7) (p < 0.05). Furthermore, the PE–BX group differed significantly from the BX group at both time points (p < 0.01) (Figure 1). Representative histological images of the apical third of the extraction socket are presented in Figure 5. The yellow arrow indicates osteoblasts along the bone margin exhibiting positive type I collagen expression.
Discussion
The topical application of the ethanol–propolis extract/bovine xenograft composite significantly enhanced bone healing in the tooth socket as compared to the control group. Overall, reduced expression levels of NF-κB, TLR2 and TNF-α were observed, along with increased expression of type I collagen, particularly in the propolis-based groups. No significant differences were found between the 2 propolis-based groups.
This study involved the random allocation of Cavia cobaya into 4 groups – a control group and 3 treatment groups. All animals included in this study were selected according to the specific criteria described in the Methods section.
A similar study by Hozzein et al. investigated the effects of topical propolis application on cutaneous wound healing in diabetic mice.19 Topical propolis demonstrated remarkable wound-healing effects, even in streptozotocin-induced type 1 diabetic mice. The levels of pro-inflammatory cytokines (IL-1β, IL-6, TNF-α, and matrix metalloproteinase-9 (MMP-9)) were significantly reduced, approaching those observed in the non-diabetic control group. Furthermore, propolis promoted type I collagen expression via the transforming growth factor-beta 1 (TGF-β1)/Smad2/3 signaling pathway.19
The remarkable effects of the propolis and bovine bone graft combination applied to the extraction socket were investigated by Kresnoadi et al.20 They reported that this combination increased bone morphogenetic protein-7 (BMP-7) expression and decreased nuclear factor of activated T cells 1 (NFATc1) expression in osteoblasts on days 7 and 14. Significant differences were observed between the combination group and the control group (p < 0.05).20
The propolis extract used in this study contained 2.56% caffeic acid, 1.55% apigenin, 1.28% flavonoids, 0.82% saponins, 1.03% quercetin, and 1.15% terpenoids. Halim et al. compared the composition of Indonesian propolis with its Brazilian counterpart.21 Qualitative analyses showed that the Indonesian propolis contained substantial amounts of flavonoids and phenolic compounds, followed by tannins, essential oils, steroids and triterpenoids, as well as saponins, alkaloids, glycosides, and sugars.21
The principal components of propolis are flavonoids and phenolic acids, including CAPE, which may constitute up to 50% of its bioactive fraction. Flavonoids, one of the largest group of naturally occurring phenolic compounds, are present in nearly all flowering plants and include many of the most common pigments found in plants worldwide. However, the flavonoid profile of propolis differs slightly from that of floral flavonoids due to enzymatic modification and processing by bees during propolis production.
The results showed that the expression levels of NF-κB, TLR2 and TNF-α decreased in all groups by day 30. The PE–BX group exhibited the lowest expression levels, followed by the PE, BX, and control groups, indicating a reduction in inflammatory markers, particularly after socket filling with the PE–BX combination. This effect may be attributed to the complex chemical composition of propolis, which contains phenolic compounds such as cinnamic acid. According to Conti et al., cinnamic acid can downregulate TLR2, human leukocyte antigen – DR isotype (HLA-DR), and cluster of differentiation 80 (CD80) expression while upregulating TLR4 expression in human monocytes.22
Flavonoids (such as quercetin, galangin and pinocembrin), along with caffeic acid, benzoic acid and cinnamic acid, may act on microbial membranes or cell walls, causing structural and functional damage.23 Parolia et al. characterized propolis as having antibacterial properties due to its ability to inhibit bacterial RNA polymerase.24 In addition, it exhibits immunomodulatory, antioxidant and wound-healing activities, largely attributable to its capacity to neutralize free radicals. Moreover, propolis contains trace elements such as iron and zinc, which are essential for collagen synthesis. Aromatic compounds present in propolis, including caffeic acid, are known to possess antimicrobial, antibacterial, anti-inflammatory, immunomodulatory, and hepatoprotective properties.24
Type I collagen plays a crucial role in osteogenesis, as it is assembled into fibers that form the structural and mechanical scaffold of bone.25 It is also the most abundant organic component of the bone matrix.
In general, the molecular mechanisms that may occur following propolis administration include: (1) reduced expression of RANKL and the inhibition of the macrophage colony-stimulating factor (M-CSF)-induced RANK signaling pathway, leading to suppressed differentiation of osteoclast precursor cells; and (2) the upregulation of osteoblast-related gene markers, including Alp, Runx2, Osx, Col-1, thereby promoting osteoblast proliferation, differentiation and mineralization.
Flavonoids and CAPE in propolis are known to decrease osteoclast numbers while increasing osteoblast populations. They also exhibit anti-inflammatory effects by reducing the levels of pro-inflammatory cytokines.26 Similarly, Iqbal et al. demonstrated that a 40% propolis extract reduced osteoclast numbers in guinea pigs by day 14 after tooth extraction.27 This effect is attributed to the high flavonoid content in the 40% propolis gel, which inhibits osteoclastogenesis while promoting osteoblast maturation and enhancing bone remodeling activity.27
The study demonstrated promising results for the presented combination material. However, it was limited by the observation period, which lasted only 30 days, rather than extending until the complete healing of the alveolar bone and the full resolution of the lesion.
Conclusions
The administration of a combination of a natural propolis extract and a bovine bone graft during the extraction socket healing process can enhance type I collagen expression while reducing the NF-κB, TLR2 and TNF-α levels, thereby promoting alveolar bone regeneration.
Ethics approval and consent to participate
This research was approved by the Ethical Clearance Commission at the Faculty of Dental Medicine of the Airlangga University, Surabaya, Indonesia (approval No. 014/HRCC FODM/III/2018.)
Data availability
The datasets supporting the findings of the current study are available from the corresponding author on reasonable request.
Consent for publication
Not applicable.
Use of AI and AI-assisted technologies
Not applicable.